Classifications
The Charcot neuropathic osteoarthropathy has been classified in various systems using anatomical landmarks on x-rays and clinical symptoms.
Sanders and Frykberg Classification
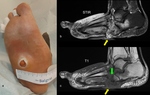
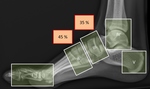
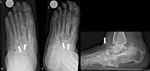
Sanders and Frykberg identified 5 zones of disease distribution according to their anatomical location,
as demonstrated in Fig.
9.
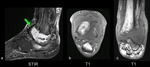
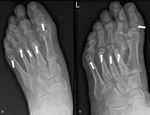
Zone I: metatarsophalangeal and interphalangeal joints (Fig.
10),
Zone II: tarsometatarsal joints (Fig.
11),
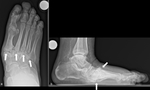
Zone III: tarsal joints (Fig.
12),
Zone IV: ankle and subtalar joints (Fig.
13),
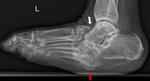
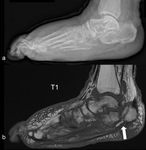
Zone V: calcaneus (Fig.
14).
Most commonly involved are zone II (tarsometatarsal articulations) in about 45% and zone III (cuneonavicular,
talonavicular and calcaneocuboid articulations) in about 35 % of CN cases  [2].
[2].
Brodsky Classification
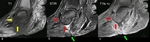
The Brodsky classification is another anatomy-based classification.
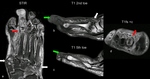
Brodsky differentiated 4 anatomical areas mostly affected in CN as shown in Fig.
15.
Type 1 (metatarsocuneiform and naviculocuneiform joints) is considered the most common (60%) and is often associated with symptomatic exostosis.
Type 2 (subtalar,
talonavicular or calcaneocuboid joints) is the second most common type (30-35%),
symptomatic exostosis is less often observed here.
Type 3 is divided into two subtypes: Type 3a (ankle) is seen in about 9% of CN cases,
Type 3b (Calcaneus) is only seen in about 2% with the clinical manifestation being a pathologic fracture of the calcaneus tuberosity  [13].
[13].
Eichenholtz Classification
Traditionally,
the natural history of CN has been divided into three stages according to the Eichenholtz classification.
However,
the classification does not include the whole range of the disease,
therefore the addition of the clinically important stage 0 has been proposed in the literature,
the so called “pre-stage 1” or “Charcot in situ” (Table 1)  [14,
15].
[14,
15].
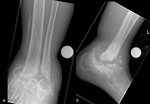
Fig.
16 – 18 show the radiographic appearance of left foot in the same patient through the stages 1,
2 and 3 according to the Eichenholtz classification.
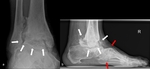
Stage 1 (Fig.
16),
Stage 2 (Fig.
17),
Stage 3 (Fig 18).
Because of the missing radiographic findings,
patients in Stage 0 are often misdiagnosed with other diseases,
like gout,
cellulitis or deep vein thrombosis.
MRI allows an early diagnosis,
which is crucial,
because early off-loading in patients with CN Stage 0 can prevent the progression into higher debilitating stages.
Limitations of Eichenholtz Classification.
Need for MRI-Classification?
Since medical imaging technologies rapidly advance,
Chantelau and Gruetzner  [15] proposed to replace the classic Eichenholtz classification with a newer one,
including especially MRI features.
[15] proposed to replace the classic Eichenholtz classification with a newer one,
including especially MRI features.
Limitations of the Eichenholtz classifications are:
-
Eichenholtz-Stages do not correlate well with the clinical symptoms of CN
-
Eichenholtz-Stages do not cover the whole spectrum of CN and adding a Stage 0 as proposed by Mautone and Naidoo  [14] does not differentiate between an early active stage and a properly healed inactive stage
[14] does not differentiate between an early active stage and a properly healed inactive stage
-
Plain radiographs are not capable of diagnosing acute pathologies like cortical foot fractures,
which often are only visible on CT or MRI scans.
The bone marrow edema pattern can exclusively be detected on MRI.
Chantelau and Gruetzner proposed a new classification which is divided in two stages and two grades as seen in Table 2  [15].
[15].
This MRI-classification offers a possibility to predict the outcome.
Grade 1 stages usually take longer to heal than grade 0.
Therefore,
a longer duration of off-loading is needed.
Once healed,
grade 1 cases show a higher degree of deformity and foot function is much more reduced  [16].
[16].
Because of the significance of MRI-Imaging in the management of Charcot neuropathic osteoarthropathy classification and grading systems are necessary and should be further developed for future use in daily routine.
Role of conventional radiographs
X-Rays of the Charcot foot are traditionally the standard imaging technique to establish the diagnosis,
to stage and to monitor the disease.
But the main value of plain radiographs is to assess the position of the bones to each other in general,
and in particular under load (Fig.
19).
Loading techniques in advanced imaging modalities like CT and MRI are still not sufficient to replace radiographs,
because of their limited availability and the difficulty concerning standardization.
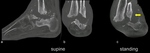
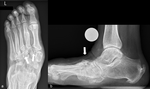
Typical measurements that help to determine the severity of deformation in Charcot foot,
especially in follow up studies are: 1) Meary’s angle or lateral talo-first metatarsal angle (angle between the line originating from the center of the body of the talus,
bisecting the talar neck and head,
and the line through the longitudinal axis of 1st metatarsal),
which normally should be around 0°,
2) cuboid height (perpendicular distance from the plantar aspect of the cuboid to a line drawn from the plantar surface of the calcaneal tuberosity to the plantar aspect of the 5th metatarsal head) and 3) calcaneal pitch (angle between a line extending from the plantar aspect of the calcaneus to the plantar surface of the 5th metatarsal head and the line extending from the most plantar portion of the calcaneal tuberosity to the most plantar portion of the anterior calcaneus)  [17] (Fig.
20).
[17] (Fig.
20).
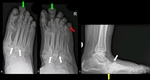
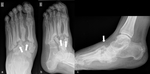
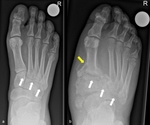
Dorsoplantar (dp) radiographs can reliably show the (sub)luxation in the Lisfranc’s joint,
especially the medial aspect of the joint (Fig 21).
Oblique conventional radiographs are superior to dp-radiographs in visualizing the lateral aspect of the Lisfranc’s joint (3rd to 5th tarsometatarsal joint) (Fig.
18).
Dorsoplantar radiographs in follow-up studies show the increase in forefoot abduction relative to the hindfoot over time,
the so-called hindfoot-forefoot angle (Fig.
21).
As Hastings at al.
 [17] showed there are 3 steps to get this measurement done: 1) quantify the talocalcaneal angle,
which is the angle formed between the line that bisects the talar neck and head and the line parallel to the lateral cortex of the calcaneus,
2) bisect the talocalcaneal angle,
3) get the hindfoot-forefoot angle,
which is the angle between a line parallel to the bisector of the talocalcaneal angle and a line through the longitudinal axis of the 2nd metatarsal (Fig.
21).
[17] showed there are 3 steps to get this measurement done: 1) quantify the talocalcaneal angle,
which is the angle formed between the line that bisects the talar neck and head and the line parallel to the lateral cortex of the calcaneus,
2) bisect the talocalcaneal angle,
3) get the hindfoot-forefoot angle,
which is the angle between a line parallel to the bisector of the talocalcaneal angle and a line through the longitudinal axis of the 2nd metatarsal (Fig.
21).
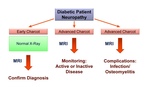
Role of Magnetic Resonance Imaging
MRI is the standard procedure in order to establish an early diagnosis in CN (when x-rays still appear normal).
MRI also allows to determine the course of the healing process and the success of the off-loading treatment.
Another very significant role of MRI is its ability to further evaluate complications of a Charcot foot,
in particular soft tissue infections and osteomyelitis (Fig.
22).
As already mentioned above,
there is no established MRI classification system.
First attempts have been demonstrated by Chantelau and Gruetzner  [15].
The main benefit of MRI is to describe the course of the disease,
since it is the only reliable qualitative method to image changes inside of the bones,
e.g bone marrow edema.
Most of the other options,
like nuclear medicine techniques,
are less sensitive.
[15].
The main benefit of MRI is to describe the course of the disease,
since it is the only reliable qualitative method to image changes inside of the bones,
e.g bone marrow edema.
Most of the other options,
like nuclear medicine techniques,
are less sensitive.
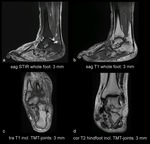
MRI-Protocol
In order to assess all important information about the Charcot foot by MRI,
the protocol doesn’t need to be extensive.
Essential is the use of large Field of View (FoV) to get a good impression of the entire foot.
It is necessary to use a fluid sensitive sequence,
like STIR (short tau inversion recovery) for assessing edema in the bone marrow and soft tissue.
A classic T1 TSE (turbo spin-echo) sequence is irreplaceable to demonstrate the anatomy and the fat signal of the bone marrow.
T2 weighted sequences can demonstrate the presence of subchondral cysts and help to identify fluid collections and sinus tracts.
Especially sagittal large FoV images give a quick superior impression about the overall situation.
Axial images are useful to assess the Lisfranc’s joint disease.
An MRI protocol proposal for Charcot foot evaluation is demonstrated in Fig 23.
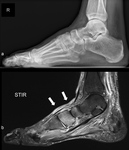
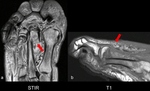
MRI-benefit 1: Diagnosis of early stage Charcot neuropathic osteoarthropathy
Establishing an early diagnosis and therefore an early off-loading treatment is crucial for the prognosis and outcome of an acute Charcot foot (Fig.
24).
MRI early stage Charcot neuropathic osteoarthropathy
MRI is the best imaging modality to confirm diagnosis of suspected early active Charcot disease. Early signs of a Charcot foot in MRI are (subchondral) bone marrow edema,
soft tissue edema,
involvement of the Lisfranc’s joint,
joint effusion,
and microfractures (subchondral).
It is useful to directly assess the Lisfranc ligament (which runs from the medial cuneiform to the second metatarsal bone) which will be affected prior to Lisfranc’s joint subluxation.
No cortical fractures and no gross deformity is to be seen (Fig.
25)  [19].
[19].
MRI-benefit Nr.
2: Monitoring of disease activity
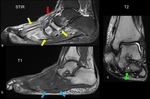
MRI of late stage Charcot neuropathic osteoarthropathy
MRI of late stage disease of Charcot neuropathic osteoarthropathy shows less or complete regression of bone marrow edema.
Joint destruction and joint dislocations are present.
Especially the involvement of Lisfranc’s joint leads to a typically superior and lateral dislocation of the metatarsals leading to a complete collapse of the longitudinal arch (Fig.
26).
The talus head is tilted towards the sole of the foot (Fig.
27),
the navicular bone dislocates into a medial position.
The cuboid bone becomes the most inferior and therefore most prominent part of the foot on the plantar side,
leading to an increased weight-bearing stress in the cuboid bone.
Beneath the cuboid bone callus and ulcer formation is common.
Prominent well-marginated subchondral cysts are a typical feature of the Charcot foot (Fig.
28b).
Gross cortical fractures are present (Fig.
29),
especially calcaneus fractures are common.
Bone proliferation / sclerosis,
debris and intraarticular bodies can occur  [2,
19].
[2,
19].
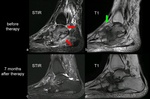
Monitoring of disease activity with MRI
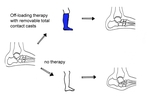
MRI is the best modality to monitor the disease activity of CN.
As long as a significant amount of bone marrow edema is seen on MRI,
consequent off-loading therapy with removable total contact casts (rTCC) has to be continued  [20].
After a significant decrease or complete disappearance of bone marrow edema,
the next step in the therapy can be done,
with installation of the orthopedic footwear (Fig.
30).
[20].
After a significant decrease or complete disappearance of bone marrow edema,
the next step in the therapy can be done,
with installation of the orthopedic footwear (Fig.
30).
MRI-benefit Nr.
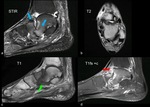
3: Imaging of Complications: Infection/Osteomyelitis
MRI has been shown to have a high diagnostic accuracy in diagnosing osteomyelitis of the foot,
with a high sensitivity (77-100%) and high specificity (80-100%) rate  [14].
MRI has in particular a very high negative predictive value (98%): if there are no signs of osteomyelitis on MRI you can practically exclude osteomyelitis
[14].
MRI has in particular a very high negative predictive value (98%): if there are no signs of osteomyelitis on MRI you can practically exclude osteomyelitis  [21].
Besides making the diagnosis,
MRI is capable of defining the extent of the infection.
Although in the presence of a Charcot foot discriminating active CN from acute osteomyelitis remains challenging.
Both entities have similar image characteristics like bone marrow edema,
soft tissue edema,
joint effusions,
fluid collections,
and contrast-enhancement in bone marrow and soft tissues.
Even the degree of signal drop in T1 sequences might be quite similar in both conditions.
But making the correct diagnosis is crucial to prevent a potential disaster because the therapeutic approach is very different.
Active CN needs off-loading therapy while osteomyelitis requires antibiotic therapy and possibly surgical debridement
[21].
Besides making the diagnosis,
MRI is capable of defining the extent of the infection.
Although in the presence of a Charcot foot discriminating active CN from acute osteomyelitis remains challenging.
Both entities have similar image characteristics like bone marrow edema,
soft tissue edema,
joint effusions,
fluid collections,
and contrast-enhancement in bone marrow and soft tissues.
Even the degree of signal drop in T1 sequences might be quite similar in both conditions.
But making the correct diagnosis is crucial to prevent a potential disaster because the therapeutic approach is very different.
Active CN needs off-loading therapy while osteomyelitis requires antibiotic therapy and possibly surgical debridement  [3] or amputation.
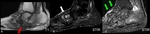
There are some imaging features that help the radiologist to find the correct diagnosis - as listed in table 3 (Fig.
31,
Fig.
32).
[3] or amputation.
There are some imaging features that help the radiologist to find the correct diagnosis - as listed in table 3 (Fig.
31,
Fig.
32).
Advanced MR-Imaging techniques
The usefulness of MR-Imaging in differentiation between active phase of neuropathic arthropathy and osteomyelitis has been shown in the past  [23].
Unfortunately,
there is still some overlap in the imaging appearance between both entities.
Due to technical progress there are more and more advanced MR-Imaging techniques available with the possibility to add functional and quantitative information
[23].
Unfortunately,
there is still some overlap in the imaging appearance between both entities.
Due to technical progress there are more and more advanced MR-Imaging techniques available with the possibility to add functional and quantitative information  [24].
[24].
Diffusion-weighted imaging may contribute in the detection and extension of osteomyelitis: pure edema does not show diffusion restriction,
whereas the presence of pus and inflammatory cells in infection leads to restricted diffusion with lower ADC-values than in pure edema  [25].
Dynamic Contrast Enhancement (DCE) Perfusion may help in the discrimination between viable tissue and necrosis.
Furthermore,
the enhancement pattern in DCE-Perfusion seems to be different between osteomyelitis and osteoarthropathic changes,
increasing the potential of differencing lesions with bone marrow edema
[25].
Dynamic Contrast Enhancement (DCE) Perfusion may help in the discrimination between viable tissue and necrosis.
Furthermore,
the enhancement pattern in DCE-Perfusion seems to be different between osteomyelitis and osteoarthropathic changes,
increasing the potential of differencing lesions with bone marrow edema  [24].
Diffusion Tensor Imaging (DTI) may have the potential in evaluating peripheral nerve disease
[24].
Diffusion Tensor Imaging (DTI) may have the potential in evaluating peripheral nerve disease  [26],
although it is a very challenging technique,
especially in the foot.
MR Angiography (MRA) allows to non-invasively evaluate peripheral vascular impairment,
which is frequently observed in diabetic patients.
This can be done by contrast-enhanced 3D-MRA and even without contrast using nonenhanced MRA (gated 3D fast spin-echo MRA based on the acquisition of images during the diastolic and systolic phases of the cardiac cycle)
[26],
although it is a very challenging technique,
especially in the foot.
MR Angiography (MRA) allows to non-invasively evaluate peripheral vascular impairment,
which is frequently observed in diabetic patients.
This can be done by contrast-enhanced 3D-MRA and even without contrast using nonenhanced MRA (gated 3D fast spin-echo MRA based on the acquisition of images during the diastolic and systolic phases of the cardiac cycle)  [27].
[27].
MRI-Limitations
There are some strict contraindications for MRI studies in general,
e.g.
implanted electronic devices like heart pacemakers (in particular older types),
insulin pumps,
neurostimulators and implanted hearing aids but also some types of intracranial metal clips and metallic foreign bodies close to the eye.
Therefore,
not all patients may undergo an MRI examination.
Claustrophobia might be a limitation as well,
but fortunately examinations of the foot are usually well tolerated,
because the main part of the body and especially the head of the patient are positioned outside the scanner during the examination.
Alternatively in those patients,
CT and nuclear medicine imaging can be performed.
In comparison to other radiological techniques the main disadvantage of the MRI is,
that the images cannot be obtained under load.
Therefore,
there is still a need for conventional radiographs.
In some cases,
the MRI might not be able to distinguish severe charcot-activity from osteomyelitis with the need for additional biopsy performance or nuclear imaging methods.
Special Case 1
A 58-year-old male presented in the emergency department with a sudden shortening of the left leg.
The history of the patient revealed a long-standing insulin-dependent diabetes mellitus (IDDM) type I for 26 years with polyneuropathy (Fig.
33,
34,
35).
Special Case 2
66-year-old male patient with known Charcot foot and insulin-dependent diabetes mellitus type II present with a new ulcer formation at the tip of the second and at the fifth toe (Fig.
36,
37).

![Table 1: Modified Eichenholtz classification from Mautone and Naidoo 2015 [14] References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779531?maxheight=150&maxwidth=150)
![Table 2: Proposed MRI-classification for CN modified from Chantelau and Grützner 2014 [15] References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779536?maxheight=150&maxwidth=150)
![Fig. 20: Lateral weight bearing radiographs showing the typical course of Charcot foot disease over time (a: Baseline, b: 10 month later). Note the continuous increase of Meary’s angle or lateral talo-first metatarsal angle (yellow angle), the diminishment of cuboid height, which is becoming negative (blue distance) and the decrease of the calcaneal pitch (red angle) [18] References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779538?maxheight=150&maxwidth=150)
![Fig. 21: Radiograph in dorsoplantar projection showing the changes in foot morphology in a typical Charcot foot patient over time (a: baseline, b: 10 months later). Note the increase in forefoot abduction relative to the hindfoot (hindfoot-forefoot angle) which is the angle between the longitudinal axis of the 2nd metatarsal bone (red arrow) and the bisection (yellow dotted line) of the angle which is formed by the following two lines: one through the talar neck and head and the other parallel to the lateral cortex of the calcaneus (green arrows) [18] References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779539?maxheight=150&maxwidth=150)
![Table 3: MRI features for differentiating an active Charcot foot from osteomyelitis. Information collected from Ahmadi et al 2006 [22] , Donovan and Schweitzer 2010 [23], Ergen et al. 2013 [2], Martín Noguerol et al. 2017 [24], Mautone and Naidoo 2015 [14], Schoots et al. 2010 [3] References: Balgrist University Hospital](https://epos.myesr.org/posterimage/esr/essr2018/145916/media/779549?maxheight=150&maxwidth=150)