Introduction
The Charcot foot or Charcot neuropathic osteoarthropathy (CN) has been first described 1868 by Jean-Martin Charcot,
a French pathologist and neurologist,
in patients with tabes dorsalis,
a syphilitic myelopathy  [1].
Nowadays,
the detailed pathomechanisms of this disease remain unclear,
but there is consensus that polyneuropathy is the underlying basic condition of this disease.
In industrialized countries diabetes mellitus is the main cause of neuropathy in the lower limb
[1].
Nowadays,
the detailed pathomechanisms of this disease remain unclear,
but there is consensus that polyneuropathy is the underlying basic condition of this disease.
In industrialized countries diabetes mellitus is the main cause of neuropathy in the lower limb  [2].
The prevalence of CN in a general diabetic population is estimated between 0.1% and 7.5%,
but regarding diabetic patients with apparent peripheral neuropathy,
this prevalence is increasing up to 35%
[2].
The prevalence of CN in a general diabetic population is estimated between 0.1% and 7.5%,
but regarding diabetic patients with apparent peripheral neuropathy,
this prevalence is increasing up to 35%  [3].
The risk of getting a Charcot foot is not related to the type (I or II) of diabetes mellitus
[3].
The risk of getting a Charcot foot is not related to the type (I or II) of diabetes mellitus  [2].Nevertheless,
there are much more causes than diabetes for neuropathy and therefore for CN,
e.g.
alcohol abuse,
heavy metal poisoning,
traumatic injury,
multiple sclerosis,
malnutrition (Vit.
B12 and folic acid deficiency) and many more
[2].Nevertheless,
there are much more causes than diabetes for neuropathy and therefore for CN,
e.g.
alcohol abuse,
heavy metal poisoning,
traumatic injury,
multiple sclerosis,
malnutrition (Vit.
B12 and folic acid deficiency) and many more  [2].
That’s why the often-used term diabetic neuropathic osteoarthropathy (DNOAP) is only partially correct.
The incidence of bilateral involvement of the feet in CN has been reported to be between 9% and 75%
[2].
That’s why the often-used term diabetic neuropathic osteoarthropathy (DNOAP) is only partially correct.
The incidence of bilateral involvement of the feet in CN has been reported to be between 9% and 75%  [2].
The mean age of patients with CN present is in their fifties or sixties and most of them have had diabetes mellitus for at least 10 years
[2].
The mean age of patients with CN present is in their fifties or sixties and most of them have had diabetes mellitus for at least 10 years  [2].
[2].
Course of disease
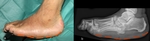
Pedal CN has an active phase (inflammation and fragmentation) and a chronic inactive phase (coalescence and consolidation).
The active phase is characterized by a hot,
red and swollen foot (=inflammation),
often without pain,
due to the polyneuropathy (Fig.
1).
A (minor) trauma is often reported prior to the onset of CN  [3].
The skin temperature on the affected foot is reported to be more than 2°C higher than on the contralateral foot.
But this sign might not always be reliable in clinical practice because of its susceptibility to environmental influences.
Furthermore,
it can only be used reliably in unilateral Charcot foot involvement
[3].
The skin temperature on the affected foot is reported to be more than 2°C higher than on the contralateral foot.
But this sign might not always be reliable in clinical practice because of its susceptibility to environmental influences.
Furthermore,
it can only be used reliably in unilateral Charcot foot involvement  [4].
[4].
In the active phase the bone gets fragile (=fragmentation) due to temporary osteopenia leading to fractures and even to the collapse of the longitudinal arch of the foot,
if loaded unprotected.
This can occur in a very short period of time (Fig.
2).
Differential diagnosis for an active Charcot foot may be infection (especially osteomyelitis),
stress-fracture and physical overload in cases with fat pad atrophy  [5].
[5].
The chronic inactive phase of CN shows no longer a warm,
red foot,
although some soft tissue edema may last.
Prominent osteophytes and palpable loose bodies are the consequence of a substantial joint and bone destruction followed by bone proliferation (coalescence and consolidation).
Due to the biomechanical roll-movement of the foot during the gait sequence,
the foot often presents with a so called “rocker-bottom-deformity”.
This deformity is a result of deformity and subluxation/dislocation of the metatarsal bones in the Lisfranc-Joint: he cuboid bone becomes the most inferior bone in the foot  [3] (Fig.
3).
The resulting changes of the pedal shape can lead to bony prominences (so called pseudoexostoses).Combined with the reduced loss of sensation in polyneuropathy,
and a diminution of the elasticity of the soft tissue (due to alterations in the collagen structure
[3] (Fig.
3).
The resulting changes of the pedal shape can lead to bony prominences (so called pseudoexostoses).Combined with the reduced loss of sensation in polyneuropathy,
and a diminution of the elasticity of the soft tissue (due to alterations in the collagen structure  [6]),
the foot is prone to extensive callus formation,
blisters and ulcerations.
This can lead to infections like cellulitis and osteomyelitis,
which may result in amputation.
[6]),
the foot is prone to extensive callus formation,
blisters and ulcerations.
This can lead to infections like cellulitis and osteomyelitis,
which may result in amputation.
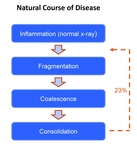
For a long time,
there was the leading opinion,
that once the Charcot foot activity has disappeared,
recurrence of CN activity in the same foot was unlikely.
However,
newer studies show that CN recurrence rates are about 23% with a mean interval of 27 months  [7] (Fig.4).
[7] (Fig.4).
Pathogenesis
The exact pathomechanism for the development of CN is not known yet and is thought to be multifactorial (Fig.
5)  [8].
Both two main theories that have been discussed in the past are based on the presence of neuropathy.
One is focusing on the neurovascular changes in blood supply (neurovascular theory) the other one on repetitive traumatic events due to the lack of sensory feedback (neurotraumatic theory)
[8].
Both two main theories that have been discussed in the past are based on the presence of neuropathy.
One is focusing on the neurovascular changes in blood supply (neurovascular theory) the other one on repetitive traumatic events due to the lack of sensory feedback (neurotraumatic theory)  [9].
One current model of CN origin supports the hypothesis that once the disease is triggered in susceptible individuals e.g.
due to a fractured bone,
an unregulated inflammatory process is started,
which leads to an abnormally intense stimulation and maturation of osteoclasts
[9].
One current model of CN origin supports the hypothesis that once the disease is triggered in susceptible individuals e.g.
due to a fractured bone,
an unregulated inflammatory process is started,
which leads to an abnormally intense stimulation and maturation of osteoclasts  [10].
Increased osteoclastic activity is responsible for the unrestrained bone turnover which causes more bone damage.
This bidirectional relationship between inflammation and repetitive traumata has been shown to be essential.
[10].
Increased osteoclastic activity is responsible for the unrestrained bone turnover which causes more bone damage.
This bidirectional relationship between inflammation and repetitive traumata has been shown to be essential.
Nevertheless,
it still remains inexplicable why CN is a quite rare condition,
mostly unilateral and usually a self-limiting process  [6].
[6].
Treatment
Current state-of-the art treatment is the off-loading of the affected foot.
The goal is to maintain a plantigrade foot with minimal deformity to avoid increased pressure points  [1].
There are several off-loading techniques,
which depend on the availability of the infrastructure and the training of the treating staff.
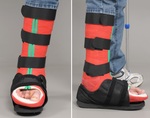
One commonly used method is the treatment of patients with custom-made removable total contact casts (rTCC) until the activity signs of the Charcot foot are significantly reduced or gone (Fig.
6).
This might take up to 18 months,
therefore compliance is a major issue.
So far,
there is no clinical value,
which would allow to determine the completed healing process.
Monitoring of the disease activity using MRI (e.g.
in 3-month-intervalls),
gives the opportunity to decide when the time has come to end the off-loading treatment.
After the off-loading treatment,
the patients should be equipped with orthopedic shoes,
which are professionally adapted to their feet [7].
[1].
There are several off-loading techniques,
which depend on the availability of the infrastructure and the training of the treating staff.
One commonly used method is the treatment of patients with custom-made removable total contact casts (rTCC) until the activity signs of the Charcot foot are significantly reduced or gone (Fig.
6).
This might take up to 18 months,
therefore compliance is a major issue.
So far,
there is no clinical value,
which would allow to determine the completed healing process.
Monitoring of the disease activity using MRI (e.g.
in 3-month-intervalls),
gives the opportunity to decide when the time has come to end the off-loading treatment.
After the off-loading treatment,
the patients should be equipped with orthopedic shoes,
which are professionally adapted to their feet [7].
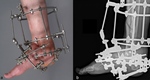
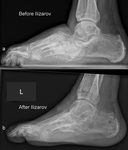
The stabilization with the  Ilizarov external fixator frame is considered as an alternative treatment option for the off-loading
Ilizarov external fixator frame is considered as an alternative treatment option for the off-loading  [11] (Fig.
7 and Fig.
8).
It is particularly used in feet with severe deformity or after the removal of osteomyelitic bone fragments
[11] (Fig.
7 and Fig.
8).
It is particularly used in feet with severe deformity or after the removal of osteomyelitic bone fragments  [12].
Surgical treatment options like reconstructive surgery in a stable,
nonplantigrade foot or resection of bony prominences in a stable plantigrade foot are available.
Major amputations need to be done in case of severe bone destruction including osteomyelitis or failed previous surgery
[12].
Surgical treatment options like reconstructive surgery in a stable,
nonplantigrade foot or resection of bony prominences in a stable plantigrade foot are available.
Major amputations need to be done in case of severe bone destruction including osteomyelitis or failed previous surgery  [9].
The main goal of all reconstructive procedures is to form a foot,
which can be adequately equipped with an orthopedic shoe or an orthotic device.
[9].
The main goal of all reconstructive procedures is to form a foot,
which can be adequately equipped with an orthopedic shoe or an orthotic device.


![Fig. 5: Multifactorial pathogenesis of Charcot foot (adapted from [8]) References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779520?maxheight=150&maxwidth=150)