Typical findings of CNS lymphoma:
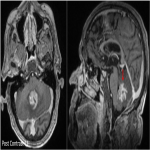
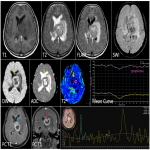
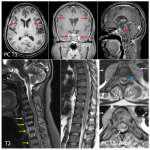
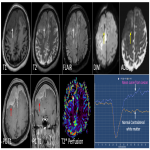
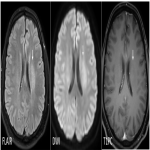
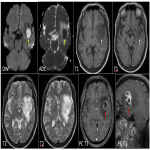
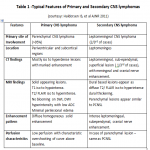
PCNSL have predominantly parenchymal involvement. It is a solitary, hypercellular tumor with disruption of blood brain barrier showing homogenous enhancement, poor neovascularity and predilection for periventricular or subcortical regions(figure 1). The imaging features of primary and secondary CNS lymphoma are illustrated in table 1.
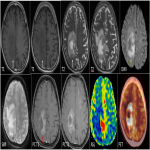
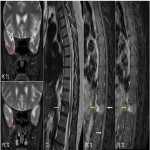
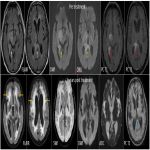
Key features- Characteristic deep depression on the surface of the enhancing tumor termed as “Notch sign”is not uncommon(figure 1).This pattern of enhancement suggest irregular growth pattern as well as pliable and infiltrative property of the tumor. Other pattern of enhancement seen is“serration shape” in the margin of lesions especially in cerebellar lesions(figure 2).
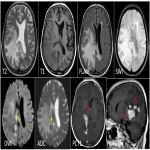
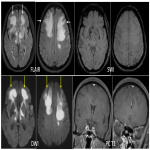
Secondary CNS lymphomas have predominant leptomeningeal involvement showing leptomeningeal and subependymal enhancement.(table 1)(figure 3)
Atypical findings of primary CNS lymphomas:
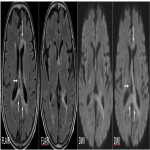
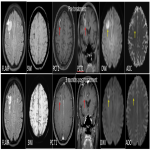
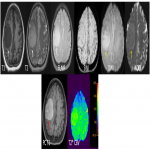
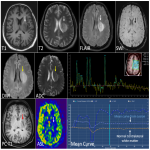
Presence of central necrosis, hemorrhage, calcification, areas of cystic degeneration, non-enhancement(figure 12)are unusual in case of PCNSL.(figure 4 – 7, 10)
Dural based lymphomas are rare, diffusely infiltrating plaque-like meningeal lesions with extensions into falx, tentorium and cerebral sulci and can mimic extra-axial hemorrhages or meningiomas at presentation.(figure 11)
Advanced imaging findings in primary CNS lymphomas:
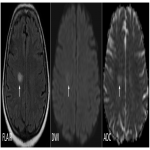
Diffusion Weighted Imaging(DWI):Lymphomas being highly cellular, they are usually hyperintense on DWI, with low ADC. ADC values not only have utility in differentiating PCNSL from other brain tumors but also have role in prognostication. Higher ADC values is an excellent pretreatment predictive biomarker for clinical outcome. Shorter progression free survival(PFS) and overall survival(OS) were predicted by low ADC levels.Patients with prolonged PFS and OS after therapies had a significant increase in post-therapeutic ADC values.
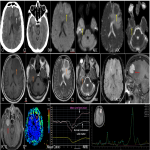
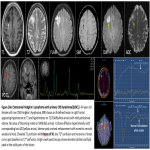
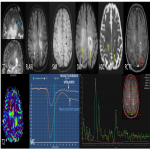
Dynamic Contrast Enhanced(DCE) Perfusion –Lymphomas are highly cellular lesions with disruption of blood brain barrier, providing high flux for contrast. Ktrans (flux constant) in lymphomas is generally reported to be moderate to high(figure 10a). Individual studies have shownhigher values of Ktrans in lymphoma when compared to other brain lesions like glioblastoma and metastases. However, this is not always the casedue to the tumor's hypovascular nature.Ve is defined as the volume of the extravascular extracellular space and is thought to correlate with tumor cellularity. Ve is consistently been found to be higher in lymphoma than in glioblastoma and brain metastases
Dynamic Susceptibility Enhanced(DSC) Perfusion- Lymphoma being a tumor with poor neovascularization show a low rCBV with mean curve showing minimal fall in signal intensity.
Characteristic “overshooting above the baseline” of mean curve noted in lymphomas is attributable to superadded T1 effect due to extravascular leakage of contrast(figure 1 & 4). However this is not specific to lymphomas, can occur in mural nodule of pilocytic astrocytoma’s.
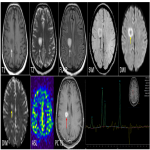
MR spectroscopy: They show depleted NAA, elevated choline peak and high Cho/Cr ratio. Characteristic elevated lipid peak noted in the solid component in the lesion. Another differentiating feature from other mimics, where necrotic core shows lipid peak.(figure 1& 5)
Common differentials or Mimics of primary CNS lymphomas and key differentiating features:
Common mimics are- High grade gliomas(figure 15), Metastases, pyogenic abscesses(figure 17) and tumefactivedemyelination(figure 14 & 16). They general imaging features and key differentiating features are as described in Table 2 & Table 3.
International Primary CNS Lymphoma Collaborative Group (IPCG) – recent guidelines for lymphoma treatment response assessment:
Imaging Features of treatment response:
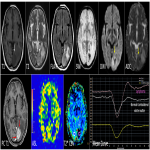
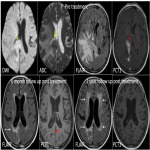
- Disappearance of contrast enhancement is imaging hall mark of treatment response.(figure 7b & 10b)
- Decrease in size, number and extent of visible lesions.
- Subsequent higher ADC values on follow up scan-emerging indicator of response assessment.
Assessment of treatment response:
All studies for response assessment should be performed ideally every 2 months during active therapy or at the time when therapeutic modality is changed. Imaging should also be performed 2 months after completion of all planned therapy to assess overall treatment response. All complete responses should be confirmed by repeat imaging.
Assessing treatment response of PCNSL, includes post treatment contrast enhanced brain imaging, presence or absence of corticosteroid administration, ocular examination and CSF findings. Here describing only, the imaging findings, relevant to our exhibit.(figure 7b & 10b)
- Complete response [CR]- defined as complete disappearance of enhancing lesion. Should have discontinued use of all corticosteroids for at least 2 weeks.(figure 7b)
- Complete response -unconfirmed [CRu]- fulfills criteria for CR with minimal abnormality- possibly post interventional or continue to require corticosteroid therapy.
- Partial response [PR]-defined as > 50% decrease in the enhancing lesion as compared with baseline imaging.(figure 10b)
(**In the setting of primary leptomeningeal lymphoma, PR is not recognized; all patients should be categorized as CR, CRu, stable disease, or progressive disease)
- Progressive disease [PR] - defined as >25% increase in the contrast enhancing size of the lesion seen as compared with baseline (comparison should be made to the smallest lesion, in case of multiple lesions). Appearance of any new lesions.
- Stable disease [SD]- Defined when the response can neither be categorized into partial response or progressive disease.
Post treatment changes in lymphomas:
- Progressive white matter hyperintensity - Progressive white matter changes even when tumors continue to show response based on enhancement. The risk factors of developing these white matter changes include the elderly (age >60), those receiving neurotoxic chemotherapies, and those who have received prior radiation. Impaired biopterin metabolism as well as accumulation of homocysteine and adenosine may be contributing to this phenomenon. Some have proposed that the impact of methotrexate on the glial cells may have an influence and they also have an impact on neurocognitive outcomes. Radiation-induced white matter injury varies from demyelination, gliosis, edema to coagulation necrosis.(figure 18)
- Cerebral volume loss – progressive white matter changes leads to cerebral volume loss on a long term basis.(figure 18)