Although controversial and constantly evolving, pulmonary aspergillosis can be subdivided into five main categories, each with different risk groups, clinical manifestations and radiographic findings:
- Aspergilloma (saprophytic or non-invasive aspergillosis), which arises in abnormal airways or pulmonary cavities and is the most common form;
- Allergic bronchopulmonary aspergillosis (ABPA), which affects patients with asthma and cystic fibrosis;
- Chronic necrotizing aspergillosis (or semi-invasive) described in patients with chronic pulmonary pathology and mild immunodeficiency;
- Invasive aspergillosis associated with markedly immunocompromised patients, which can be divided into two forms:
- Angioinvasive aspergillosis;
- Bronchopneumonic aspergillosis (airway-invasive).
Chest computed tomography is the imaging method of choice, as it allows the evaluation of the pulmonary parenchyma and the detection of features of fungal disease.
Aspergilloma (saprophytic aspergillosis):
Saprophytic aspergillosis is a fungal infection without tissue invasion and occurs in immunocompetent host.
It usually occurs in a pre-existing pulmonary cavity or ectatic bronchus. Pathologically, the fungus grows inside the cavity, creating a ball of hyphae, mucus and cellular debris. They are usually solitary lesions, most often in the upper lobes.
The most common underlying causes are tuberculosis, sarcoidosis, emphysema and occasionally bronchogenic cyst and pneumatoceles secondary to Pneumocystis jirovecii pneumonia in patients with acquired immunodeficiency syndrome.
These patients are often asymptomatic. Hemoptysis is the most common clinical manifestation (reports of up to 40%) and may be life-threatening.
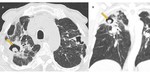
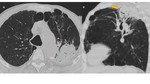
In CT, aspergilloma is characterized by a solid mass with a soft-tissue density of round or oval morphology that arises within a pulmonary cavity (Fig. 1). This fungus ball may be mobile. Aspergilloma is often associated with thickening of the cavity wall and adjacent pleura.
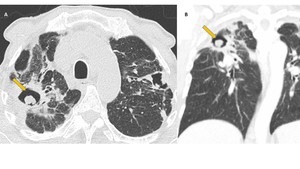
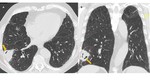
Fig. 1: Saprophytic aspergillosis. Axial (A) and coronal (B) CT images of a patient with a history of pulmonary tuberculosis. Note the presence of two cavitated lesions (in both upper lobes). The right lobe lesion has a solid mass inside, compatible with aspergilloma (arrow) and shows “air crescent” sign. There is thickening of the cavity walls and adjacent pleura.
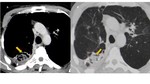
The “air crescent” sign is a typical sign of aspergilloma (Fig. 2), in which the mass separates from the pulmonary cavity wall by the airspace of variable size and shape. Occasionally the fungus ball may occupy the entire cavity without evidence of the “air crescent” sign. Angioinvasive aspergillosis may also have increasing air sign and rarely lung abscess and cavitated tuberculosis.
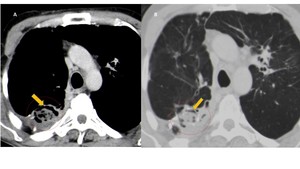
Fig. 2: Aspergilloma. Axial CT images show a cavitated lesion in the right upper lobe with a solid mass with soft tissue density and round morphology inside (circle). Presence of air crescent sign (arrow) and thickening of the adjacent pleura.
Treatment varies depending on the clinical condition of the patient. Patients with episodes of severe haemoptysis may require surgical removal of the cavity or embolization of the feeding vessels.
Prognosis is generally good and approximately 10% resolve spontaneously.
Allergic Bronchopulmonary Aspergillosis (ABPA)
Allergic bronchopulmonary aspergillosis (ABPA) represents a complex hypersensitivity reaction to inhaled Aspergillus spores (most often Aspergillus fumigatus), occurring almost exclusively in patients with asthma (70- 95%) and cystic fibrosis.
Pathologically, this form of aspergillosis is characterized by the presence of plugs of mucus containing Aspergillus organisms and eosinophils.
A severe type I hypersensitivity reaction with bronchial wall edema and bronchospasm is triggered by IgE release.
The subsequent inflammatory response leads to the deposition of immune complexes and inflammatory cells in the bronchial mucosa, causing damage to the bronchial wall, with bronchiectasis and pulmonary fibrosis.
Excessive mucus production and abnormal ciliary function lead to mucoid impaction.
Clinically it manifests with cough, wheezing and fever (non-specific symptoms). Eosinophilia and elevated serum IgE levels are typically found in ABPA.
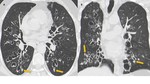
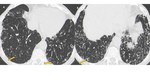
ABPA findings on CT include the presence of central saccular bronchiectasis (Fig. 3). Tubular or saccular “finger-in-glove” areas of increased opacity in a bronchial distribution representing mucus plugging within bronchiectasis, predominantly involving the upper lobes and they are highly suggestive of ABPA and is considered the hallmark of the disease.
Bronchial wall thickening is common.
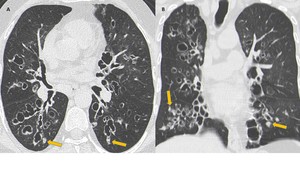
Fig. 3: ABPA. Axial (A) and coronal (B) CT images show multiple varicose bronchiectasis scattered throughout the parenchyma. Note the parietal thickening of bronchiectasis and mucoid impaction (arrows).
In chronic untreated cases, it may progress to pulmonary fibrosis, predominantly in upper lobes.
Alveolar pulmonary opacities may arise (due to deposition of immune complexes and inflammatory cells within the lung parenchyma).
Semi-invasive (Chronic Necrotizing) Aspergillosis
Semi-invasive aspergillosis (also known as chronic necrotizing aspergillosis) is histologically characterized by the presence of tissue necrosis and granulomatous inflammation.
Characteristically, patients susceptible to semi-invasive aspergillosis are mildly immunocompromised or have chronic diseases that predispose them to infection.
Risk factors include chronic debilitating diseases, advanced age, diabetes mellitus, malnutrition, alcoholism, prolonged corticosteroid therapy, and chronic obstructive pulmonary disease.
Progression is slow over months or years.
Clinically, it is an insidious disease and manifests with chronic cough, sputum production, fever and constitutional symptoms. Haemoptysis may also occur.
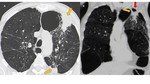
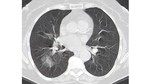
Initially, semi-invasive aspergillosis manifests with unilateral or bilateral segmental areas of consolidation in the upper lung fields, associated with pleural thickening (Fig. 4). Areas of internal necrosis may then arise, with the sign of air crescent (Fig. 5) and multiple nodular areas of increased opacity.
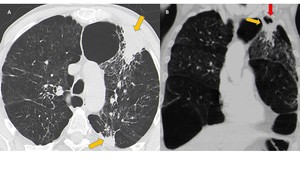
Fig. 4: Semi-invasive Aspergillosis. Axial (A) and coronal (B) CT images of a patient with chronic obstructive pulmonary disease with complaints of hemoptysis. In the left upper lobe there are two areas of consolidation (arrows), the anterior segment with cavitation area (red arrow) and with associated pleural thickening. Note the presence of thickening of some bronchial walls and multiple nodular areas of increased opacity. Areas of centrilobular emphysema are also observed.
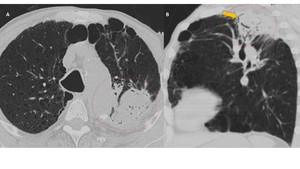
Fig. 5: Semi-invasive (Chronic Necrotizing) Aspergillosis. Axial (A) and sagittal (B) CT images of an alcoholic patient with chronic obstructive pulmonary disease. There is an area of consolidation in the left upper lobe with small areas of necrosis (early cavitation), outlining the air crescent sign (arrow). Note the presence of associated pleural thickening and emphysema (related to the underlying disease).
At CT, necrotizing aspergillosis also manifests with bronchial wall thickening and bronchial obstruction. In practice, the diagnosis is made through a bronchoscopic biopsy, which shows tissue invasion.
Treatment options depend mainly on the patient's status. In patients with hemoptysis, surgical resection of the lesion provides a more definitive therapeutic alternative.
Prognosis is often good, but some report up to 40% mortality.
Invasive pulmonary aspergillosis
It occurs almost exclusively in immunocompromised patients with severe neutropenia and has high rates of death (up to 85%).
Depending on its route of spread, invasive aspergillosis can be divided into two forms: angioinvasive aspergillosis and airway-invasive aspergillosis.
Airway-invasive Aspergillosis
Airway-invasive aspergillosis is histologically characterized by the presence of Aspergillus deep to the basement membrane of the airways. It is a potentially lethal opportunistic infection. The extent and severity of infection in susceptible individuals are of course predicated on the degree of immunodeficiency.
Clinically, it manifests with acute tracheobronchitis, bronchiolitis, and bronchopneumonia, without evidence of vascular invasion.
Acute tracheobronchitis usually has no imaging manifestations.
Bronchiolitis is characterized on CT by the presence of centrilobular nodules (with random distribution) and linear branching or nodular areas of increased attenuation having a “tree-in-bud” appearance (Fig. 6).
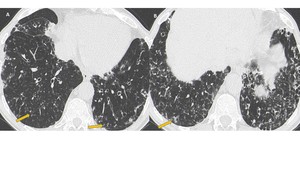
Fig. 6: Airway-invasive aspergillosis in immunosuppressed patient. Presence of multiple centrilobular nodules and linear branching of increased attenuation having a “tree-in-bud” appearance (arrows).
Aspergillus bronchopneumonia manifests with predominantly peribronchial consolidation areas. It is indistinguishable from bronchopneumonias caused by other microorganisms.
Angioinvasive Aspergillosis
Angioinvasive aspergillosis occurs almost exclusively in immunosuppressed patients with severe neutropenia. Histologically, it is characterized by invasion of small to medium-sized pulmonary arteries by fungal hyphae. This leads to thrombus formation with occlusion of the arteries, with consequent tissue necrosis and systemic dissemination.
The clinical diagnosis is difficult and the mortality rate high. Clinically, it manifests as fever, nonproductive cough, pleuritic pain and hemoptysis (nonspecific symptoms).
CT findings consist of small nodules surrounded by a halo of ground-glass attenuation (“halo sign”) (Fig. 7 - 8) and areas of wedge-shaped and pleural-based consolidation (correspond to hemorrhagic infarcts) (Fig. 9), also with the halo sign. The halo sign represents alveolar hemorrhage.
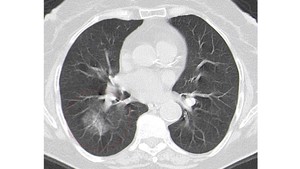
Fig. 7: Angioinvasive aspergillosis. Severely immunosuppressed patient after kidney transplantation. Chest CT shows nodule surrounded by a halo of ground-glass attenuation (“halo sign”), highly suggestive of angioinvasive aspergillosis in the appropriate clinical context.
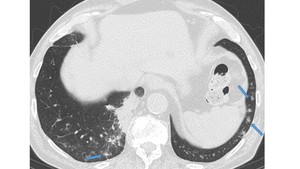
Fig. 8: Angioinvasive aspergillosis in immunocompromised patient. CT image shows multiple small solid nodules surrounded by a halo of ground-glass attenuation (“halo sign”) (arrows) and an area of consolidation in the posterior segment of the right lower lobe.
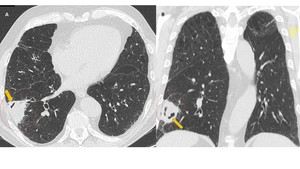
Fig. 9: Angioinvasive aspergillosis under treatment in a neutropenic patient. CT images in axial (A) and coronal (B) show consolidation area with pleural base and wedge-shaped (circle) corresponding to hemorrhagic infarct. Note the presence of “air crescent sign” (arrow), which typically appears after initiation of treatment due to progression to cavitation.
The halo sign is highly suggestive of angioinvasive aspergillosis in severely neutropenic patients. However, it may be associated with other pathologies, for example, Wegener granulomatosis, hemorrhagic metastases and Kaposi sarcoma.
Air crescent sign may appear within the nodule during the convalescence period (2-3 weeks after therapeutic institution), similar to Aspergilloma.
In the right clinical context, nodules or consolidations surrounded by a ground-glass halo, progressing to cavitation or Air crescent sign are considered typical of angioinvasive aspergillosis (Fig. 10).
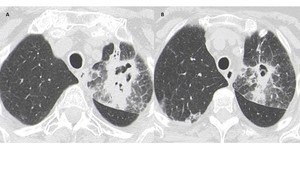
Fig. 10: Angioinvasive aspergillosis in a severely neutropenic patient. In the left upper lobe there is a consolidation with small central cavitation areas and ground-glass surrounding the lesion. Note the presence of interlobular septa thickening and small nodules surrounded by a halo of ground-glass attenuation.
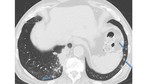
Angioinvasive aspergillosis affects only the lung in about 60% of patients. Due to hematogenous spread, other organs may be affected, including the brain, liver, spleen, kidney, and gastrointestinal tract (Fig. 11).
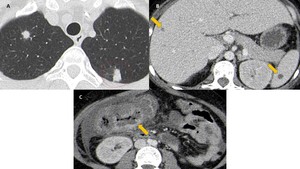
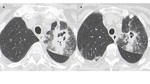
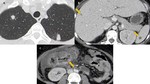
Fig. 11: Angioinvasive aspergillosis in a young patient with aplastic anemia. Presence of pulmonary nodules, some surrounded by a halo of ground-glass attenuation (circle). Some hypodense nodules in the liver and spleen are observed (arrows), suggestive of involvement by aspergillosis. Similarly, there is involvement of the ascending colon with marked parietal thickening and intraperitoneal free fluid.