I.Introduction
Anderson Fabry disease (FD) is a progressive X-linked genetic disorder (mutations in the GLA gene) caused by absent or deficient alpha-galactosidase A. It results in the deposition of globotriaosylsphingosine (Gb3) and its derivative (lyso-Gb3) within lysosomes (1).
Multi-organ involvement is a characteristic during FD cardiac variant is usually limited to the heart. The renal and cardiac disease may be life-threatening.
Cardiac damage is a turning point in FD.
Common clinical manifestations of classic form are:
- neuropathic pain
- hypohidrosis
- angiokeratoma
- cornea verticillata (pathognomonic)
- lymphoedema
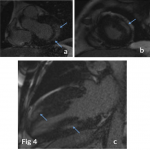
-Myocardial damage includes hypertrophy, inflammation, and ultimately fibrosis at an advanced stage (2).
-Clinical presentation of cardiac involvement goes from arrhythmia, restrictive cardiomyopathy to heart failure. The leading cause of mortality in FD is arrhythmia.
-Enzyme replacement therapy and chaperone molecule therapy may be used during FD.
-Medications' effectiveness is a particularity of FD, as opposed to other infiltrative cardiomyopathies. Treatment stabilizes or even reverses the disease's course (3). Several randomized studies have established the medications' efficacy and safety (4,5).
II.Diagnosis and monitoring
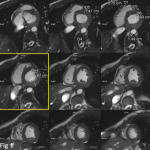
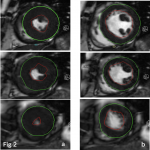
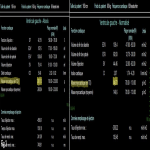
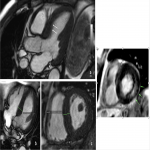
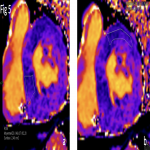
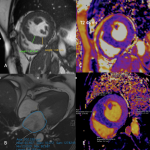
- In case of unexplained left ventricular (LV) hypertrophy, ⍺-galactosidase A activity on dried blood spots on filter paper can be used for FD screening (6). CMR showing a decreased T1 mapping can be a mean of diagnosing FD.
- The case of common clinical manifestation or genetically proven FD, plasmatic biomarkers, such as lyso-globotriaosylceramide (lyso-Gb3), are useful but need to be associated with GLA gene analysis for the diagnosis of FD (6).
- When FD is suspected and during the follow-up, transthoracic ultrasound with strain measurement and CMR with gadolinium injection is indicated (6).