Imaging plays a crucial role when patients with critical conditions arrive at the emergency department with an acute confusional state that can subtend a cerebral dysfunction. When clinical data suggest the presence of a toxic and\or metabolic disorder, many exogenous or endogenous causes and substances must be considered.
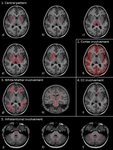
A systematic approach based on the topographical distribution of imaging findings (patterns) and also on a specific signal appearance, is preferable to face this large group of diseases and it could suggest faster the most likely diagnosis in a restricted group of possibilities(Fig.1).
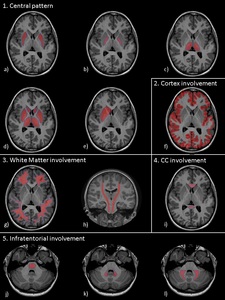
Fig. 1: a) Putamina; b) Globi-Pallidi; c )Thalami; d) Diffuse Basal-Ganglia involvement; e) Unilateral exceptions; f) Cortex involvement; g) Periventricular White-Matter; h) Cortical-spinal tracts; i) Corpus-Callosum; j) Brain stem; k) Dentate-Nuclei; l) Butterfly-wing pattern
1) CENTRAL PATTERN (BG and thalami):
The “central pattern” is the largest and most heterogeneous group. Despite the topographical distribution of the lesions is an important start-point, the analysis of different MRI signal characteristics is also an ultimate approach to deal with the various diagnostic possibilities and contains the comprehensive evaluation of T1-WI, T2-WI, SWI and DWI.
Paramagnetic substances (e.g. iron) catalyze the nuclear-spin-relaxation of neighboring water-protons and appear hypointense on T2-WI. Calcium deposits may mimic iron deposits or hemorrhages, but the SWI filtered-phase-images could be helpful to differentiate them: the dark regions in the magnitude-image appear bright and have an opposite signal to the veins.
Manganese and copper are metals that may be deposited in neurodegenerative disorders and they appear hyperintense on T1-WI. Tab.3 summarizes the above-mentioned MRI characteristics.
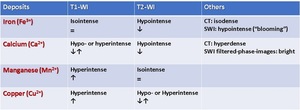
Table 3
1a. Central pattern: topographical approach
(prevalent affected areas)
Putamina:
- Methanol poisoning(+ WM)(Fig.2)
- Uremic encephalopathy: three patterns: BG involvement, acute toxic leukoencephalopathy(ATL), PRES-like
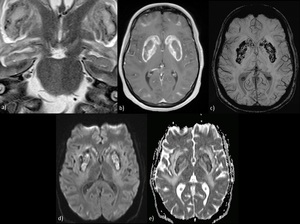
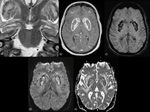
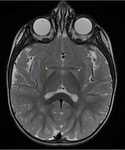
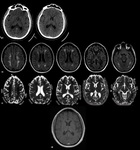
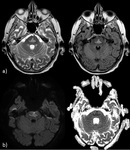
Fig. 2: Methanol poisoning caused by accidental ingestion of methanol-containing agents or from the intake of adulterated alcoholic beverages (moonshine). Bilateral symmetrical signal alteration of the BG: mainly the putamen. a) Bright T2 signal; b) CE in this case; c) SWI: necrotic hemorrhage; d) Restricted diffusion.
References: Case courtesy of Dr Annibale Botto, UOC di Neuroradiologia - AOU S. Giovanni di Dio e Ruggi d’Aragona - Salerno/IT
Globi-Pallidi:
- Carbon-monoxide(CO) poisoning(Fig.3)(+ WM in the subacute-phase)
- Chronic hepatic encephalopathy(+ substantiae nigrae, perirolandic cortex (laminar T2-hyperintensities), cortical-spinal tracts)
- Vigabatrin-associated toxicity(+ thalami)
- Manganese accumulation(Fig.4)
- Toluene use(+ thalami, substantiae nigrae)
- Kernicterus(Fig.5)
- NBIA(Fig.6)
- Gadolinium-deposits(lack of clinical evidence\neurologic effects)
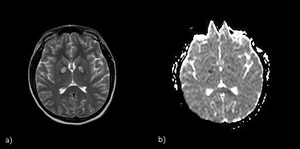
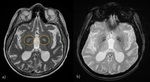
Fig. 3: CO poisoning. a) Simmetric hiperintensities in globi pallidi on T2-WI; b) ADC-map: restricted diffusion.
References: Case courtesy of Dr A Mehmet, Radiopaedia.org. From the case rID: 15968
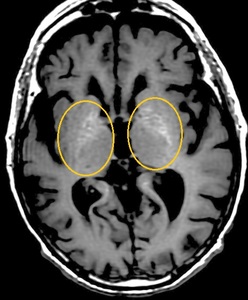
Fig. 4: Manganese accumulation. It occurs in chronic hepatic encephalopathy or caused by occupational exposure or hyperalimentation. T1-WI hyperintensities of Globi Pallidi (short T1).
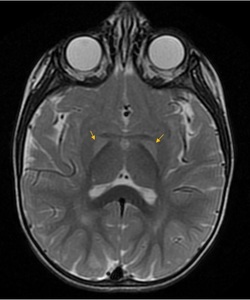
Fig. 5: Kernicterus (Bilirubin Encephalopathy in the newborn). T2-WI hyperintensities in both Globi Pallidi (subacute and chronic phases). They appears hyperintense in T1-WI in the acute phase.
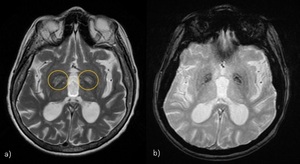
Fig. 6: NBIA, Neuroferritinopathy (NFT): autosomal dominant form of adult onset NBIA (symptoms: dystonia–chorea and dementia). a)T2-WI: "eye of the tiger" sign: the “eye” corresponds to a region of rarefaction surrounded by more preserved iron-laden neuropil, neurons, and astrocytes; b) SWI: paramagnetic effect of iron deposits.
Lenticular-Nuclei “Lentiform-fork-sign”(Fig.7):
- Metabolic acidosis: uremic encephalopathy, diabetes mellitus, methanol and ethylene glycol intoxications, acidopathies
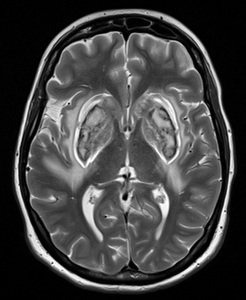
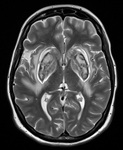
Fig. 7: Lentiform fork sign (on T2-WI). Kumar et al. described the elements of the lentiform fork: 1) Lateral arm: edematous external capsule (from the anterior end of the putamen to the stem); 2) Stem: blending of edematous external and internal capsules at the infero-posterior end of the putamen; 3) Medial arm: from the stem to one third of the medial edge where it split into two hyperintense branches engulfing the globus pallidus.
Thalami:
- Wernicke-encephalopathy(+ mammillary bodies, hypothalamus, tectal plate and periaqueductal GM, perirolandic cortex)(Fig.8)
- Vigabatrin-associated toxicity (+ GP and dentate nuclei)
- Toluene use(+ GB and substantiae nigrae)
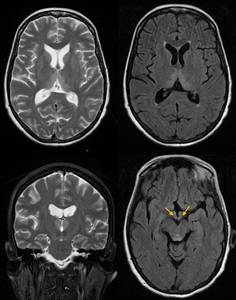
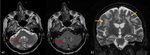
Fig. 8: Wernicke-encephalopathy (WE). a)T2-WI show bilateral involvement of the thalami, mammillary bodies (arrows), hypothalamus and periaqueductal GM. Also tectal plate and perirolandic cortex can be affected; b) restricted diffusion; c) CE of mammillary bodies in 80% of cases (pathognomonic). WE is related to chronic alcohol abuse, but also to all the conditions that lead to malnutrition and thiamine deficiency (e.g. hyperemesis, bariatric surgery), an important vitamin (B1) involved in cell membrane integrity.
Diffuse BG involvement:
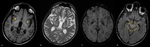
- Wilson disease(+ thalami and “Face of the Giant Panda” sign)(Fig.9)
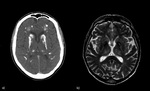
- Parathyroid disorders/Fahr's Syndrome(Fig.10)
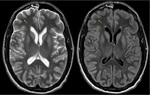
- Extrapontine myelinolysis(Fig.11)
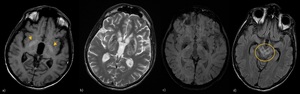
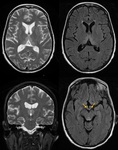
Fig. 9: Wilson disease (WD) in two different patients. a) Patient 1, Wilson disease with hepatic dysfunction without neurologic symptoms: T1-WI show hyperintesities of Globi Pallidi; b,c,d) Patient 2, Wilson disease with simultaneous neurologic symptoms and hepatic dysfunction: (b) symmetrical increased T2 signal in the basal ganglia (ventro-lateral thalami involvement can be present);(c) hypointense deposits on SWI in WD patients are mainly caused by iron deposition (Yang J. et al); (d) “Face of the Giant Panda” sign: T2-hyperintensity of the midbrain excluding the red nuclei (eyes) and lateral pars reticulata of the substantia nigra (ears) which are hypointense.
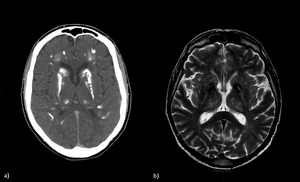
Fig. 10: a) Patients with Fahr's Syndrome (rare, genetically dominant condition): TC shows the idiopathic iperdense calcifications in basal ganglia; b) Patient with hypoparathyroidism: T2-WI shows bilateral hypointensities (calcium deposits) involving the basal ganglia.
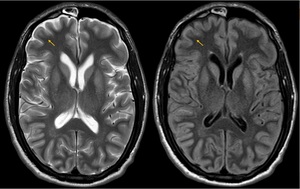
Fig. 11: Osmotic Demyelination Syndrome (ODS) - Extrapontine myelinolysis. Axial T2\FLAIR images shows diffuse Basal ganglia involvement (symmetric and bilateral hyperintensities) and juxtacortical hyperintense lesions (arrows). Cortex can be also involved in extrapontine myelinolysis.
Unilateral exceptions:
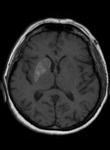
- Non-ketotic-hyperglycemic-hemichorea-hemiballismus (striatum)(Fig.12)
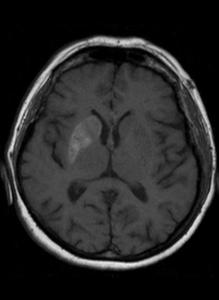
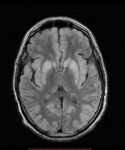
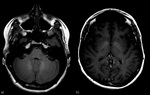
Fig. 12: Non-ketotic hyperglycemic hemichorea (NHH) or diabetic striatopathy. T1-WI shows hyperintensity of the right lentiform and caudate nuclei. No mass effects. Chorea and ballismus develops rapidly during episodes of non-ketotic hyperglycemia (> in type 2 diabetes mellitus). Symptoms resolve after normalization of glucose levels.
References: Case courtesy of Dr Ayaz Hidayatov, Radiopaedia.org. From the case rID: 56241
1b. Central pattern: signal characteristic approach
T2-WI\FLAIR hyperintensity:
- Wernicke-encephalopathy
- Methanol poisoning: restricted diffusion on DWI, hemorrhages on SWI
- CO poisoning: T2\FLAIR hyperintensity of GP with hypointense rim (hemorrhage), restricted diffusion
- Uremic encephalopathy
- Vigabatrin-associated toxicity: restricted diffusion
- Wilson disease (simultaneous neurologic symptoms and hepatic dysfunction)
- Kernicterus (subacute and chronic phases)
- Pantothenate-kinase-associated-neurodegeneration (PKAN) and Neuroferritinopathy (NFT): “Eye of the tiger” sign (Fig.6)
T2-WI hypointensity:
- Toluene use (iron deposition?)
- Parathyroid disorders (calcium deposits)
- NBIA (iron deposits)
T1-WI hyperintensity:
- Non-ketotic-hyperglycemic-hemichorea-hemiballismus
- Chronic hepatic encephalopathy
- Manganese accumulation
- Wilson disease (hepatic dysfunction without neurologic symptoms)
- Kernicterus (acute phase)
- Gadolinium-deposits(Fig.23)
Tab.4 is a useful attempt to merge these two different approaches.
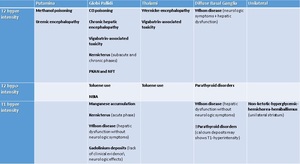
Table 4
2) PREVALENT CORTEX INVOLVEMENT:
- Hyperammonemia(Fig.13)
- Hypoglycemic brain injury (in adults)(Fig.14)
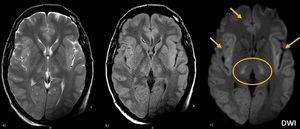
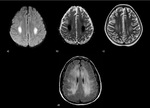
Fig. 13: Hyperammonemia. a, b) Symmetric T2\FLAIR hyperintensities and restricted diffusion (c) in the insular and cingulate gyri. Also basal ganglia and thalami can be involved.
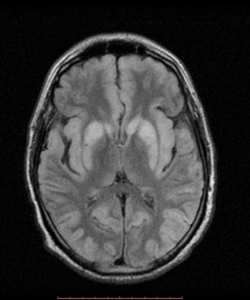
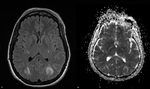
Fig. 14: Hypoglycemic brain injury (in adults) FLAIR axial image shows symmetric hyperintensities in the parieto-occipital and temporal regions. Basal ganglia are also involved, thalami are spared. Lesions also present Restricted diffusion.
References: Case courtesy of Dr Nikos Karapasias, Radiopaedia.org. From the case rID: 25687
3) WHITE-MATTER INVOLVEMENT:
Symmetric Periventricular WM:
- Methotrexate-leukoencephalopathy(Fig.15)
- Inhaled Heroin (chasing the dragon)(Fig.16)
- Cocaine-leukoencephalopathy
- Delayed post-hypoxic leukoencephalopathy(Fig.17)
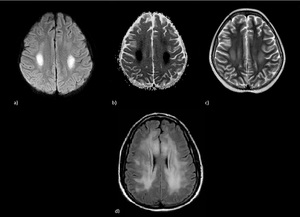
Fig. 15: Methotrexate leukoencephalopathy: a, b, c) Patient 1: Bilaterally hyperintensities on T2-WI in the centrum semiovale with diffusion restriction; d) Patient 2: bilaterally asymmetric (across multiple vascular territories) high T2 signal alterations of the deep white matter, subcortical U-fibers are spared.
References: Case courtesy of A.Prof Frank Gaillard, Radiopaedia.org. From the case rID: 27698 and Case courtesy of A.Prof Frank Gaillard, Radiopaedia.org. From the case rID: 4438
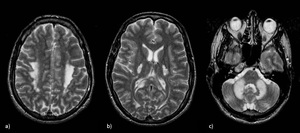
Fig. 16: Inhaled Heroin (chasing the dragon). Axial T2-WI show confluent, bilateral and symmetric hyperintensities involving posterior limb of internal capsule, perirolandic subcortical white matter, occipital lobes and also the cerebellar white matter sparing the dentate nuclei (butterfly-wing pattern). The anterior limb of the internal capsule and subcortical U-fibers are usually spared. Restricted diffusion is usually present in the acute phase.
References: Case courtesy of A.Prof Frank Gaillard, Radiopaedia.org. From the case rID: 4442
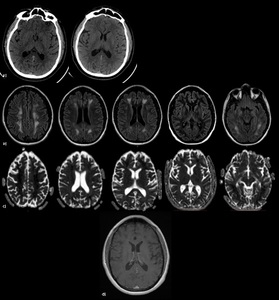
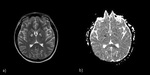
Fig. 17: Delayed Post-hypoxic Leukoencephalopathy (DPHL) after Benzodiazepine overdose - The pathomechanism of DPHL is not fully understood (myelin-sheath damage?). DPHL is mainly described in the context of CO intoxication and other causes of hypoxia (drug overuse, cardiac arrest, strangling, seizures). a) Unenhanced CT images of a patient found comatose at home show symmetrical and bilateral hypointensities of the globus pallidus, no involvement of withe matter. After few days the patient clinically improved and was extubated; b, c, d)MRI after 4 weeks show symmetric T2-hyperintensities (b) of the white matter with a marked diffusion restriction (c) and no contrast-enhacement (d). The cortex, U-fibers and infratentorial structures are spared.
Cortical-spinal tracts:
- Cobalamin(B12) deficiency(Fig.18)
- Chronic hepatic encephalopathy
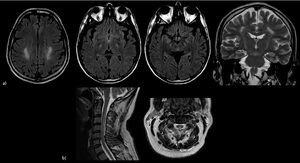
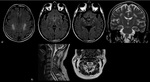
Fig. 18: Cobalamin (B12) deficiency. a) Intracranial findings: axial and coronal T2\FLAIR images show bilateral hyperintensities of the corticospinal tracts; b) Medullary findings: dorsal and lateral spinal cord columns are affected (inverted V-shape sign in axia image). Usually there is a complete resolution after treatment.
4) CORPUS CALLOSUM INVOLVEMENT
- Marchiafava-Bignami-disease(Fig.19)
- Cytotoxic Lesions Of the Corpus-Callosum (CLOCCs)(Fig.20)
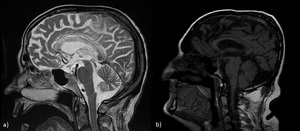
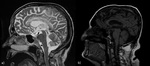
Fig. 19: Marchiafava-Bignami disease (MBD). Related to chronic ethanol abuse: osmotic demyelination and necrosis of the corpus callosum. a) Patient 1: sagittal T2-WI show hyperintensities in the corpus callosum ; b) Patient 2: sagittal T1-WI show selective involvement (hypointensities) of the middle layers of the corpus callosum (sandwich sign).
In the chronic MBD, the CC appears thinner.
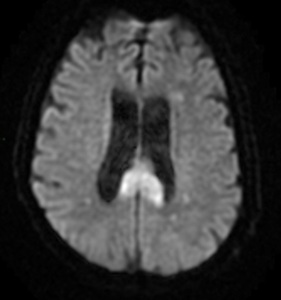
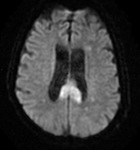
Fig. 20: Cytotoxic Lesions Of the Corpus-Callosum (CLOCCs). Oval lesion with restricted diffusion in the splenium. CLOOCs are associated with drug therapy, malignancy, infections, subarachnoid hemorrhage, metabolic abnormalities, trauma and represent an important imaging differential diagnosis for MBD. Typically shows one of three patterns: 1) a small round or oval lesion in the center of the splenium, 2) a lesion centered in the splenium but extending through the callosal fibers, 3) a lesion centered in the posterior portion of CC but extending anteriorly. They are usually reversible probably due to excitotoxic intracellular and/or intramyelinic edema.
5) INFRATENTORIAL INVOLVEMENT:
Brain stem:
- Pontine-myelinolysis(Fig.21)
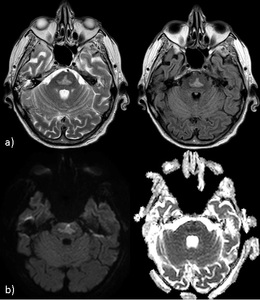
Fig. 21: Pontine myelinolysis. a) T2\FLAIR axial images and b) DWI and ADC-map show well-demarcated trident-shaped lesion on the central pons (sparing the periphery); restricted diffusion appears 24 hours after the onset of Osmotic Demyelination Syndrome (oligodendrocytes of the pons are more vulnerable to osmotic changes).
Cerebellum:
- Metronidazole-associated toxicity(Fig.22)
- Gadolinium-deposits(Fig.23)
- Inhaled Heroin (butterfly-wing pattern)(Fig.16)
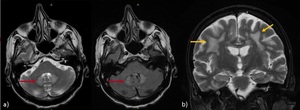
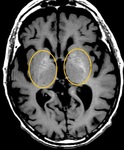
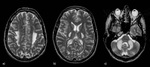
Fig. 22: Metronidazole associated toxicity. a) Axial T2\FLAIR images (red arrows) show symmetric hyperintensities of dentate nuclei (without CE); b) Coronal T2-WI (yellow arrows) shows involvement of periventricular white matter. The splenium can be also involved.
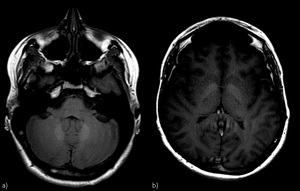
Fig. 23: Gadolinium deposits. a) T1-WI hyperintensities of dentate nuclei; b) also globi pallidi are involved.
Lack of clinical evidence\neurologic effects of gadolinium deposition.
RELATED REACTIVE CONDITIONS:
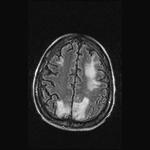
- PRES (typical\atypical): commonly described as a cerebrovascular-self-regulatory disorder caused by hypertension, but 20–30% of patients are normotensive. This neurotoxic state is also explained by endothelial dysfunction secondary to cytotoxic\immunogenic causes (Tab.5), resulting in vasogenic edema (interstitial-fluid accumulation in the subcortical WM). It’s not always reversible or posterior (typical). Tab.6 sums up typical (Fig.24) and atypical (Fig.25) PRES features.
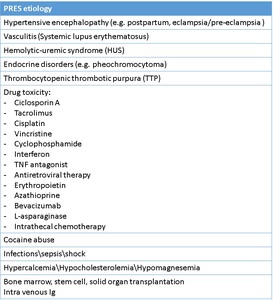
Table 5
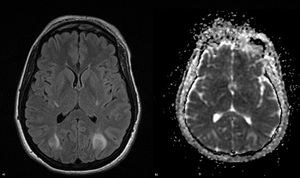
Fig. 24: Typical PRES. a) FLAIR; b) ADC-map. Imaging findings: bilateral, asymmetric hyperintensities on T2-WI\FLAIR involving the subcortical White Matter. ADC-maps are often negative (vasogenic edema). Patchy or variable CE. SWI may show microhemorrhages.
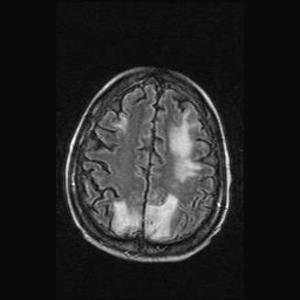
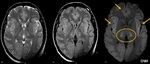
Fig. 25: Atypical PRES. FLAIR: Superior frontal involvement
References: Case courtesy of Dr Alessandro Spano Mello, Radiopaedia.org. From the case rID: 32300
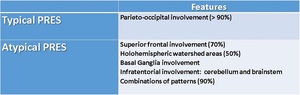
Table 6
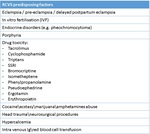
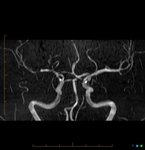
- RCVS: Thunderclap headache of a few hours is the usual primary symptom and it periodically recurs over days or weeks. It’s characterized by reversible vasoconstriction of the cerebral arteries(Fig.26). RCVS should not be confused with PRES, although they have similar presumed underlying mechanisms and predisposing factors (Tab.7), and with vasospasm related to subarachnoid-hemorrhage, in fact in RCVS subarachnoid-hemorrhage is believed to be a secondary phenomenon and not the inciting event.
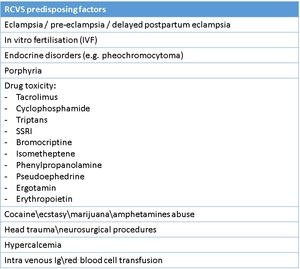
Table 7
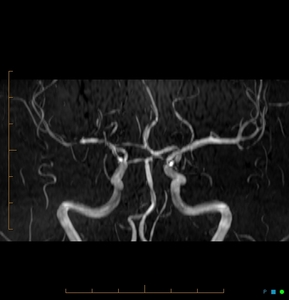
Fig. 26: RCVS. Coronal MRA shows mutiple sites of arterial narrowing (> right side, MCA)
References: Case courtesy of Dr Chris O'Donnell, Radiopaedia.org. From the case rID: 43356