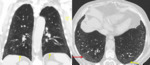
UIP Pattern
Histologically, it is characterized by the presence of a heterogeneous pattern with different parts of the biopsy showing normal lung, interstitial fibrosis, fibroblast proliferation, and honeycombing. They each represent a different stage in the temporal evolution of fibrosis, reflecting spatial and temporal heterogeneity, typical of UIP.
Definite UIP Pattern
- Subpleural, basal predominance
- Reticular changes
- Honeycombing with/without traction bronchiectasis
- Absence of characteristics considered inconsistent with the UIP pattern
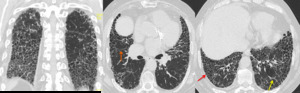
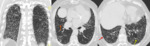
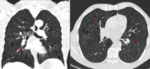
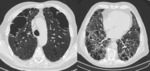
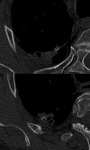
Fig. 1: HRCT of a patient with a definite UIP pattern displaying all features for the diagnosis: subpleural, basal predominance, reticulation (yellow arrow), bronchiectasis (orange arrow)
and extensive honeycombing (red arrow).
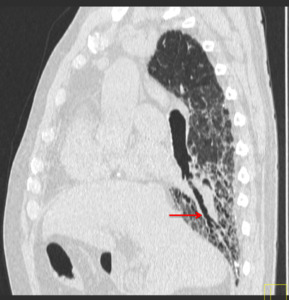
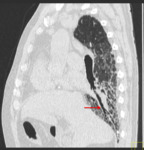
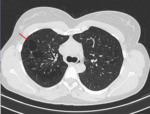
Fig. 2: HRCT - sagittal reformat. Reticulation, honeycombing pattern and traction bronchiectasis (red arrow) are all elegantly displayed.
Findings considered inconsistent with UIP pattern
• Upper/middle lobe predominance
• Peribroncovascular predominance
• More extensive ground-glass component than reticulation
• Profuse micronodules
• Bilateral mosaic attenuation
• Bronchopulmonary consolidation
NSIP
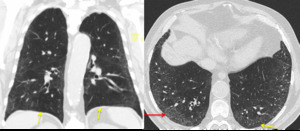
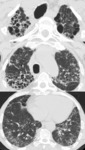
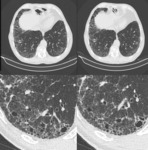
Fig. 3: HRCT of a 64 year-old man displaying a NSIP pattern. There is a small reticulation component and some septal thickening. There is a relatively symmetrical and bilateral distribution of the findings across the lung. Lung density is significantly increased, with the presence of ground glass (red arrows). Note the relative sparing of the subpleural lung parenchyma (yellow arrows) and the absence of cysts.
Honeycombing
Honeycombing represents destroyed and fibrotic lung tissue.
Criteria for identification on HRCT
- Air cysts
- Easily recognizable wall
- Diameter: usually between 3 and 10mm
- Localization: subpleural
- Cysts share walls
- Presence of two(?)/three(?) cysts in a line: minimum for diagnosis
Interobserver variability in the diagnosis of honeycombing is still problematic.
The radiologist should be alert to cystic lung diseases that can mimic honeycombing, resulting in potential pitfalls:
Lymphangioleiomiomatosis
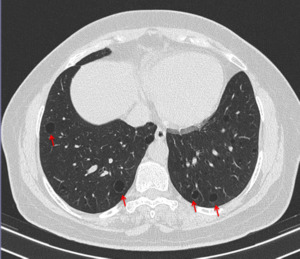
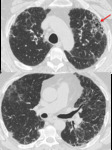
Fig. 4: HRCT of a 35 year old woman with LAM. Several cysts distributed randomly across the lung parenchyma (red arrows) are observed. No signs of fibrosis, volume loss or other lung changes are visible.
Lymphoid Interstitial Pneumonia
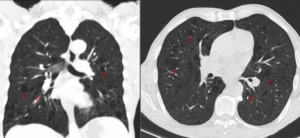
Fig. 5: HRCT of patient with lymphoid interstitial pneumonia. Well defined cystic formations, scattered in the parenchyma, not matching the honeycombing pattern.
As the name suggests, IPF does not have a known cause. Therefore, other fibrosing lung diseases should be ruled out before considering this diagnosis:
Chronic Hypersensitivity Pneumonia
All patients with an interstitial lung disease displaying a fibrotic pattern should be screened for antigens that may cause hypersensitivity pneumonia.
If there is a temporal association between antigen exposure and symptoms OR if symptoms change over time, hypersensitivity pneumonia should come across as the most likely diagnosis.
The presence of lymphocytosis in bronchoalveolar lavage may also aid in diagnosis.
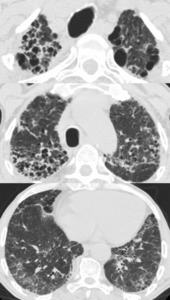
Fig. 7: HRCT of a 78-year-old male patient with chronic hypersensitivity pneumonia: subpleural, predominantly reticular densities, primarily located in the upper lobes. Honeycombing pattern: "possible". There is discreet ground-glass and no significant micronodules.
Collagen related diseases, including rheumatoid arthritis and Sjogren's, should also be included in the differentials.
Rheumatoid Arthritis
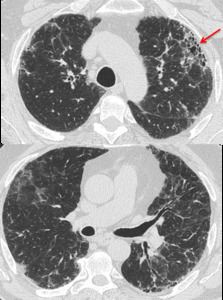
Fig. 8: HRCT of a 76 year-old woman with Rheumatoid Arthritis. There is substantial reticulation and honeycombing in the upper lobes, mainly located anteriorly (red arrow). This finding is known as the anterior upper lobe sign, described as being more common in patients with autoimmune disease.
Sjogren Syndrome
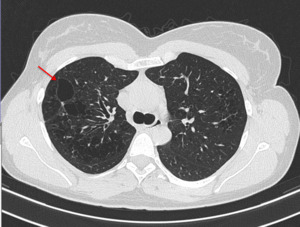
Fig. 6: HRCT of a patient diagnosed with Sjögren syndrome. Imaging features include centrilobular emphysema and several irregularly sized cysts (red arrow).
Additional difficulties in the diagnosis
Emphysema and fibrosis
Diagnosing IPF may be difficult in smoking patients with concomitant emphysema, this being present in up to one-third of patients with IPF.
Areas of emphysema coexistence should always be described in the report as they have implications for patient prognosis and treatment.
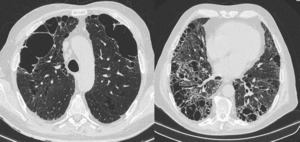
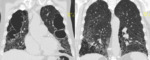
Fig. 9: HRCT of a 77-year-old male taking pirfenidone for a year, O2: 5L / min at rest; SatO2: 89%. Bullous emphysema takes up a significant part of the anterior segments of the upper lobes. In the lower lobes, fibrotic changes predominate over emphysema, with honeycombing and traction bronchiectasis.
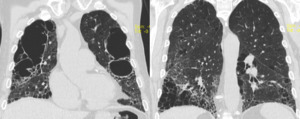
Fig. 10: HRCT of the same patient of the previous picture: coronal reformat. There is lower lobe volume reduction and the presence of a relative spatial independence between processes of fibrosis and emphysema.
Antifibrotic drugs
Pirfenidone and nintedanib have shown to be effective in reducing rates of disease progression compared with placebo. Despite that, the disease is neither fully stopped and, unfortunately, patients continue to experience lung function decline while on treatment.
Both agents target the fibrotic cascade, acting on the reduction of fibroblast and myofibroblast production.4
Pirfenidone has been shown to act on multiple targets, including transforming growth factor-β-triggered events, mediated through glioma-associated oncogene homolog 2.4
Nintedanib is a tyrosine kinase inhibitor that blocks intracellular signalling of platelet-derived growth factor receptors, fibroblast growth factor receptors and vascular endothelial growth factor receptor.4
After antifibrotic treatment, it is not expected to occur any improvement in the fibrosis extent in HRCT due to the irreversibility associated with these findings. Despite this, serial CT examinations can detect a slowdown of the progression of pulmonary fibrosis. Radiologists should clearly state on the reports if there are changes in lung volume, a progression of bronchiectasis, the extension of lesions, with an emphasis on honeycombing, reticulation, and appearance of consolidations.
The process of analyzing an HRCT of a patient with IPF (whether he is taking an antifibrotic drug or not) involves a thorough analysis of each lobe, assessing lung density, volume, reticular, fibrotic changes and search for potential complications such as infection.
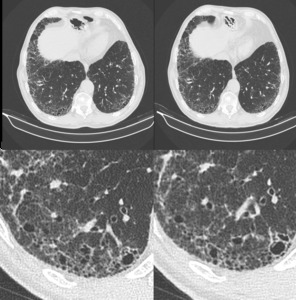
Fig. 15: HRCT of a 72 year-old patient taking pirfenidone for IPF for over 18 months. Left: before treatment. Right: follow-up. Lung volume is similar as well as the honeycombing and reticulation component of disease. Also noteworthy is that the use of a magnified view over the affectted regions leads to better assessment to possible small changes, that did not occur in this particular case.
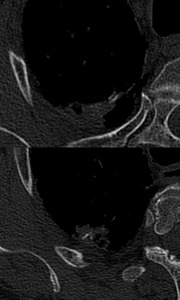
Fig. 14: Diffuse pulmonary ossification. High attenuating structures, displaying a dendriform pattern, are seen across the areas of fibrosing lung, in a patient with IPF. Although ossification is also present in fibrosing lung diseases other than IPF, this finding is more prevalent in the latter.
Patients with IPF often have often an unpredictable and heterogeneous disease course.
The evolution may be continuous or according to the occurrence of acute episodes that accelerate the decline of pulmonary function.
Progression can be assessed according to the forced vital capacity (FVC) rate of decline. Rapid progressors tend to show more pronounced effects when treated with antifibrotic drugs.
Currently, there is a lack of studies focusing on radiological changes after the initiation of antifibrotic drugs. The endpoints of major studies tend to focus on respiratory function tests and the slowdown of the decline of CVF.1
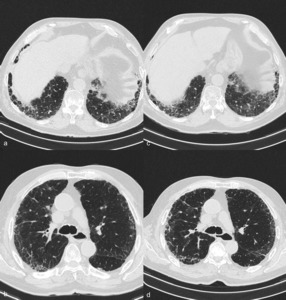
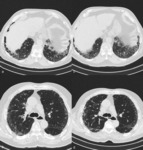
Fig. 11: HRCT of a 80 year-old male patient, under pirfenidone. Follow up time: 9 months. Direct comparison between the two studies shows moderate stability of the findings. The honeycombing component is mild and heterogeneously distributed. Ground glass is moderate as well as the reticulation component. Functional respiratory tests were almost the same between both studies, with DLCO decreasing by 2% (from 76 to 74%).
According to a study by Ballestra et al, fibrotic changes such as honeycombing tend to increase despite “stable disease.” 1
The presence of ground-glass opacities (GGO) in IPF is still an ongoing debate. According to the same study, GGO tend to increase with disease progression and stabilize when the disease is unchanged.
In the context of IPF, ground glass reflects initial fibrosis when located close by already established fibrosis. It is associated with the presence of cells in the alveolar/interstitial space. Patients with rapid progression tend to have more extensive cellular/immune-inflammatory infiltrates. 1
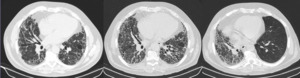
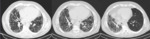
Fig. 12: Serial HRCT of a patient with IPF under antifibrotic drug treatment. From left to middle image (follow-up time: 7 months): there is an increase in fibrosis and lung density and reticular pattern despite the use of pirfenidone. Eventually, the patient required a lung transplant. Right image: after transplant - there is compensatory left hyperinflation and reduction of the right lung volume with a significant improvement in the patient's quality of life.
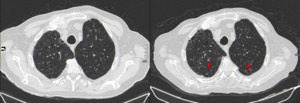
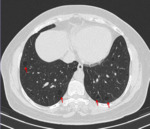
Fig. 13: HRCT of a patient treated with nintedanib. Follow-up time: 1 year. There is a global stability of the fibrosis. Despite this, there are numerous micronodules on both upper lobes (red arrows), related to respiratory infection.
Imaging plays a particularly important role in the monitoring of patients treated with antifibrotic drugs in three main areas:
- HRCT captures early changes in density and morphology that may be present before pulmonologist takes notice.
- Identifying subtle abnormalities that are not captured by a lung function test.
- Identifying complications of IPF, such as lung cancer or indirect signs of pulmonary hypertension.