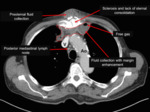
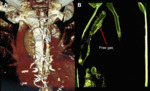
Patients: Data of 134 patients with clinically suspected acute mediastinitis after sternotomy were evaluated in our study.
All patients had received a contrast enhanced chest CT prior to deep revision surgery with collection of microbiological samples .
CT-Protocol: All CT scans were performed on a dual source CT (Somatom Definition Flash,
Siemens Healthcare,
Erlangen,
Germany) according to the standard protocol in our facility.
Dose modulation was used for all scans.
In total 70 ml (400 mg Iodine/ml) of nonionic iodinated contrast medium were applied.
In cases which included an abdominal scan,
90 ml of contrast agent were applied.
Contrast medium flow rate was 2.5ml/s.
The scan was initiated after standardized delay of 90 seconds and performed in cranio-caudal direction capturing the entire thorax.
CT data evaluation: CT data were assessed using commercially available post-processing software.
Qualitative parameters were evaluated using OsiriX software.
Quantitative analysis was performed with "Medical Image Processing,
Analysis and Visualization" (MIPAV,
National Institutes of Health,
Bethesda,
Maryland,
USA) software.
Exclusion criteria for quantitative analysis were mediastinal drainage at the time of CT exam or incompatibility with the software used.
Qualitative parameters: All qualitative image parameters were assessed visually.
22 different CT-parameters were examined,
including mediastinal tissue composition,
sternal condition,
lymph node size and secondary paramediastinal findings.
Quantitative parameters: In every evaluated dataset a VOI (volume of interest) of the anterior mediastinal region was generated.
Employing density-based threshold segmentation,
the volume of fluid,
air and fat in the anterior mediastinal region as well as its total volume was measured.
Furthermore,
we measured maximum lymph node sizes and sternal dehiscence distances.
Clinical data: We recorded evidence of thoracic infection and its location seen during revision surgery as described in the surgical report.
Furthermore,
we noted inflammatory markers values,
which were taken as close as possible prior to the time of revision surgery,
with a maximum time frame of two weeks.
Microbiological data: Microbiological samples were taken during revision surgery and their analysis was performed according to the standards of our clinic.
Type and amount of pathogen as well as sampling point were recorded.
Frequencies of pathogen types were evaluated.
Clinical standard of reference: Patients with evidence of acute retrosternal inflammatory signs in revision surgery or positive retrosternal microbiological samples were classified as mediastinitis positive,
in line with CDC guidelines.4