We can broadly divide such findings into normal (age-related) intracranial calcification, normal cystic variants, vascular variants and normal meningeal/calvarial variants.
A. Normal intracranial calcifications: Occur as part of normal ageing of brain, without a pathological cause. As expected, they are more prevalent in older age groups.
The common locations involving such calcifications include:
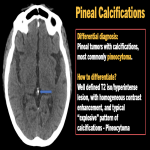
- Pineal gland [1] (fig.1)
- Most common ( ~ 71% ).
- Morphology varies with age- young children showing single and punctate calcification while bigger and relatively coarse calcification in older individuals.
MIMIC: In young children, larger and multiple pineal calcifications (> 1 cm) tend to be suspicious and warrant further investigation of the pineal gland for detection of any underlying tumor.
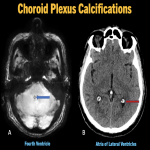
- Choroid Plexus [1] (fig. 2)
- Second most common site.
- Usual sites- atria of lateral ventricles; third and fourth ventricles(less common), especially in children less than 9 years of age.
MIMIC: Further assessment may be indicated in cases of the extremely unusual sites, which include body of lateral ventricles, foramen of Monroe and roof of third ventricle.
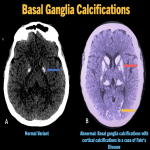
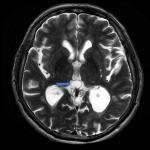
- Basal ganglia (fig. 3)
- Another rather common finding, usually involving globus pallidi. They appear symmetric with a fine punctate pattern.
MIMICS:
- Idiopathic conditions like Fahr’s disease
- Toxic involvements (Carbon monoxide, lead poisoning),
- Infections like TORCH, CNS tuberculosis
- Metabolic disorders like parathyroid dysfunction and hypothyroidism
- Mitochondrial diseases like MELAS, and
- Tuberous sclerosis.
A clinical, laboratorical and radiological approach can help make a diagnosis of the same. [2]
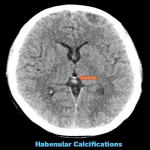
- Habenula (fig. 4)
– Usually present with curvilinear calcification, anterior to the pineal gland. A higher existence of habenular calcifications are seen in cases of those suffering from Schizophrenia.
- Dural – Common locations include tentorium cerebelli and falx cerebri.
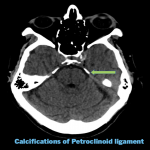
- Petroclinoid ligaments (fig. 5)
- Dentate nucleus of cerebellum
- Superior Sagittal Sinus
B. Normal cystic variants
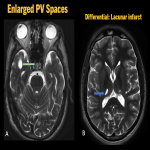
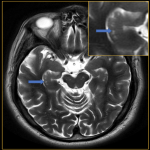
a. Perivascular Spaces (PV Spaces) [3] (fig. 6)
:
-
- Also known as Virchow-Robin (VR) spaces, these are pia lined spaces surrounding the vessels, along their subarachnoid course.
- Enlarged perivascular spaces can be detected on MR and CT imaging, with common locations along the lenticulostriate arteries (basal ganglia region), perforating medullary arteries (centrum semiovale) and in the midbrain.
- Enlarged VR spaces are well-defined, oval to round structures with bilaterality, and size of less than 5 mm.
- They usually do not show surrounding parenchymal gliosis, and occasionally demonstrate a vessel coursing through them.
Catch: Atypical VR spaces introduce a diagnostic dilemma, as they can present with bigger size, unilaterality, irregular morphology, surrounding gliosis and mass effect.
Differential diagnoses: Lacunar infarcts (figure. 6), infections like cryptococcosis and neurocysticercosis, cystic periventricular leucomalacia, multiple sclerosis and cystic neoplasms.
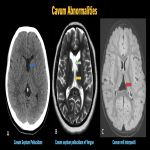
b. Cavum septum pellucidum (et vergae) and Cavum veli interpositi (fig. 7)
: These are abnormalities seen due to persistence of embryonic midline cavities.
Cavum septum pellucidum (CSP) - triangular space existing between the two leaflets of the septum pellucidum, seen as a cystic area between the frontal horns of lateral ventricles.
Cavum vergae (CV) - posterior extension of CSP extending posteriorly till the splenium of corpus callosum, and bounded by the body of corpus callosum superiorly.
Cavum veli interpositi (CVI) - more posteriorly, between the atria of lateral ventricles, posterior to the foramen of Monro and bounded on either side by the crura of the fornix.
c. Pineal cysts (fig. 8)
: These are homogenous fluid containing cystic structures. usually homogenous in attenuation/intensity, thin walled and show occasional peripheral calcification, and rim enhancement. The prevalence is highest for teens and young adults.
Catch: Usually they are clinically insignificant and do not require follow up, sometimes it is difficult to distinguish them from tumors like pineocytoma and germ cell tumors, especially those having large size and nodular enhancement.
d. Hippocampal cysts (Hippocampal sulcus remnant cysts) (fig. 9)
: Occur due to incomplete closure of the hippocampal fissures, and appear bilateral, small (1-2 mm) with same attenuation/signal intensity as CSF on imaging.
e. Rathke cleft cyst: Most commonly asymptomatic and incidentally detected, these demonstrate T1 variable and T2 hyperintense signals with no post contrast enhancement, and a typical intracystic nodule appearance.
f. Mega cisterna magna (fig. 10)
: a size criteria of > 10 mm is taken to define mega cisterna magna, which has no clinical implications.
Differentials: Blake pouch cyst and vermian hypoplasia, which can be distinguished from mega cisterna magna by the presence of hydrocephalus and vermian abnormalities, respectively.
C. Vascular variants:
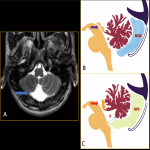
a. Variations involving venous sinuses
: The transverse sinus is the most common cortical venous sinus to show anatomic variations, with hypoplasia of one of the sinuses seen quite commonly (fig. 11). A close differential of the same is transverse sinus thrombosis, which can be distinguished from the normal variant by the presence of filling defect and a normal bony groove for the transverse and sigmoid sinuses. The groove is usually hypoplastic along with the sinus in the normal variant.
Hypoplasia of the superior sagittal sinus is another not so uncommon entity, with variations primarily affecting the anterior extent of the sinus. [4]
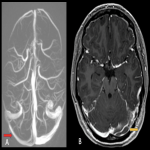
b. Developmental venous anomaly (fig. 12)
: Previously known as venous angiomas, these developmental anomalies are characterised by their classic “medusa head” appearance on venographic studies. The appearance is produced by multiple veins draining into a larger collecting vein, which further drain into superficial or deep veins.
Catch: The occasional association of DVAs with the co-existence of cavernomas should warrant a careful search for ruling out the same.
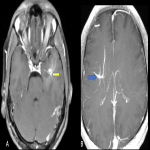
c. Capillary telangiectasia [5] (fig. 13)
: Most commonly located in the pons and cerebellum, they do not require any further management. On MR imaging, these are usually inconspicuous on non-enhanced sequences (rarely they may be mildly T2-hyperintense and T1-hypointense), while on enhanced scans, they show lace-like enhancement without any mass effect, with blooming artefact often seen on susceptibility-weighted images.
D. Dural and meningeal variants:
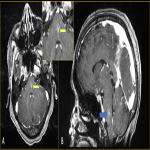
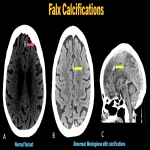
a. Osseous metaplasia of falx/falx ossification (fig. 14)
: Osseous areas within the falx are a rather normal finding, with no usual clinical significance.
Catch: Gorlin-Goltz syndrome (Nevoid Basal cell carcinoma syndrome) is an autosomal dominant condition with a high incidence of ossified falx, along with findings like basal cell carcinomas, odontogenic keratocysts and medulloblastomas. The radiologist should be aware of the same and should search for these findings to rule this entity out.
Small calcified meningiomas (parafalcine) can mimic osseous falx, requiring contrast studies for typical enhancement patterns and dural tails.
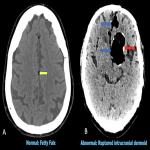
b. Fatty falx (fig. 15)
: Well-defined fat-attenuation areas within falx cerebri. They are benign, and differentials include ruptured dermoid cysts and intracranial lipomas.
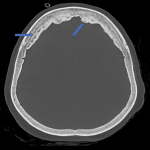
c. Hyperostosis frontalis interna/frontoparietalis (fig. 16)
: Visualised as irregular and nodular thickening of the frontal bone, along its inner table. If the parietal bones are involved, the term hyperostosis frontoparietalis is used. Differentials include: Paget's disease, sclerotic metastasis, fibrous dysplasia and acromegaly.
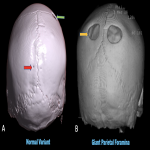
d. Parietal foramina [6] (fig. 17)
: Seen in the paramidline regions of bilateral parietal bones, through which parietal emissary veins pass. Normally they are very small in size, very large giant parietal foramina are sometimes seen, which can have co-existing intracranial midline abnormalities like persistent falcine sinus, occipital cortical malformation and atretic occipital encephalocele.