In this section we will focus on different components of Structured reporting format for CT and MR liver dynamics and we will discuss the differences in the end.
1. History:
It is the most important component. For liver dynamics we need detailed history especially of the following aspects;
1. Viral serology status (HBV and HCV)
2. Prior imaging information whether CT, MR or USG.
3. Current complaint of patient and clinical question that need to answered.
4. Any history of CLD, HCC and if present any MDT decision regarding its management.
5. Any treatments done for prior HCC, if perfomed mention with dates.
Here is an example ( Fig. 6) of good and short history for the CT liver dynamic.

Fig. 6: History of CT liver dynamic.
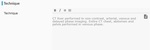
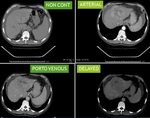
The technique of the scan should be mentioned here. Any artifects or technical problems limiting the interpretation of scan should be mentioned here. At our institute we perform quad phase CT scan of liver
Fig. 7 with venous phase covering entire chest, abdomen and pelvis.
Fig. 7: Technique of CT liver dynamic.
Comparison to prior studies is important for evaluation of response of disease. So comparison with dates and modality of scan must be mentioned as in
Fig. 8 .
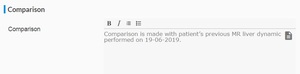
Fig. 8: Comparison heading.
Now we will discuss subheadings of Findings which include;
4. Cirrhosis:
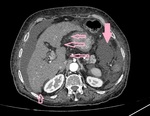
It is important for clinicians to know whether liver has radiological features of cirrhosis or not. The CT is not sensitive modality for early cirrhosis. However, advance cirrhosis can be picked up easily on CT as shown in
Fig. 9 . (2)
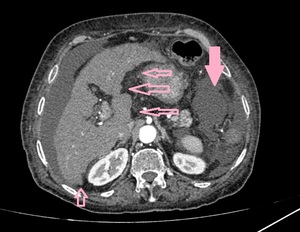
Fig. 9: CT scan arterial phase axial image shows nodular liver margins consistent with cirrhotic liver morphology. The surrounding fluid is marked with solid rose coloured arrow while the margins of liver are marked with other rose coloured arrows.
In this section we discuss radiological features of portal hypertension, which include portal vein dilatation, splenomegaly, ascites and varices.
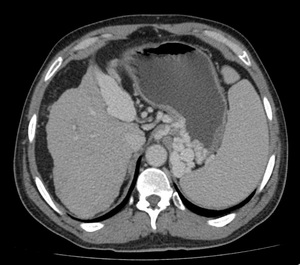
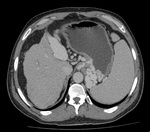
Fig. 10: CT scan axial image in venous phase shows recanalization of umbilical vein with enlarged spleen and multiple upper abdominal varices. Features of portal hypertension.
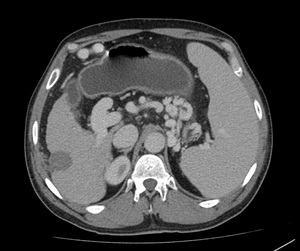
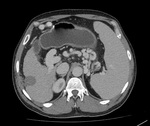
Fig. 11: CT scan axial image in venous phase shows dilated portal vein with enlarged spleen and multiple upper abdominal varices. Features of portal hypertension.
6. Liver Lesions:
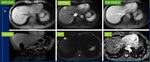
In this segment we describe all the lesions with maximum dimension size and their segment. Then characterize the lesion as HCC, other diagnosis (hemangioma etc) or indeterminate lesions. In case of MR, we also classify dysplastic and regenerative nodules.
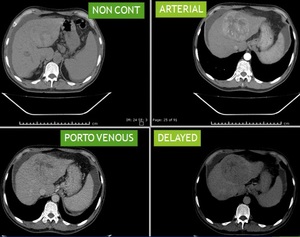
Fig. 12: CT scan axial images shows a large lesion in segment IVa of liver with typical features of HCC i.e, arterial phase enhancement with venous or delayed phase washout. Cirrhotic liver morphology and mild ascites is also noted.
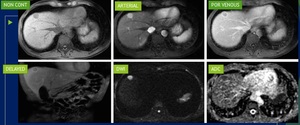
Fig. 13: MR liver dynamic shows a small lesion in segment IVa of liver which is showing arterial phase enhancement with washout on venous and delayed phase images. The lesion is also showing restricted diffusion. All the features favour HCC.
In this section, we review for any variant hepatic arterial anatomy. It will help the interventional radiology doing an embolization procedure.
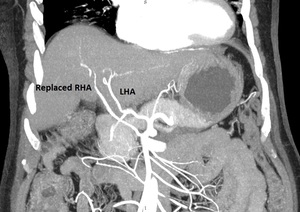
Fig. 14: Coronal MIP arterial phase image of upper abdomen showing replaced right hepatic artery (RHA) supplied by SMA and conventional left hepatic artery (LHA) from celiac axis. The patient has an HCC, which is not shown in the image. This is important information for interventional radiologist going to perform embolization of this lesion.
We discuss the biliary system including gall bladder in this section for any associated abnormalities.
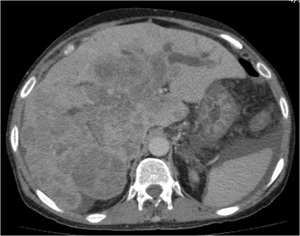
Fig. 15: CT scan axial image delayed phase image showing infilrative type of HCC with infiltration into biliary ducts showing mild dilatation, prominent in left lobe.
Here we discuss, whether a tumor thrombus is present or not and what is the extent of tumor thrombus.
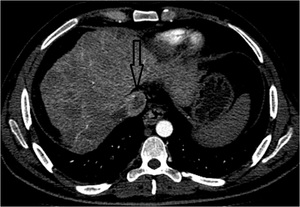
Fig. 16: CT scan axial image showing an ill defined infiltrative type of HCC with tumor thrombus in IVC (arrow).
In this section we discuss any extra hepatic spread of HCC, especially lymph nodal involvement. Any metastatic lesion is discussed here.
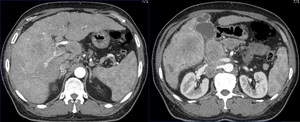
Fig. 17: Known case of HCC. Multiple liver lesions (HCCs) with enlarged upper abdominal lymph nodes in porta hepatis and retroperitoneal locations.
11. Overall tumour burden:
Overall tumor burden is calculated in our department as per Modified Response Evaluation Criteria in Solid Tumors (mRECIST) for response evaluation. We measure only the arterialized component of HCC along the maximum dimension. We add all the arterialized components of HCC to calculate total tumor burden.
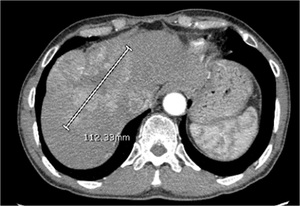
Fig. 23: CT scan arterial phase showing large HCC which has been measured through its largest dimension of enhancing component as per mRECIST.
Rest of abdomen and pelvis assessment comes under this heading.
13. Chest bases or Chest:
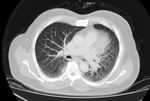
We look for any chest nodule or mass lesion in this section. MPR MIP images are used for evaluation of any lung nodule.
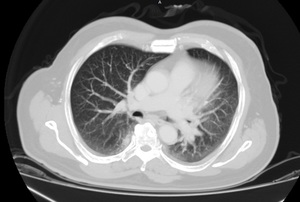
Fig. 18: CT chest MRP MIP axial images for evaluation of lung nodules.
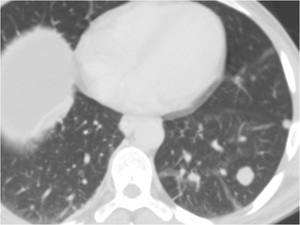
Fig. 19: Known case of HCC. An enhancing nodule in left lower lobe, suspicious of lung metastasis.
In this section, we review bones in MPR settings to look for any bone metastatic lesion.
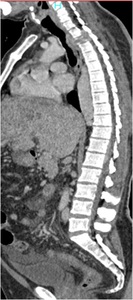
Fig. 20: A patient with known HCC. CT scan venous phase sagittal image showing a lytic destructive lesion in T2 verterbral body projecting into spinal canal, suggestive of metastatic lesion.
We conclude by mentioning tumor burden of HCC. Any extra hepatic disease, lung or bone mets and vascular invasion.
We do response evaluation according to mRECIST for followup scans. [3]
We have not started applying LIRADS at our institute, but it is the next step forward.
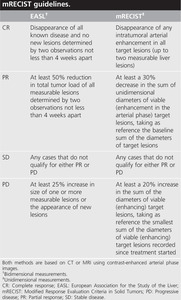
Fig. 21: EASL and mRECIST response criteria for HCC.
References: Kim M, Kim B, Han K, Kim S. Evolution from WHO to EASL and mRECIST for hepatocellular carcinoma: considerations for tumor response assessment. Expert Review of Gastroenterology & Hepatology. 2014;9(3):335-348.
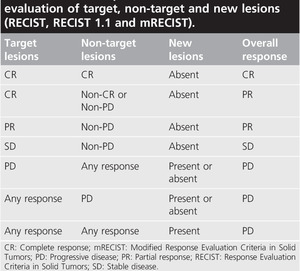
Fig. 22: Evaluation of target, non-target and new lesions as per different criteria.
References: Kim M, Kim B, Han K, Kim S. Evolution from WHO to EASL and mRECIST for hepatocellular carcinoma: considerations for tumor response assessment. Expert Review of Gastroenterology & Hepatology. 2014;9(3):335-348.
We advice regarding followup or further workup of any indeterminate lesion. Also mention to discuss it in MDT meeting.