I/ The breast imaging reporting and Data system(BIRADS)
The most widely used system for classification is the American College of Radiology Breast Imaging Reporting and Data System (BI-RADS).
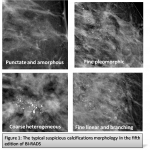
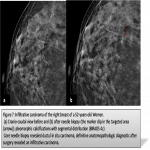
In the fifth edition of BI-RADS, The typical suspicious calcifications morphology include: ‘’amorphous, coarse heterogeneous,fine pleomorphic and fine linear branching’’.(figure1)
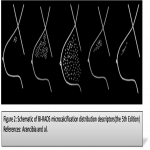
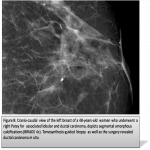
Distribution of calcifications has been simplified in the fifth edition by eliminating the terms scattered and clustered. The remaining distributions are termed “diffuse, segmental, regional, grouped, and linear”. The number of calcifications in the “grouped” distributionhas been revised to include a minimum of five calcifications within 1 cm of each other and a larger number of calcifications within 2 cm of each other (figure2)
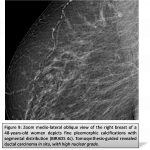
When microcalcifications present a grouped or regional distribution, amorphous, coarse heterogeneous, and fine pleomorphic calcifications represent a lower risk of malignancy and should be assigned to category 4b opposed to fine linear calcifications, which should be assigned to category 4c.Iflinear calcifications present a segmental, linear or branching linear distribution, theyshould be assigned to category BI-RADS 5.(figure3)
A solitary group of punctate calcification was regarded and biopsy was recommended if the calcification is new, increasing, in a segmental or linear distribution, or adjacent to known cancer
When there is an association of different types of microcalcifications, the final BI-RADS category will be that of those microcalcifications of higher grade.
The morphology of calcifications is far more important than stability and stability can only be relied on if the calcifications have a probably benign form.
II/ Techniques of histological proofand management of histological results
Biopsy is recommended when imaging of calcifications is not typically benign.
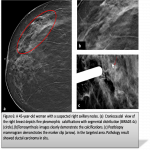
In general, calcification is biopsied using a stereotactic approach for localization.More recently, tomosynthesis-guided biopsy technology has become available; this is reported to be quicker and more effective for sampling low-contrast soft-tissue lesions because it requires less repositioning. Tomosynthesis-guided core biopsy may also be more comfortable for patients. For tomosynthesis-guided core biopsies, the patient may be sitting or be in the right or left lateral decubitus position (figure4)
When appropriate, biopsy can be performed using ultrasound. Successful identification of calcification on ultrasound relies on accurate localization of the cluster in the correct quadrant, distance from the nipple and depth below the skin surface. Calcification tends to be more conspicuous if there is any change in the adjacent soft tissue, and ultrasound-guided biopsy of calcification therefore may have a height yield of malignancy
The size of the biopsy needle varies with local protocols;the shortcomings of 14-G biopsies led to the introduction of larger cores assisted by vacuum to ensure retrieval, which also allowed multiple samples to be collected with a single percutaneous introduction. This allows the pathologist considerably more tissue for analysis improving diagnostic accuracy.
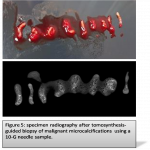
Immediate specimen radiography is invaluable to assess the adequacy of the specimen.(figure5)
A needle sample may be confidently used to establish a benign diagnosis, or a malignant diagnosis of invasive cancer, but underestimates the nature of disease in approximately 27% of cases when DCIS and indeterminate lesions such as atypia are present.
The diagnosis and management of indeterminate lesions will be discussed in a subsequent review. The distinction between atypia and low-grade in situ carcinoma depends on the extent of changes. If the abnormality measures >2 mm, or more than one duct system is involved, the lesion is best described as low-grade DCIS rather than atypia.
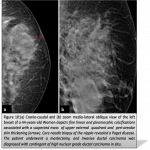
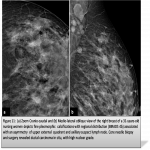
In light of the multidisciplinary discussion, surgical excision for lesions may be considered.The pre-operative radiological localization of the lesion with guide wires, or other means, is an essential step to aid the surgeon in removing the lesions with sufficient margins.(figure6-7)
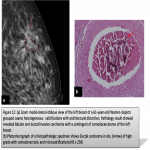
III/Diagnostic and prognostic value of microcalcifications (figure8-12)
In addition to their value in the early detection of breast carcinoma and accurate radiological diagnosis, the presence of microcalcifications often affects the extent of surgical intervention.
Microcalcifications are important in the pre-operative assessment of lesion extent and distribution and malignant-type calcifications should be included in the radiological extent of the tumor.
Multiple clusters of malignant microcalcifications indicate the involvement of multiple TDLUs in a malignant process and, as such, indicate multifocality of the tumor’s in situ component.
Patients with calcification-associated breast carcinomas have an elevated risk of metastatic spread compared to non-calcified cancers. Microcalcifications may appear in metastatic deposits of breast carcinoma especially lymph nodes.
Certain types of microcalcifications are associated with negative genetic and molecular characteristics of the tumor and unfavorable prognosis. Associations between duct-centric calcifications and hormone receptor negativity and HER2 positivity are well-documented in CIS, especially in cases with neoductgenesis.
Microcalcifications localized in the larger ducts (duct-centric, casting-type microcalcifications) represent an independent negative prognostic marker compared to lesions containing other types of microcalcifications and to non-calcified lesions and a higher risk of breast cancer death has been shown in the presence of casting-type calcifications compared to cancers not associated with this mammographic abnormality.
Casting-type calcifications are dystrophic deposits of calcium salts in the necrotic debris filling the lumen of the ducts involved with CIS. They follow the branching pattern of the ductal tree of the sick lobe (called comedo CIS). This subtype of microcalcifications associated with unfavorable genetic and molecular characteristics, complex subgross morphology, high histology tumor grade, increased metastatic potential, and decreased disease-free and overall survival.
All dystrophic calcifications represent a late event in the natural history of cancer development. They appear after tumor cell proliferation, cell death, and the formation of central necrotic debris within the lumina, which are all required for calcium salt deposition. This means that the cancer may exist long before it becomes calcified and is detected by mammography.
Powdery calcifications reflect the presence of a large number of psammoma body-like calcifications within TDLUs. They do not require necrosis for their development. Their appearance is associated with chemical alteration of the secretion within the lumina of the acini of the TDLUs. As such, they represent an early event in the natural history of CIS development. Many benign lesions also include such calcifications (adenosis, involution, apocrine change), leading to difficulties in radiologically differentiating lesions calcified in this manner.