Patient Selection:
A retrospective review of all TACE performed between Jan 2010 and Dec 2017 was performed. (Figure 1)

Fig. 1
References: Department of Radiology, Queen Elizabeth University Hospital, Glasgow 2020
Patients receiving TACE for the first time were then selected out.
From this group, any patient who had previous treatments were excluded.
To allow for same modality imaging comparison, only patients with pre- and post-treatment multi-phase CT imaging within 3 months of the treatment were included in the final cohort, giving a study sample size of 110 patients.
There was no significant difference in survival between the group with satisfactory pre-/post-treatment imaging and non-satisfactory imaging. (Figure 2)

Fig. 2
References: Department of Radiology, Queen Elizabeth University Hospital, Glasgow 2020
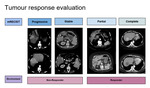
Tumour Response Evaluation
Tumour response was evaluated based on two categories (Figure 3)

Fig. 3
References: Department of Radiology, Queen Elizabeth University Hospital, Glasgow 2020
1) modified Respose Evaulation Criteria in Solid Tumours (mRECIST)
- Complete response: No tumour enhancement in target lesion
- Partial response: >= 30% reduction of the sum of diameters of viable (arterial enhancement) target lesion
- Stable disease: Features classified as neither partial or progressive disease
- Progressive disease: >= 20% increase in sum of diameter of viable target lesion
2) Dichotomised Responder versus Non-Responder
- Responder: Complete and Partial response
- Non-Responder: Stable and progressive disease
Statistical Analysis
To allow for common statistical procedures, analysis was restricted to examination of the index HCC that was defined as the largest tumour. (if more than 1 tumour was present)
ANOVA used to compare means between groups Kaplan Meier survivor curves for patient survival. COX proportional hazard regression used for multivariable adjusted analysis and to estimate survival curves adjusting for demographic and clinical baseline characteristics