Imaging
CT of the brain often fails to show the early changes of central pontine myelinolysis because it results from subtle alteration in the tissue water content, which is further obscured by artifact occurring in this region of the brain; after the onset of symptoms there is a delay of one-two weeks for the detection of the lesions on CT images, with an appearance of hypoattenuating lesions.
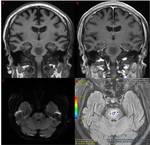
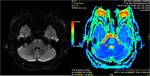
The lesions, under MR imaging mode, appear as areas of hyperintensity on T2-weighted images and FLAIR images, corresponding to areas of hypointensity on T1-weighted images. The most sensitive and early sequence is DWI, that shows restricted diffusion with associated reduced Apparent Diffusion Coefficient (ADC) Values; this finding reflects the intramyelinic edema that determines impairment of free water molecules movement in extracellular space. DWI can also show restricted diffusion in the early 24 hours after the onset of ODS, which is an important finding for early diagnosis. The administration of contrasting agents is not conclusive: peripheral contrast enhancement may occur but is not a prominent imaging feature.
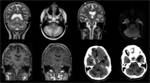
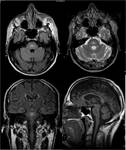
The extent and distribution of the abnormality may vary. The most characteristic appearance consists of well demarcated rounded, or trident-shaped lesions in the center of the pons, with a characteristic sparing of the peripheral pons and corticospinal tract regions. The lesions can extend from the pontomedullary junction into the midbrain, with the characteristic sparing of the tegmentum. In more severe cases, almost the entire central pons may be involved with only a thin rim of normal signal around it.
The most commonly involved extrapontine sites are the basal ganglia, thalami, and hemispheric white matter, where the characteristic symmetry of the lesions is often respected. In the cases of laminar necrosis of the cortex, the damaged areas are depicted on images as multiple and diffuse punctuate lesions on juxtacortical white matter or delineating the transition line between cortical gray matter and subjacent white matter.
The variety of appearance and the unspecific signal characteristics of the lesions justify the necessity to consider also their distribution and the clinical features, that contribute to provide the basis for the diagnosis.
Differential diagnoses
Several conditions can mimic ODS, including other demyelinating diseases, therefore they must be included in the spectrum of possible diseases. In these cases, there can be involvement of the pons but it is rarely symmetric, with FLAIR scans demonstrating lesions elsewhere often extended in the supratentorial regions, and therefore clinical differentiation is not problematic.
Post-irradiation and post-chemotherapy changes can easily be diagnosed from the clinical history in the majority of the cases. Neoplasm rarely mimics ODS; among these, pontine gliomas, that are tumors of childhood and young adulthood, appear hyperintense on T2/FLAIR scans and can expand the pons, whereas demyelinating lesions in ODS do not show mass effect.
Metastatic disease, that is more typical of adulthood, when localized in the posterior fossa, affects typically the cerebellum, not the pons. Furthermore, brainstem neoplasms usually present in a more subacute manner.
Hypertensive encephalopathy (PRES) can involve the pons but does not spare the peripheral WM tracts. The major differential diagnosis of extrapontine ODS with basal ganglia and/or cortical involvement is metabolic disease. The basal ganglia are affected in Wilson disease and mitochondrial disorders, but the pons is less commonly involved.
Treatment of pontine myelinolysis
Although OM was once believed to be fatal with a survival rate of only 5% to 10% beyond 6 months, many patients survive much longer, but the clinical outcome varies widely, and recovery can be complete or partial, with the most serious manifestation being the “locked-in syndrome”.
The management of the patient is essentially supportive, since myelinolysis itself cannot be specifically treated once it develops. Supportive therapies are indicated for alleviate symptoms such as depression, psychosis and abnormal somatic movements. Recommendations for management of hyponatremia stress the importance of slow correction; gradually increasing the sodium concentration, from 4-6 mmol∙L-1 in any 24 hour period, is the most favorable therapy to treat a patient with severe hyponatremia while preventing any unwanted side effects. Therapeutic plasmapheresis has been reported as a safe and effective method of improving the clinical outcome; it is believed that it may reduce myelotoxic substances, thereby leading to clinical improvement.