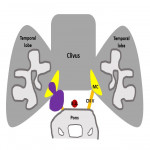
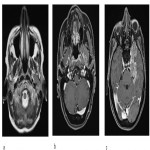
The preferred imaging modality in depicting structure, contents and relationship of Meckel's cave with adjacent structures is MRI, using protocol that includes high-resolution sequences in three planes. High-resolution heavily T2-weighted volumetric sequences are used to explore the cisternal course of the cranial nerves, Gasserian ganglion (GG) and trigeminal rootlets within MC. High resolution T1-weighted fat-suppressed imaging with and without contrast injection depicts fat planes at the skull base, characterizes and localizes lesions, visualizes bone marrow invasion, meningeal invasion and perineural spread.
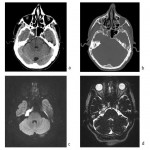
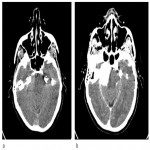
Meckel's cave presents as triangular shaped CSF-filled space near the petrous apex on the soft tissue window of non-enhanced CT (NECT). Bone window of NECT is useful in visualization of skull base and foramina.
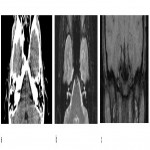
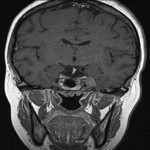
Dominant contributing factor to signal intensity of the MC on MRI is CSF making MC hypointense on T1WI and hyperintense on T2WI. Trigeminal ganglion is placed in the anterior inferolateral aspect of the MC and due to perineural vascular plexus normally enhances on post-contrast images (Figure 2).
Key abnormal imaging findings that suggest MC pathology are the absence of a normal “empty” MC on T1WI, bulging of the lateral wall on T1WI, and pathologic contrast enhancement greater than in the GG on gadolinium-enhanced T1WI. Other signs are nerve enlargement with perineural fat plane effacement, osseous foraminal erosion or enlargement, and trigeminal cistern CSF effacement.
Broad spectrum of entites can occur in Meckel's cave and its vicinity.
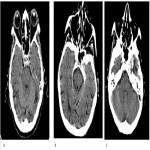
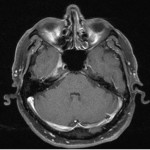
Normally empty MC can be site for deposition of calcium or fat. The latter may be seen in obese individuals, those taking corticosteroids and patients with Cushing syndrome, but in the absence of these conditions they have no significance. In traumatized patients with radiological signs of brain injury, subarachnoid hemorrhage can also be found in Meckel's cave (Figure 3).
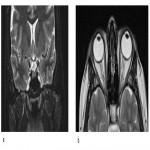
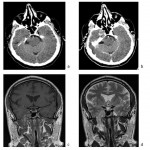
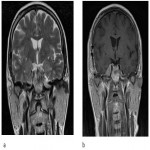
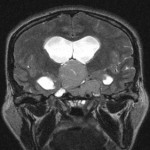
Size of the trigeminal cavity should also be assessed as it was shown that enlarged cavity with bilobed appearance together with other signs (papilledema, enlarged subarachnoid space around cranial nerves etc.) reflects idiopathic intracranial hypertension (Figure 4).
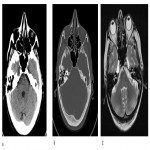
Shape of the MC can be abnormal in other conditions such as petrous apex cephalocele. It is a congenital or acquired herniation of the posterolateral wall of the MC into the petrous apex with consequent smooth remodeling of its bone. Signal intensity of cephalocele matches CSF on all sequences. (Figure 5).
Another cystic lesion affecting MC is cholesterol granuloma. Composed of granulation tissue prone to bleeding, this expansile well-marginated lesion with thinned overlying bone, unlike cephalocele shows central high signal intensity on T1WI, T2WI and FLAIR due to cholesterol and methemoglobin component.
Cholesteatoma in this location is usually congenital and causes variable degree of bone destruction, and unlike cholesterol granuloma shows low to intermediate signal on T1WI and can partially attenuate on FLAIR. However, cholesteatomas with minimal bone destruction and white cholesteatoma variant with high cholesterol component can be hard to distinguish from cholesterol granuloma. Diffusion weighted imaging is useful in this differentiation as cholesteatoma, besides name and location, is identical to epidermoid cyst, therefore appearing very bright on DWI (Figure 6)
.
Apart from congenital abnormalities, inflammatory conditions can also involve MC. An example is Tolosa-Hunt syndrome characterized by intense contrast enhancement during the active phase with resolution of enhancement following steroid treatment (Figure 7).
Tumors affecting MC display different morphological features depending on their predominant localization which can be either middle or posterior fossa. Tumors with components in both fossae i.e. in cerebellopontine cistern and trigeminal cavity exhibit dumbbell shape with waist at constricting trigeminal porus (Figure 8).
Two most common neoplasms of this region, scwannoma and meningioma, are sometimes hard to distinguish. In general, nerve sheath tumors such as schwannoma will be more central within the cave, growing along the course of the nerve causing nerve and foraminal enlargement and demonstrating high signal on T2WI with moderate-to intense heterogeneous enhancement on T1WI postgadolinium image (Figure 9).
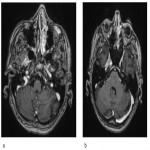
Meningiomas arise from dura in or around Meckel's cave and therefore have peripheral localization, usually low T2WI signal and uniform vivid enhancement with enhancing dural tail (Figure 10 and 11).
Despite imaging features, distinction between these entities can be unclear even at pathohistological analysis.
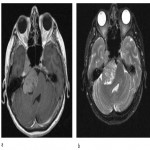
Retrograde perineural spread of head and neck tumors most commonly involves trigeminal and facial nerve. Tumors located along the distribution of mandibular division (V3) of trigeminal nerve have ability to use perineural spread to disseminate into masticator space and then through foramen ovale enter the Meckel's cave. Squamous cell carcinoma is the most common histological subtype of all head and neck tumours and therefore accounts for largest number of cases of perineural spread. However, tumor with the highest association with perineural spread is adenoid cystic carcinoma that accounts for only 1-3% of head and neck malignancies but has perineural invasion rates as high as 60%, especially if it occurs in minor salivary glands (Figure 13 and 14).
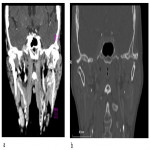
Parotid malignancies have the potential to invade auriculotemporal branch of V3 and spread into MC (Figure 15).
Additionaly, any other tumor that involves masticator space, pterygopalatine fossa or cavernous sinus can be associated with perineural spread with imaging characteristics dependent on the primary tumor .
Besides perineural spread, other mechanism of spread are hematogeneous and lymphatic dissemination from distant tumors, such as breast carcinoma (Figure 16)
, or direct extension of tumors, like pituitary macroadenoma invading Meckel's cave through cavernous sinus (Figure 17)
.