MRI is the most useful imaging tool in diagnosing viral encephalitis. We used conventional sequences like T1WI, T2WI, FLAIR and DWI in all cases. DWI showed changes even during the early phase of illness. Hypointense changes on T1WI and hyperintense T2/ FLAIR changes appeared later in the disease course. T2* sequences were used to identify haemorrhage. Post contrast T1WI (T1W C+) showed no/minimal enhancement. MRS, MTR & DTI were used in selected cases. For evaluating calcifications, CT was more helpful than MRI.
JAPANESE ENCEPHALITIS:
JE virus, an arbovirus of Flaviviridae family is transmitted through the bite of Culex mosquito. Pigs act as amplifier host.
Imaging features & pathogenesis:
T2/FLAIR hyperintensity with restricted diffusion is noted in thalamus, basal ganglia, midbrain, hippocampus, cerebral cortices, anterior horns of the medulla and cervical spinal cord.
The distinct neurotropism is due to heat shock protein-70 (Hsp-70), a specific neuronal JEV-receptors which mediates viral entry into neurons.
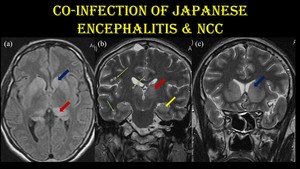
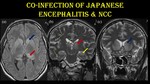
Fig. 5: Co-infection of JE & NCC.
10-year-old girl with serologically positive JE encephalitis with NCC co-infection.
Symmetrical T2 / FLAIR hyperintensities noted in bilateral caudate nuclei (blue arrow), putamen, thalamus (red arrow) and hippocampus (yellow arrow). Vesicular stage (green arrow) of NCC lesions (T2 hyperintense lesions with hypointense central dot) are noted in right peri insular cortex, inferior temporal lobe and bilateral thalamus without surrounding edema.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Co-infection of JE virus and NCC:
Due to similar etiopathogenesis (rearing of pigs), co-infection of JE & NCC is common.
Pigs are efficient amplifiers of the JE virus and the intermediate host of Taenia solium.
Co-infection with Taenia increases the BBB permeability & neuroinvasiveness of JE virus resulting in increased mortality especially in children.
DENGUE ENCEPHALITIS:
Dengue virus is transmitted through bite of Aedes mosquito.
Imaging features & pathogenesis:
Dengue, being a flavivirus also affects bilateral thalamus (double doughnut sign), globus pallidus, brainstem and cerebellum in a symmetrical fashion.
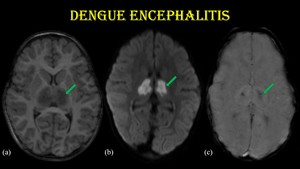
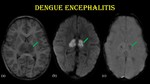
Fig. 6: Dengue encephalitis.
2-year-old male child with H/O fever for 10 days and altered sensorium for 3 days, who tested positive for NS-1 antigen & dengue IgM.
Axial sections of MR images (a, b & c) show symmetrical T1 hypointensity with marked diffusion restriction in bilateral thalamus (green arrow) with GRE blooming predominantly involving the centre of thalamus - “double doughnut sign”.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
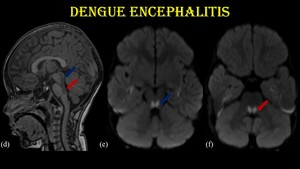
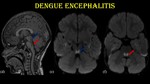
Fig. 7: Dengue encephalitis.
The same patient shows T1 hypointensity with restricted diffusion (d, e & f) in midbrain (blue arrow) and pons (red arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Its neurotropism is due to the specific expression of Hsp-70 & Hsp-90 receptors in the above-mentioned areas.
- Affinity towards similar sites in JE & dengue encephalitis is due to similar neuronal receptors.
CHIKUNGUNYA ENCEPHALITIS:
Chikungunya virus belong to Togaviridae and transmitted by Aedes.
Imaging features & pathogenesis:
Diffusion restricting T2/FLAIR hyperintensity noted in bilateral periventricular and subcortical frontoparietal white matter and corpus callosum.
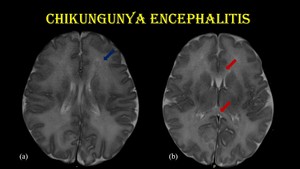
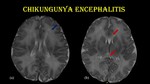
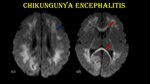
Fig. 8: Chikungunya encephalitis.
8-day-old baby with H/O fever and irritability for 3 days was referred for MRI. Mother had fever, myalgia & headache 5 days prior to delivery and was tested positive for CHIK-IgM.
Axial T2W MR images (a & b) shows hyperintensity in bilateral frontoparietal white matter (blue arrow) & rostrum and splenium of corpus callosum (red arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
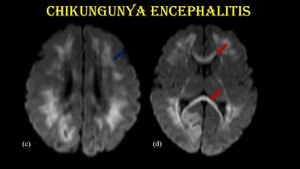
Fig. 9: Chikungunya encephalitis.
DWI shows restricted diffusion in bilateral frontoparietal white matter (blue arrow) & rostrum and splenium of corpus callosum (red arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Concentration of IL-6, IL-8, TNF-α and IFN-α are elevated in CSF and serum.
Astrocytes and oligodendrocytes are mainly affected explaining the selective involvement of white matter
PARECHOVIRUS ENCEPHALITIS:
Parechovirus, nonenveloped RNA virus of Picornaviridae family is transmitted through feco-oral & respiratory routes.
Imaging features & pathogenesis:
Parechovirus selectively affects the periventricular and subcortical white matter in a symmetrical fashion.
Corpus callosum & internal capsule are involved, sparing thalamus, and basal ganglia.
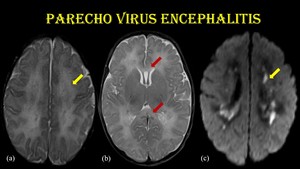
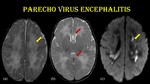
Fig. 10: Parechovirus encephalitis.
3-week-old female child presented with fever & irritability and tested positive for HPeV RNA in CSF by RT-PCR.
Axial T2W images (a & b) show symmetrical hyperintensity in bilateral periventricular and subcortical white matter (yellow arrow) & corpus callosum (red arrow) with restricted diffusion in periventricular white matter (c).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
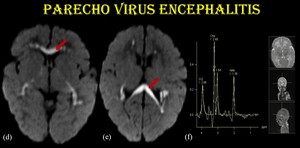
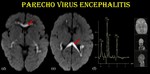
Fig. 11: Parechovirus encephalitis.
Restricted diffusion in corpus callosum (d & e) noted in the same patient. MRS (f) shows reduced NAA and elevated Choline / Creatine peaks.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
The activation of intracellular TLR-7 and 8 by the ssRNA of HPeV results in this selective neurotropism.

Fig. 12: White matter involvement in Parechovirus encephalitis.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
ENTEROVIRUS RHOMBENCEPHALITIS:
Enterovirus also belongs to Picornaviridae with similar modes of transmission.
Imaging features & pathogenesis:
Hyperintense signal changes in T2WI and FLAIR, preferably involving dorsal aspect of pons, medulla & midbrain and dentate nucleus of cerebellum and cervical spinal cord sparing supratentorial regions.
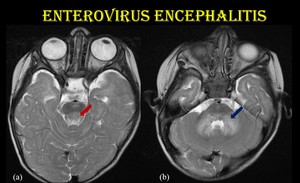
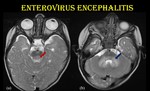
Fig. 13: Enterovirus rhombencephalitis.
3-year-old-female child with H/O fever, vesicular lesions over the dorsal surface of hands and foot, presented with myoclonic jerks with serologically positive EV infection.
Axial T2W images show hyperintense signal changes in the tegmentum of pons (red arrow) and dentate nuclei of cerebellum (blue arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
HUMAN HERPES VIRUS (HHV)
HHVs represent a group of DNA viruses which differ in surface glycoprotein and biological characteristics.
Ø α-viruses - HSV-1, HSV-2 and VZV.
Ø β-viruses - CMV, HHV-6, and HHV-7.
Ø γ-viruses - EBV & HHV-8.
After primary infection, viruses remain latent in neuronal tissues and reactivated when host immunity falls.
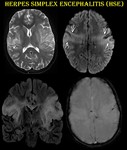
HERPES SIMPLEX ENCEPHALITIS (HSE)
Imaging features & pathogenesis:
Typical bilateral asymmetric involvement of medial temporal, inferior frontal regions, insula and limbic system sparing the basal ganglia. GRE shows blooming in temporal lobe.
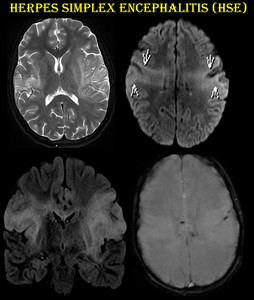
Fig. 14: Herpes simplex encephalitis.
11-year-old female child presented with fever and GTCS.
T2 / FLAIR hyperintensity noted in bilateral peri insular cortex (arrows), inferior frontal gyri, superior and medial temporal lobes with blooming in GRE. She was diagnosed as HSE and started on acyclovir.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
This selective affinity is explained by:
Intranasal inoculation and spread along olfactory pathways into nearby temporal and frontal cortices.
From its latent site in trigeminal ganglia, through tentorial branches of trigeminal nerve, HSV infects the dura of anterior and middle cranial fossae, thereby affecting frontal, temporal lobes and limbic system.
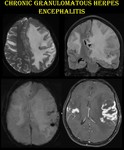
CHRONIC GRANULOMATOUS HERPES ENCEPHALITIS
HSE is an acute illness but some present with relapse and very few present with chronic course.
Early discontinuation/ inadequate antiviral therapy is the cause.
CSF-PCR analysis will be negative for HSV-DNA.
MRI shows cerebral edema, parenchymal calcification and contrast enhancement.
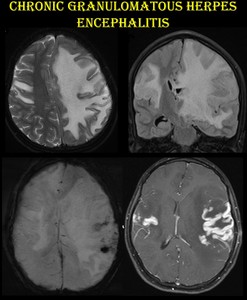
Fig. 15: Chronic granulomatous herpes encephalitis.
The same patient presented after 18 months with intractable seizures and altered consciousness. H/O discontinuation of acyclovir following symptomatic improvement was present.
MRI shows enhancement in bilateral superior temporal and left peri insular regions with blooming and massive edema causing subfalcine herniation to the right.
Biopsy revealed florid granulomatous inflammation with foci of mineralisation and necrosis.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Patients with TLR-3 deficiency prone to develop chronic granulomatous herpes encephalitis.
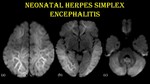
NEONATAL HERPES ENCEPHALITIS:
Vertical perinatal transmission from maternal genital infection (HSV-2).
Imaging features & pathogenesis:
Leptomeningeal inflammation with diffuse parenchymal involvement.
No temporal lobe predilection.
Can involve cerebellum, brainstem, temporal lobes, deep grey nuclei and cortex.
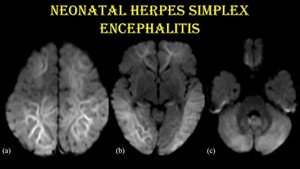
Fig. 16: Neonatal herpes encephalitis.
18-day-old male child with H/O fever and seizure for 3 days & vesicular and crusted lesions in scalp since birth.
DWI (a, b & c) shows restricted diffusion in bilateral frontal, parietal, temporal, occipital and insular regions, bilateral limbic lobes and cerebellum. No evidence of haemorrhage.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
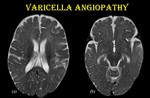
VARICELLA ENCEPHALOPATHY
Imaging features & pathogenesis:
Presents as cerebellitis, vasculitis, meningoencephalitis, nerve palsy and transverse myelitis.
VZV vasculopathy, an important cause of childhood stroke, affects small penetrating vessels.
HPE shows that VZV infects the adventitia and spreads transmurally towards lumen.
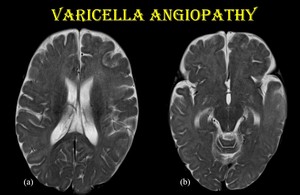
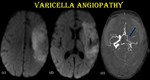
Fig. 17: Varicella angiopathy.
8-month-old female child, K/C/O chicken pox infection on native treatment presented with irritability and paucity of movements on the right side.
Axial T2WI (a & b) show hyperintensity in left frontotemporal and insular cortex.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
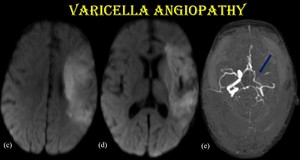
Fig. 18: Varicella angiopathy.
DWI of the same patient shows corresponding areas of restricted diffusion with diffusely narrowed petrous, cavernous & supraclinoid segments of left ICA and left MCA (blue arrow) in MRA.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
CONGENITAL CMV INFECTION
Transplacental transmission. Common in women experiencing primary infection during pregnancy.
Imaging features & pathogenesis:
Gestational age at the time of infection determines the pattern of CNS injury.
· <18 weeks → reduction in neurons and glia, lissencephaly, small cerebellum, ventriculomegaly.
· 18-24 weeks → migration abnormalities (frontal > temporal), ventriculomegaly.
· 3rd trimester → myelin delay or destruction, periventricular cysts.
· Perinatal infection → delay in myelin maturation, focal white matter injury.
- Periventricular calcification is the commonest finding in neonatal CMV infection.
- Anterior temporal cysts with white matter abnormality is specific for CMV infection.
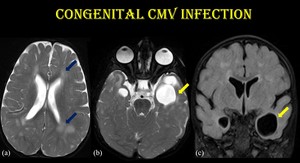
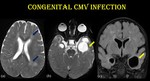
Fig. 19: Congenital CMV infection.
MR imaging features of early fetal CMV infection. Axial T2 and coronal FLAIR images show white matter hyperintensity (blue arrow) and anterior temporal cysts (yellow arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
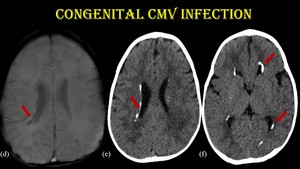
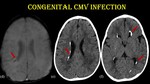
Fig. 20: Congenital CMV infection.
For the same patient, GRE shows periventricular calcifications (red arrow) which are better seen in CT.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
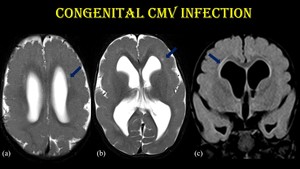
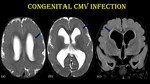
Fig. 21: Congenital CMV infection.
3-month-old infant with ventriculomegaly, pachygyria and subtle T2/FLAIR hyperintensity in periventricular frontal white matter (blue arrow) – suggestive of early fetal CMV infection.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Diagnosis should be made within 3 weeks of life by PCR analysis. After 3 weeks, presence of CMV DNA may indicate perinatal transmission which is benign. Neuroimaging supports the diagnosis in such cases.
ACUTE ENCEPHALOPATHY WITH BIPHASIC SEIZURE AND LATE REDUCED DIFFUSION (HHV-6 INDUCED)
AESD is diagnosed by its clinical and radiological features.
Influenza-A, HHV-6, adenovirus and rotavirus are the common viral causes.
Child presents with prolonged febrile seizure, followed days later by a cluster of complex seizures.
MRI following the initial episode is normal.
Restricted diffusion in bilateral frontoparietal white matter sparing the perirolandic region and basal ganglia is noted after the secondary seizure.
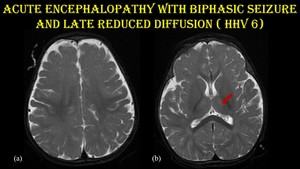
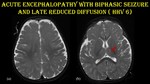
Fig. 22: Acute encephalopathy with biphasic seizures and late reduced restriction.
A 7-month-old male child presented with history of prolonged seizure lasting for 15 minutes following fever with rash for one day. On day 3, the child developed complex partial seizures. MRI was done on day 4. CSF tested positive for HHV-6 B DNA.
Axial T2 sections (a & b) shows subtle hyperintensity in bilateral thalamus (red arrow).
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
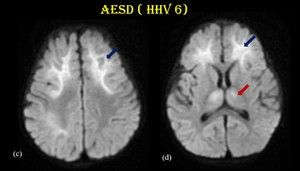
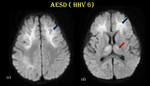
Fig. 23: Acute encephalopathy with biphasic seizures and late reduced restriction.
DWI (c & d) in the same patient shows restricted diffusion involving bilateral frontal (blue arrow), right parietal regions and bilateral anterior thalami sparing the peri rolandic region.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
INFLUENZA VIRUS
Belongs to Orthomyxoviridae family.
- Acute Necrotizing Encephalopathy of Childhood (ANEC)
ANEC affects infants and children and characterized by symmetrical diffusion restricting lesions in thalami, brain stem tegmentum and cerebellum.
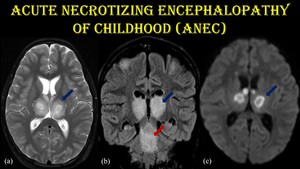
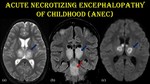
Fig. 24: Acute Necrotizing Encephalopathy of Childhood.
9-year-old male child presented with H/O fever, impaired consciousness and seizure for 4 days. The patient is a biopsy proven case of ANEC. No specific viral etiology could be made out.
(a) Axial T2WI shows hyperintensity in bilateral thalamus (blue arrow). (b) Coronal FLAIR shows hyperintensity in bilateral thalamus and brainstem (red arrow). (c) Diffusion restriction noted in bilateral thalamus in a symmetrical fashion.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
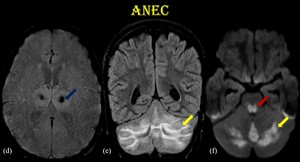
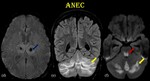
Fig. 25: Acute Necrotizing Encephalopathy of Childhood.
(d) GRE shows blooming in bilateral thalamus. (e) coronal FLAIR shows hyperintensity in bilateral cerebellar hemispheres (yellow arrow). (f) diffusion restriction noted in posterior pons and cerebellum.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Pathogenesis:
Common causes include influenza-A virus, HSV, HHV-6 and mycoplasma.
TNF-1, interleukin-1, and interleukin-6 mediate the disease.
Absence of inflammatory cells in affected brain parenchyma differentiates it from ADEM and acute haemorrhagic encephalitis.
- REversible Splenial LEsion Syndrome (RESLES)
Also known as MERS and CLOCC. Presents as diffusion restricting lesion in splenium of corpus callosum.
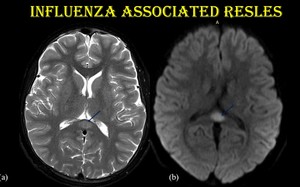
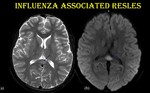
Fig. 26: Reversible splenial lesion syndrome.
5-year-old-female child admitted with disorientation and hallucinations following a 1-day prodromal illness consisting of high fever, cough, and rhinorrhea. Influenza A viral antigen was isolated from nasopharyngeal swab.
Diffusion restricting T2 hyperintensity noted in splenium of corpus callosum.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
Pathogenesis:
Causes include infections (influenza virus, HHV, EBV, adenovirus), drugs, malignancy and trauma.
Following infection, macrophages activate astrocytes which releasing glutamate.
Glutamate via NMDA & AMPA receptors cause influx of sodium, calcium and water resulting in cytotoxic edema.
High density of cytokine & glutamate receptors in Splenium of corpus callosum makes it highly vulnerable.
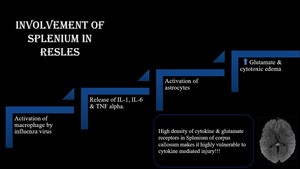
Fig. 27: Involvement of splenium in RESLES.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN
SUMMARY:

Fig. 28: Summary of predilection sites.
References: Dept. of radiology, Government kilpauk medical college, Chennai/IN