Complications after liver transplantation can be classified in vascular, biliary and others, which include: infection, abscess, hematoma, neoplasm, cirrhosis and bowel perforation.
We used several illustrative cases from our Ultrasound and Abdominal Imaging groups in order to demonstrate the main complications after liver transplantation in different imaging modalities, including:
- Hepatic artery stenosis;
- Portal vein thrombosis;
- Hepatic artery thrombosis;
- Inferior vena cava thrombosis of the donor;
- Inferior vena cava stenosis;
- Biliary cast;
- Chronic transplant ischemia;
- Hepatic artery aneurysm;
- Acute peripheral ischemia;
- Biliary fistula;
- Choledocholithiasis;
- Cholangitis;
- Biloma;
- Portal vein stenosis;
- Choledochal stenosis.
1. Vascular complications
Vascular disorders are common and have an incidence of 9%. Early detection is critical to allow graft viability.
1.1. Hepatic artery
Hepatic artery disorders include stenosis, thrombosis and pseudoaneurysm. On Doppler US, the normal hepatic artery shows a rapid systolic upstroke and a continuous diastolic blood flow. The resistive index of a normal hepatic artery varies from 0,5 to 0,8. In the early postoperative period (less than 72 hours after transplantation), increased hepatic artery resistance (resistive index greater than 0,8) is a frequent finding, but resistance ordinarily returns to a normal level within a few days. Increased hepatic artery resistance is associated with older donor age and a prolonged period of ischemia.
Stenosis
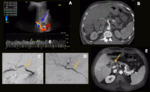
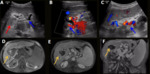
Hepatic artery stenosis usually appears at the site of the anastomosis and within 3 months after transplantation. Early diagnosis is essential for saving the graft since it can lead to hepatic ischemia, artery thrombosis, biliary stricture, sepsis and graft loss. Doppler US is the first imaging modality to be performed, as it can depict any focal increase (more than 2 to 3 times) in peak systolic velocity at the site of stenosis and turbulent flow (Fig.1).
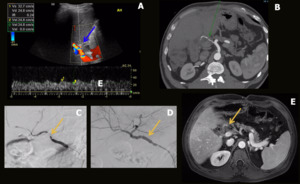
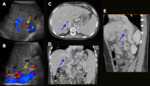
Fig. 1: Hepatic artery stenosis. Male patient, 62 years old, performed liver transplantation for chronic liver disease by B-virus and hepatocarcinoma. Control US detected tardus parvus pattern in the distal segment of the hepatic artery in the hilum associated with low resistivity indexes (RI = 0,24) (blue arrow in A). A CT performed on the same day showed a patent hepatic artery but with marked stenosis on the anastomotic topography (green arrow in B). Stent angioplasty was performed (yellow arrows in C and D). Control MRI showed patent hepatic artery with the stent on anastomotic topography (yellow arrow in E).
Thrombosis
Hepatic artery thrombosis represents one of the most feared complications after liver transplantation, as it can lead to biliary ischemia, necrosis and fulminant hepatic failure. Doppler US shows an absence of flow in the hepatic artery with intraluminal hypoechogenic material. Angiographic CT allows better evaluation of parenchymal and vascular structures (Fig. 2) and represents a noninvasive alternative method to conventional angiography. MRI is also an accurate modality for evaluating hepatic vessels but lasts too long and requires apnea.
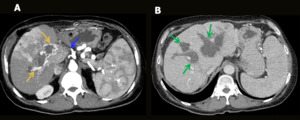
Fig. 2: Hepatic artery thrombosis. Male patient, 55-year-old, performed liver transplantation for chronic liver disease. Control CT showed hepatic artery thrombosis (blue arrow in A) with thin intra and extrahepatic collateral arteries (yellow arrows in A), associated with extensive areas of perfusion disorder (A). Diffuse intrahepatic bile duct necrosis with heterogeneous content (green arrows in B).
Ischemia and infarction
Ischemia and infarction occur after liver transplantation because collateral vessels are usually ligated during surgery. They are more associated with hepatic artery complications than portal vein occlusion and can lead to infection, abscesses and sepsis (Fig.3).
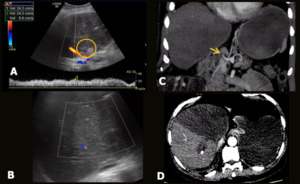
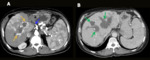
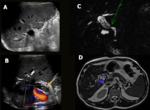
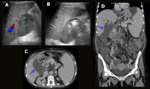
Fig. 3: Hepatic artery and right portal branch thrombosis + parenchymal ischemia (post-transplantations – living donor). A 60-year-old male patient underwent liver transplantation (right lobe – living donor). He presented with abdominal pain and fever. Abdominal US showed absence of flow in the hepatic artery (A), as well as in the right portal branch, associated with parenchymal textural alteration in the medial segments, suggesting ischemia (B). CT showed complete thrombosis of the hepatic artery (yellow arrow in C) and the right anterior portal branch, associated with extensive ischemia / infarction zone of the parenchyma in segments V, VI and VIII of the graft.
Pseudoaneurysm
This complication is relatively rare and occurs at the site of anastomosis. On US, it appears as a cystic lesion, near the normal site of the hepatic artery with turbulent arterial flow. Contrast-enhanced CT and MRI show an enhanced arterial lesion (Fig. 4).
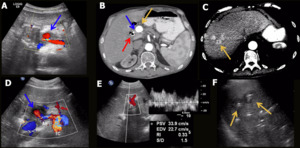
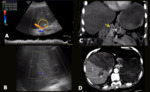
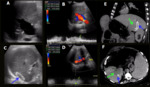
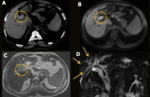
Fig. 4: Hepatic artery aneurysm. Male patient, 55 years old, chronic liver disease by C-virus, underwent a double transplantation (liver + kidney). Two months later he performed a control US that showed saccular aneurysm of the hepatic artery (blue arrow in A) near the hepatic hilum measuring 1,5 cm. Stent angioplasty was performed, unsuccessfully. A CT scan performed two months later showed critical stenosis (blue arrow in B) associated with pseudoaneurysm (yellow arrow in B) of the hepatic artery and cholangiolitic abscesses in the right hepatic lobe (yellow arrow in C). A biliary prosthesis is also observed (red arrow in B). Control US performed one month later evidenced the hepatic artery with signs of critical stenosis / occlusion and pseudoaneurysm in the hepatic hilum region (blue arrow in D). Arterial flow in the left hepatic lobe was of low resistivity (RI = 0,3) (E). No intrahepatic arterial flow was characterized in the right lobe. Liver abscesses were still present (yellow arrows in F).
1.2. Portal vein
Portal vein disorders are infrequently and include thrombosis (Fig. 5), aneurismal dilation (Fig. 6) and stenosis (Fig. 7). Stenosis can be detected by mode-B US, showing poststenotic dilation and increase in number or caliber of collateral vessels (portal hypertension); and by Color and Pulsed Doppler, depicting focal aliasing with increased velocities. Thrombosis is characterized by an echogenic filling defect in the portal vein. Color or power Doppler images and pulsed Doppler waveforms may show a lack of portal venous flow. CT or MRI angiography is an excellent method for detection of filling defects and focal narrowing of the portal vein.
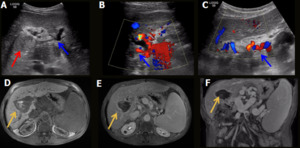
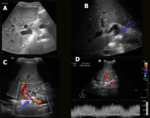
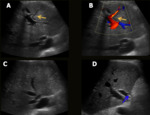
Fig. 5: Aneurysmal portal vein tromboses. Male patient, 70 years old, performed liver transplantation for chronic liver disease by C-virus and hepatocarcinoma. Control US was performed, showing a tapered portal vein in the hilum (blue arrow in A) with multiple adjacent collaterals (blue arrow in C), aspect suggestive of cavernomatous transformation but with hepatopetal flow and normal velocity (blue arrow in B). Right portal branch thrombosis was also evidenced (red arrow in A). An MRI was performed, showing partial subacute thrombosis of the portal trunk and total subacute thrombosis of the right portal branch (yellow arrows in D-F).
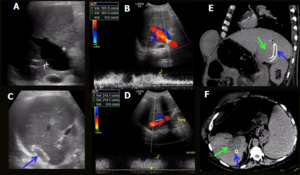
Fig. 6: Portal vein aneurysmal dilation + TIPS. Male patient, 65-year-old, performed liver transplantation for chronic liver disease by C-virus and hepatocarcinoma . Control US showed a dilated portal vein (caliber = 3,5 cm - A), with hepatopetal flow and increased velocity (100 cm/s - B). TIPS stent (C) was patent with normal flow and speed of 214 cm/s (D). CT showed the TIPS (blue arrows) communicating aneurysmal dilation of right portal vein with right hepatic vein (green arrows).
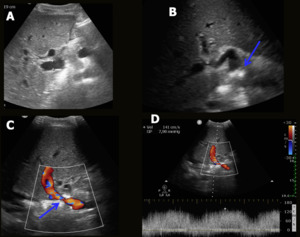
Fig. 7: Portal vein stenosis. Female patient, 24-year-old, performed liver transplantation for Budd-Chiari syndrome. Control US depicted a focal area of narrowing in the postanastomotic segment of the portal vein (blue arrows), with hepatopetal flow and increased velocity (141.0 cm/s).
1.3. Inferior vena cava (IVC) and hepatic veins
These complications are relatively rare and include thrombosis and stenosis, usually at the site of surgical anastomosis. Stenosis (Fig. 8) can be detected on B-mode US as a focal stricture and turbulent flow with increased velocity on pulsed Doppler. A triphasic or biphasic waveform may help exclude substantial hepatic vein stenosis. Thrombosis (Fig. 9) can be depicted as an intraluminal echogenic thrombus with no flow on Doppler US. CT and MRI are used when US is normal or inconclusive and can show additional findings, such as a mosaic pattern of perfusion (characteristic of Budd-Chiari syndrome).
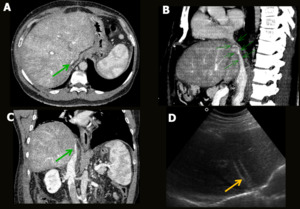
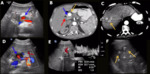
Fig. 8: IVC stenosis. Male patient, 60-year-old, performed liver transplantation for chronic liver disease. Control CT showed
stenosis of the retrohepatic portion of the inferior vena cava (green arrows in A-C), mainly in the anastomosis plane, associated with diffuse hepatic perfusion disorder with mosaic pattern. Stent placement was performed by radiointervention. Control US showed patent stent inside the medium hepatic vein (yellow arrow in D) and extending to the suprahepatic portion of the inferior vena cava.
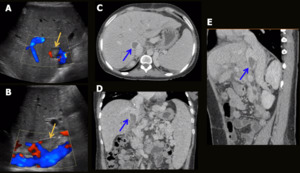
Fig. 9: Donor IVC thrombosis. Male patient, 61-year-old, performed liver transplantation 3 months ago for autoimmune cirrhosis. Control US depicted inferior vena cava with intraluminal thrombus (2 cm) in the portion near the hepatic graft (donor), with preserved flow in the anastomosis region (yellow arrows in A and B) CT showed partial hematic thrombosis of the most caudal portion of the inferior vena cava of the graft (blue arrows in C-E).
2. Biliary complications
Biliary disorders are frequent and usually occur within 3 months after surgery. Obstruction, stenosis (Fig. 10), biliary cast (Fig. 11), stone formation (Fig. 12), fistula (Fig. 13), bile leak, ductal ischemia, dysfunction of the Oddi sphincter and recurrent biliary diseases are the most common complications. The best imaging modalities in the first three months after transplant are US and cholangiography. MRI cholangiography should be used after the removal of biliary catheters and represents the best noninvasive method for evaluation of the biliary tree.
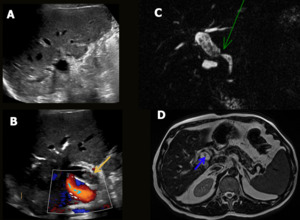
Fig. 10: Biliary anastomosis stenosis. Male patient, 61-yearold, performed liver transplantation for chronic liver disease. Control US depicted moderate dilation of the intra and extrahepatic bile ducts (A), with multiple small stones and thick content in the biliary tree, more evident in the hepatocoledoc (yellow arrow in B) associated with aerobilia. CholangioMR showed biliary anastomosis stenosis (green arrow in C) with moderate bile duct dilatation and multiple intra and extrahepatic bile duct stones (blue arrow in D).
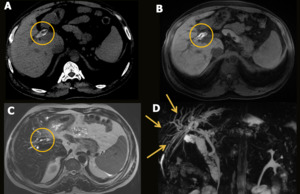
Fig. 11: Biliary cast. Male patient, 45 years old, performed liver transplantation for chronic liver disease. He presented arterial anastomosis stenosis as an acute complication. Control CT showed hyperdense material inside the central intrahepatic bile ducts (A). An MRI was performed, showing
T1 hyperintense material shaping the central intrahepatic bile ducts (B) with low sign in T2 images (C), probably corresponding to biliary cast, associated with moderate dilation of the intrahepatic bile ducts (cholangioRM - D).
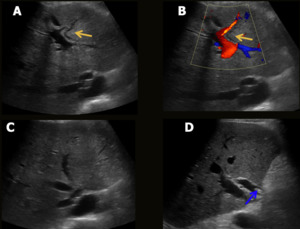
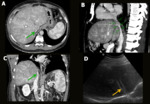
Fig. 12: Choledocolithiasis. Male patient, 58-year-old, performed liver transplantation for chronic liver disease. Control US depicted moderate dilation of the intra (A-C) and extrahepatic (D) bile ducts, with choledocolithiasis (blue arrow in D).
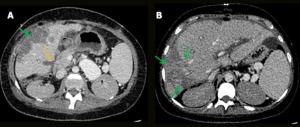
Fig. 13: Biliary fistula and peripheral parenchymal ischemia. Male patient, 18-year-old, performed liver transplantation for biliary atresia. Control CT showed a linear hypoattenuating area in the medial edge of segment V, suggestive of biliary fistula (yellow arrow); and hypoattenuating peripheral lesions more evident in the right lobe, some of them liquefied, representing ischemic areas (green arrows).
3.Others
Transplant patients have a lot of risk factors for malignant tumors, being the most relevant immunosuppression and chronic viral infection by hepatitis virus, Epstein-Barr, cytomegalovirus and herpesvirus. The most common malignant neoplasms are skin cancers other than melanoma, Kaposi sarcoma and non-Hodgkin lymphoma. Recurrence of hepatocellular carcinoma can also occur, being the most common sites the lung and second the liver.
Fluid collections like seromas and hematomas are common complications and usually occur within the first days after surgery during few weeks. US is very sensitive, but no specific, as blood, bile, pus and lymph have the same appearance. In doubtful cases, CT or MRI could be helpful in differentiating the type of fluid (Fig. 14).
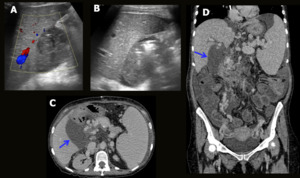
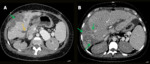
Fig. 14: Fluid collection. Female patient, 54-year-old, performed liver transplantation for chronic liver disease. 20 days after the transplant, she presented abdominal pain and fever. Control US depicted a loculated collection with thickened and heterogeneous content, located near the hepatic hilum / gallbladder site (A). There were gaseous foci inside the collection with no change in position during the exam (B). CT showed a loculated collection near the hepatic hilum, in contact with duodenal wall (blue arrows in C and D).
It is important to remember that rejection is the most common cause of graft failure, but imaging modalities as well as clinical and laboratory findings are nonspecific and play no role in this diagnosis. A definite and accurate diagnosis is performed by histological analysis.