To make a definite diagnosis the radiologist must recognize the main findings related with the most common emergencies:
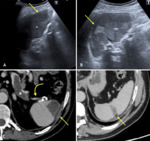
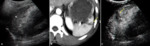
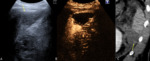
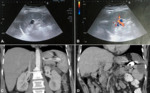
The US appearance depends on the time of the event (Fig.1). In the acute phase, the infarcted area becomes oedematous due to associated inflammation and necrosis (Fig.1), so the classic appearance is a wedge-shaped hypoechoic lesion, solitary and extending to the periphery of the spleen, with absent flow on Doppler interrogation (Fig.2). The lesion may decrease in time, becoming gradually fibrous and so hyperechoic (Fig.1), without Doppler signal, when compared with normal adjacent parenchyma. In the chronic phase, calcifications may occur and detected as highly reflective echoes. Occasionally infarcts may become infected or may bleed. CEUS is more sensitive in the identification of infarcted areas, that will appear without any enhancement in both the arterial and venous phases, and may help in differential diagnosis.
Typically will appear as a round or oval ipoechoic areas accompanied by fever, left upper quadrant tenderness and, in one-third of cases, by leucocytosis. There may also be splenomegaly and edema of the overlying soft tissues (Fig.3).
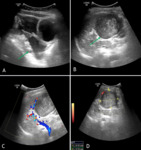
Spleen is highly vascular, so, in case of damage, there is an important risk of haemorrhagic shock and death. If not identified, delayed rupture may occur (Fig.4), determining important bleeding even a long time after the traumatic event.
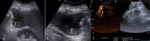
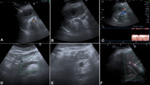
The clinical manifestation is represented by upper left quadrant pain radiating to the ipsilateral shoulder, associated with lower quadrant pain typical of hemoperitoneum. Abdominal rigidity may be present, but also no one of these signs (Fig.5)!
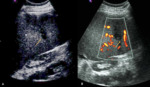
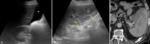
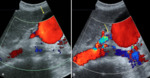
In some cases, free abdominal fluid with internal echoes (Fig.4) may be detected, associated with inhomogeneous texture of the spleen due to laceration (Fig.4), contusion or subcapsular haematoma. The laceration appear as a subtle, hyperechoic line within the spleen immediately after the injury. A frank area of haemorrhage, easily identifiable on ultrasound, may not develop until later. CD may be helpful in delimiting the area without vascularization at the laceration site. CEUS is particularly helpful in doubtful cases in patients with minor trauma, increasing the sensitivity for the detection of traumatic injuries and showing non-vascularized hypoechoic areas compared to surrounding parenchyma, that appears hyper-reflective for the presence of contrast medium (Figs.5,6).
- Spontaneous splenic rupture
This is a very rare, potentially lethal condition. There are several underlying pathologies that may lead to spontaneous rupture (haematological, neoplastic, inflammatory and infectious) without a history of trauma, due to the massive spleen or mass enlargement. Findings are represented by free abdominal fluid, splenomegaly and inhomogeneous texture of the spleen related to the lesion (Fig.7).
- Splenic artery aneurysm and pseudoaneurysm
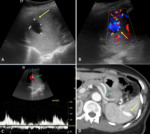
Arterial aneurisms and pseudoaneurisms are seen as an outpouching of the arterial vessel wall, with or without peripheral calcifications (Fig.8). CD confirm the arterial flow (Fig.9) within the aneurysm whereas PW help in differentiating it from other possible cystic masses near the splenic hilum, such as pancreatic pseudocysts. Aneurysm and pseudoaneurysm are clinically significant if over 2 cm in diameter, when the risk of rupture and fatal haemorrhage is present. Pseudoaneurysm may be distinguished from true aneurysm if an underlying cause (surrounding inflammation, haemorrhage) is detected.
Splenic intraparenchymal pseudoaneurysm may occurs following splenic trauma. A ‘cystic’ area may be observed at B-mode examination (Fig.10), demonstrating vascular flow in the central anechoic area at CD, with the characteristic “yin-yang” flow, and bidirectional waveform pattern at PW. In rare cases, pseudoaneurysm may manifest as complication of splenic infarct, infiltration of the spleen by malignancies or inflammatory disease such as pancreatitis or infections.
- Splenic arteriovenous fistula
It may occur following trauma or percutaneous procedure and is frequently associated with pseudo aneurysm in which a turbulent and pulsatile blood flow with increased velocity is detected, together with a dilated and elongated afferent splenic artery and draining splenic vein (Fig.10). The hyperdynamic blood flow state through the arteriovenous shunt leads to the sudden increase in pressure and congestion within the portal and mesenteric venous system, determining a characteristic “machinery bruit” auscultated in the epigastric, left upper quadrant or left flank area.
- Splenic vein thrombosis (SVT)
Because of the close anatomical relationship between the splenic vein and pancreas, it is often related to pancreatic pathologies, like acute and chronic pancreatitis and pancreatic malignancies. Splenomegaly is due to an increased pressure within the splenic vein from the thrombosis. Increased splenic venous congestion and splenic arterial blood flow can cause haemorrhage, infarction and rupture. SVT will appear echogenic without detectable flow on Doppler-US in cases of complete obstruction (Fig.11). Recent partial or total thrombosis in the first days appears hypoechoic and is quite difficult to diagnose only at B-mode US, so Doppler signs are crucial for the detection. CEUS examination may constitute a further step, showing a higher accuracy when doubts persists on the vessels patency.
- Splenic vein aneurysm (SVA)
US is the initial imaging choice for diagnosing SVA, demonstrating either a saccular or fusiform, and hypo- or anechoic splenic vein dilatation. For extrahepatic portal vein aneurysms, which include SVAs (near the confluence with the superior mesenteric vein), a diameter greater than or equal to 2 cm is considered the diagnostic standard for cirrhotic patients, whereas it is at least 1.5 cm for the non-cirrhotic patients. The aneurysm will normally fill up with color flow at an increased flow velocity, with the “Yin-Yang sign” flow during systole and diastole. CEUS may help in better delineation of the vessel lumen and patency.
- Spontaneous spleno-renal shunt (SSRS)
SSRS will appear as tortuous inferiorly directed blood vessels originating from the splenic hilum to the left kidney with evidence of splenofugal blood flow (Fig.11) associated with increase of splenic volume, dilated left renal vein with phasic blood flow at a velocity of more than 20cm/sec, and dilated splenic vein(greater than 5 mm) with phasic blood flow at a velocity of more than 15cm/sec.