1) Ultrasound guided compression:
60 year-old lady with underlying diabetes and hypertension presented with non-healing infected 5th toe ulcer. She was referred to interventional radiology for lower limb angioplasty to aid the healing of her wound.
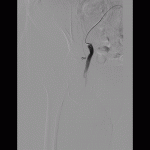
An antegrade puncture approach was made to obtain access to the right common femoral artery (CFA). Following uneventful plain old balloon angioplasty (POBA), arterial sheath was removed followed by 15 minutes manual compression. No immediate haematoma observed post-compression.
However a small pulsatile haematoma was detected during routine next day follow up by the interventional radiology team. Doppler assessment revealed the presence of a CFA pseudoaneurysm with a long but thin neck.
After studying the pseudoaneurysm morphology, an ultrasound guided compression was then performed. Adequate fentanyl and midazolam were administered to reduce pain and anxiety, thus aiding in accurate compression.
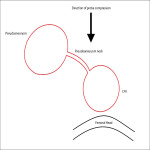
The compression was made with the aim of compressing the neck of the pseudoaneurysm, purposely focusing the pressure and visualization of the ultrasound probe at the site of the neck without needing to visualize the pseudoaneurysm sac.
As with routine manual compression, compression was done against a bony structure (femoral head). Compression was performed for 30 minutes, following which the pseudoaneurysm neck was no longer visualized. The pseudoaneurysm sac became smaller but appeared heterogenous with no signal detected on Doppler interrogation, indicative of clot formation.
Ultrasound guided compression can be a cheap and useful tool in dealing with pseudoaneurysms. Factors predicting failure of ultrasound guided compression are as follow1:
- Ongoing anticoagulation
- Short aneurysm neck length (<10mm)
- Obesity
- Pain
- Operator fatigue
2) Vascular closure device:
45-year-old gentleman with hypertension, diabetes and morbid obesity underwent lower limb angioplasty for a non-healing left foot ulcer. The patient had an incomplete angioplasty 1-week prior, abandoned due to mid-procedure allergic reaction. The procedure was repeated successfully. Both procedures were performed via antegrade approach. Post-procedure manual compression was applied both times.
Two days following the second procedure, the patient started having puncture site pain, with a small vague pulsatile lesion clinically palpable.
A CFA pseudoaneurysm is detected on ultrasound. However visualization of the entire pseudoaneurysm was difficult owing to the presence of subcutaneous air just anterior to the false aneurysm. Visualization of the neck of the pseudoaneurysm, whilst possible, was also quite challenging.
A catheter angiogram was performed from the contralateral side to assess the pseudoaneurysm, with a view for embolization. The catheter angiogram demonstrated the presence of a relatively small neck, multilobulated left CFA pseudoaneurysm. The pseudoaneurysm was also relatively aligned to the CFA parent vessel.
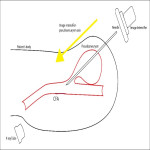
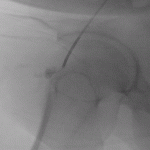
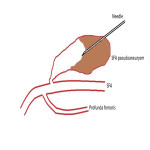
Decision was then made to percutaneously puncture the pseudoaneurysm under roadmap fluoroscopic guidance using an 18G puncture needle. The frontal image intensifier-tube axis was angled caudo-cranially so that it became parallel to the axis of the pseudoaneurysm neck. The lateral tube was maintained at its standard parallel to the table position.
The caudo-cranially angulated image intensifier-tube was used at the main guide, making sure that the needle appearance remaining pinpoint on the angulated frontal image. The lateral axis was used to make sure that a single wall puncture was made. This enabled exact puncture at the pseudoaneurysm neck and into the CFA. An 0.035” glide guidewire was then advanced retrogradely into the CFA.
Using the guidewire, a Perclose Proglide™ (Abbott Vascular Devices, Redwood City, CA, USA) suture mediated vascular closure device was introduced into the CFA through the pseudoaneurysm neck. The suture-based vascular closure device was then deployed in its usual fashion.
A check catheter angiogram was performed showing closure of the pseudoaneurysm. A small indentation on the CFA on the lateral view – indicating the site of the closure.
3) Glue embolization
35-year-old lady with hypertension and morbid obesity was referred for left flank haematoma and haematuria following renal biopsy. 4-phase CT revealed an actively bleeding left renal artery pseudoaneurysm, for which she underwent emergency embolization via right CFA access. The procedure went smoothly and the bleeding pseudoaneurysm was successfully embolized. Her right CFA sheath was subsequently removed with manual compression performed for 20 minutes.
However she started complaining of pain at her femoral access site on day 1 post-procedure. A small neck CFA pseudoaneurysm was detected on ultrasound, with visualization being partially obscured by subcutaneous air.
Catheter angiogram was performed from the contralateral side to better depict the pseudoaneurysm morphology, with a view to stem the pseudoaneurysm.
A microcatheter was advanced through the parent 5Fr Cobra catheter to cannulate the pseudoaneurysm.
A gentle confirmatory angiogram was performed with the microcatheter within the pseudoaneurysm neck. Two factors were examined:
- placement accuracy of the microcatheter, and
- pseudoaneurysm flow haemodynamics, estimating the volume of contrast that refluxed into the CFA. This acted as a mock run prior to liquid embolization.
Considering the 2 factors, histoacryl glue embolization was then performed via the microcatheter. A mixture of 50% glue and 50% lipiodol was used, ensuring high viscosity thus reducing the risk of backflow/reflux. This enabled stasis of glue within the pseudoaneurysm with no glue reflux into the CFA.
Prior to microcatheter retrieval following complete closure of the pseudoaneurysm, the parent catheter was advanced over the microcatheter towards the pseudoaneurysm neck. This was done to wedge the glue cast within the pseudoaneurysm and reduce the risk of glue migration.
Microcatheter was then retrieved and confirmatory catheter angiogram performed showing complete closure of the pseudoaneurysm.
4) Endovascular stenting and percutaneous injection
75-year-old lady presented with features of intra-abdominal sepsis and acute renal failure which required urgent haemodialysis. The nephrology team performed multiple unsuccessful attempts to insert a femoral dialysis catheter on both her groin regions. She subsequently developed an enlarging pulsatile haematoma on both groin regions, worse on the right.
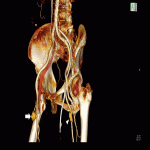
A CT angiogram revealed bilateral superficial femoral artery (SFA) pseudoaneurysms (larger on the right) and a right SFA-femoral vein arteriovenous fistula (AVF).
An ultrasound assessment 2 days later showed enlarging right SFA pseudoaneurysm, with the left SFA pseudoaneurysm being partially thrombosed. Decision was then made to insert a covered stent over the right SFA via left CFA approach in order to simultaneously close both the pseudoaneurysm and AVF.
Following cannulation of the right external iliac artery and placement of a stiff Hi-Torque Supra Core® Guidewire (Abbott Vascular, part of Abbott Laboratories, Abbott Park, Ill), a 6Fr Fortress crossover sheath (Biotronik, Berlin, Germany) was placed for better support during stent delivery. An 6mm BeGraft Peripheral Stent System (Bentley Innomed, Hechigen, Germany) balloon expandable stent was deployed. Whilst the pseudoaneurysm was closed, some residual AVF persisted thus stent post-dilatation performed resulting in total AVF closure.
Percutaneous puncture of the smaller left SFA pseudoaneurysm was then performed using an 18G puncture needle under ultrasound guidance. Gelfoam was then delivered percutaneously, under continuous ultrasonography.
Care was taken not to cause gelfoam dispersal into the parent SFA. Gentle introduction of the gelfoam, along with preferential blood flow into the pseudoaneurysm will aid in compacting the gelfoam from within the distal portion of the pseudoaneurysm. The needle was then subsequently advanced with further gelfoam injection until most of the pseudoaneurysm was packed. Residual pouch persisted, which was seen to spontaneously thrombosed on ultrasound evaluation a few days later.
The cases shown showcased a few common risk factors for iatrogenic pseudoaneurysms. A more extensive list is as compiled by Stone et al2:
- Advanced age
- Female>Male
- Increased BMI
- Low platelet count
- Anticoagulation/antiplatelet
- Procedure-related factors:
- Puncture below CFA bifurcation
- Bigger sheath size
- Interventional as opposed to diagnostic procedures
- Urgent as opposed to elective procedures
- Femoral artery and vein cannulation
Many factors influence the armament of choice in treating iatrogenic pseudoaneurysm. In our anecdotal experience; amongst the important factors are:
- ability to achieve ideal sonographic visualization of the pseudoaneurysm morphology
- assessing neck width and length
- volume of the pseudoaneurysm sac
- depth of the pseudoaneurysm
- local device availability
- patient’s BMI
- coagulation profile (e.g. INR, anticoagulation medications)