Population study:
We analyze pre-treatment CT scan of histologically proven stage IV lung adenocarcinoma patients who tested positive for EGFR mutations (exons 18-21), by polymerase chain reaction and direct DNA sequencing methods.
A total of 68 cases (39 women and 29 men), diagnosed at our institution between 2010 and 2019, were retrospectively reviewed.
[Table 1] resumes CT features analyzed:
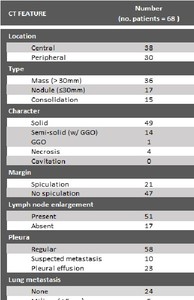
Table 1: CT Imaging features among EGFR-mutant patients (n = 68).
Primary tumor:
- Main tumors were evaluated for location (central or peripheral), margins (spiculated/non spiculated) and morphology, being divided into three types, specifically nodular lesions (≤30mm), masses (>30mm) and consolidations. Additionally, internal features like cavitation or necrosis were recorded.
- Centrally located tumors, extending to peribronchovascular main structures, represented 56% of total cases. The remaining 44% were peripherally distributed, mostly solid nodules/masses.
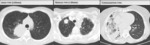
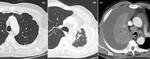
- In 53 of the 68 patients (78%), primary tumors presented as predominant solid masses (53%) and nodules (25%). Almost a quarter of cases (22%) presented with consolidation, often showing ground glass opacity (GGO) component and bilateral involvement. This percentage significantly outgrowed other studies8, where consolidation affected only a minority of EGFR mutated adenocarcinomas [Fig. 2].
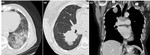
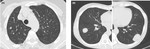
- Previous studies demonstrated that GGO is significantly more prevalent in EGFR-mutated adenocarcinomas comparing to adenocarcinomas without EFGR mutation4,6. In our review pure GGO was not a frequent feature, with most of the GGO lesions presenting as semi-solid tumors. This pattern remits for lepidic-predominant adenocarcinoma, formerly known as non-mucinous bronchoalveolar carcinoma [Fig. 3].
- In our cohort, tumor necrosis was rare (being present in only 3 tumors, none of which showing cavitation), even in locally advanced disease, which may be attributable to angiogenic growth factors [Fig. 3].
- Lesion margins were regular or lobulated in 69%; the remaining 31% presented spiculated margins.
Lymph node enlargement:
- We determined a 10 mm cut-off in short axis (12mm for infracarinal lymph nodes) for targeting highly suspicious metastatic lymph nodes, corroborated by decreased lymph node size after therapy.
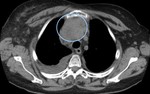
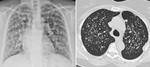
- Three quarters of patients showed mediastinal or hilar lymph node enlargement, some of them with areas of necrosis/liquefaction. Exuberant or conglomerate lymph nodes were not a frequent feature [Fig. 4], even in patients with pulmonary metastasis or tumor bilaterality.
Pleural involvement [Fig.5]:
- Pleural metastasis, presenting as pleural nodularity, were found in 10 patients (15%), a lower incidence comparing with previous study7. However this number may be underestimated, since it was difficult to differentiate pleural from peripheral lung involvement in patients with disseminated lung metastasis.
- Although pleural effusion was almost universal with disease progression, it was observed in only 23 of 68 patients at diagnosis.
Pulmonary dissemination:
- For classification purposes, we divided lung metastasis into disseminated and non-disseminated. Disseminated was subdivided into miliary (≤5 mm) and nodular scattered (>5 mm). Non-disseminated metastasis included unique or multiple but sparse secondary lesions [Fig.6].
- A miliary pattern is defined as having profuse, tiny and innumerable pulmonary micronodules, generally uniform in size (≤5 mm), diffusely distributed throughout the lungs [Fig.7]. The scattered nodular pattern was similar, but with slightly larger nodules, with 2–10 mm in diameter. [Fig.8]. Both represent hematogenic spread and are an uncommon phenomenon in the setting of lung adenocarcinoma8.
- A total of 44 patients presented with lung metastasis, the majority with sparse nodular pattern (44%). 21% presented disseminated military and scattered nodular pattern, overlapping prior clinical- and population-based studies1,2,5,6 suggesting a higher incidence in EGFR mutated carcinoma comparing to non-mutated EGFR tumors. This has been attributed to angiogenic growth factors produced by EGFR mutated tumors.
- This finding of diffuse lung nodules requires differential diagnostic consideration, as it may be seen in numerous infectious and inflammatory etiologies (tuberculosis, histoplasmosis, silicosis, sarcoidosis) an in the setting of metastatic disease from primary thyroid cancer, renal cell carcinoma and melanoma. The concomitant presence of a dominant primary lung mass or nodule is highly suggestive of an EGFR mutated lung adenocarcinoma.
- EGFR mutated tumors with miliary metastasis included exon 19 insertions/deletions and exon 21 L858R mutations, with one patient showing the rarer exon 20 S768I mutation, overlapping previous studies2. Exon 20 mutated EGFR is irresponsive to EGFR-TKI, being associated with dismal prognosis3.