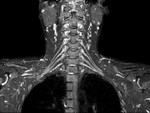
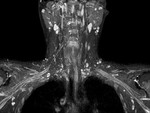
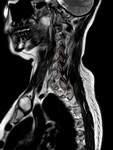
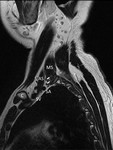
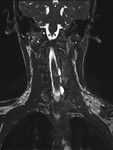
Our centre uses the Philips Ingenia 1.5 Tesla MRI scanner to image the brachial plexus.
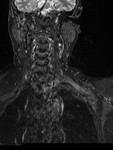
Bilateral brachial plexuses are imaged to allow comparison and better detection of abnormalities.
MRI protocol:
|
Coronal 3D STIR (include both shoulders) FOV – 350mm TR,TE – 3000ms,
194ms,
ST/GAP – 1.4 mm,
0.7mm Matrix – 252/249
|
|
| Coronal MIP FOV- 350mm TR,TE- 3000ms,
194ms,
ST/GAP -2mm,
2mm Matrix 252/249 |
| Coronal T1 (include both shoulders) FOV – 320mm,
TR,TE – 647ms,
6ms,
ST/GAP – 3mm,
0.3mm Matrix – 415/341 |
| Axial STIR (C5 to inferior axilla,
single-side) FOV – 200mm TR,TE – 3222ms,
30ms ST/GAP – 4mm,
0mm Matrix – 348/242 |
| Axial T1 (single-side) FOV – 200mm TR,TE – 645ms,16ms ST/GAP – 4mm,
0mm Matrix – 460/368 |
| Sagittal T2 (C-spine to midpoint of arm) FOV – 240mm TR,TE – 4578ms,90ms ST/GAP – 4mm,
1 mm Matrix –388/298 |
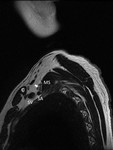
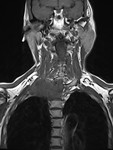
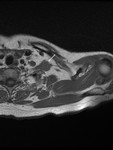
Normal Brachial Plexus Anatomy
The brachial plexus (Fig.
1) is predominantly formed by the ventral rami of the spinal nerves of C5 through T1 (with variable contribution from C4) which exit the neural exit foramina and form the roots (Fig.
4).
The roots then combine to form the three trunks: superior,
middle,
and inferior .
The superior trunk is formed by roots of C5 and C6 ,
the inferior trunk is formed by roots of C8 and T1 and the middle trunk is formed solely from the C7 root (Fig.
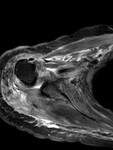
2) The roots and trunk course between in the anterior and middle scalene muscles which form the interscalene triangle bounded by the first rib inferiorly (Fig.
5).
The interscalene triangle also contains the subclavian artery and its branches.
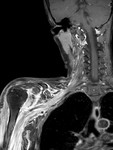
Laterally the trunks give rise to three anterior and three posterior divisions which course between the clavicle anteriorly and the first rib posteriorly,
otherwise known as the costoclavicular region (Fig.
6)
Three cords are formed by means of a combination of anterior or posterior divisions of the trunks.
The individual cords are named according to their relationship to the adjacent subclavian artery: lateral,
posterior,
and medial (Fig.
7).
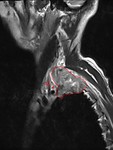
The cords run inferior to the clavicle and divide into individual branches providing motor and sensory innervation to the upper extremity (Fig.
3).
The various divisions of the brachial plexus appear as linear structures with low signal intensity on MR images obtained with all sequences. The brachial plexus can be identified on images obtained in both the axial and coronal planes,
but images obtained in the sagittal plane are often the most helpful in demonstrating divisions and cords adjacent to the subclavian artery [1].
Pancoast tumour
Pancoast tumors are non–small cell carcinomas that originate in the superior sulcus and that often cause signs and symptoms known collectively as Pancoast syndrome.
Manifestations of this syndrome include pain in the shoulder girdle and arm,
as well as Horner syndrome,
which is characterized by ipsilateral anhidrosis of the face,
miosis,
and ptosis [2].
The superior sulcus is more clearly divided by the scalene muscles into anterior,
middle (interscalene),
and posterior compartments [3].
Classic Pancoast tumors associated with Pancoast syndrome usually invade the posterior compartment of the superior sulcus,
due to involvement of the stellate ganglion.
The tumour tends to involve the first,
second and third ribs posteriorly and the vertebral bodies medially (Fig.
8).
Pancoast tumours involving the brachial plexus tend to be sited in a lateral location and involving the interscalene triangle (middle compartment) [3].
Because the brachial plexus is surrounded by a connective-tissue sheath,
a tumor may indent the brachial plexus and displace the nerve roots or trunks superiorly without actually invading them.
Sensory dysfunction may occur because of extrinsic nerve compression,
whereas a loss of motor function is more likely to be indicative of frank invasion of the nerve [3].
MRI evaluation plays an in evaluation of brachial plexus involvement and to avoid overestimating the local extension of a tumor,
it is important to correlate the imaging findings with the patient’s symptoms.
The earliest sign of brachial plexus involvement is the invasion of the interscalene fat pad by tumour.
The fat pad lies between the anterior and middle scalene muscles above the lung apex (Fig.9), and loss of the normal fat signal would preclude surgical excision [4].
Neurogenic Tumour
Primary neurogenic tumors of the brachial plexus can be benign or malignant and include neurofibroma,
plexiform neurofibroma,
schwannoma,
and malignant peripheral nerve sheath tumor.
Neurofibromas are the most common neural tumor to involve the brachial plexus; one-third of these lesions occur in patients with neurofibromatosis type 1,
whereas two-thirds of cases are sporadic [5].
The syndromic type is characteristically multiple and plexiform in appearance,
with diffuse involvement of the brachial plexus
The imaging features of solitary neurofibromas overlap those of schwannomas,
and often they are indistinguishable.
At MR imaging,
both lesions are isointense to muscle on T1-weighted images (Fig.
10) and hyperintense on T2-weighted images and may have central areas with low signal intensity (Fig.
11),
the so-called target sign.
Neurofibromas and schwannomas enhance intensely after administration of gadolinium-based contrast material [6].
Malignant peripheral nerve sheath tumors develop in 3%–13% of patients with neurofibromatosis [7,8].
These lesions occur most commonly in patients with neurofibromatosis type 1,
especially after radiation therapy [9].
Findings that favor a malignant neural tumor include large size,
irregular margins,
and heterogeneity [6].
Metastatic Tumours
Breast carcinoma is the most common source of metastatic disease causing brachial plexopathy; other metastatic sources include lung carcinoma and head and neck cancers [10].
Metastatic lymphadenopathy may surround the neurovascular bundle,
resulting in vascular or neural compromise [6].
Metastatic disease from all causes typically appears as masses with low signal intensity on T1- weighted images and increased signal intensity (greater than that of muscle) on T2-weighted images (Fig.12,
13).
Radiation-induced brachial plexopathy
Radiation therapy to the neck and axilla may result in brachial plexopathy either due to radiation neuritis or due to nerve compression by surrounding fibrous connective tissue.
The median interval between irradiation and occurrence of brachial neuropathy has been reported to be one to four years,
although neuropathy has been reported even 21 years after radiation therapy [11].
Clinical symptoms and electromyography cannot always differentiate a recurrence from brachial plexus injury,
MRI plays an important role in determining the diagnosis.
Imaging features suggestive of radiation-induced plexopathy (Fig.13) include diffuse,
uniform,
symmetric swelling,
and T2 hyperintensity of the plexus within the radiation field [12].
Mild contrast enhancement may also be present,
sometimes making differentiation from tumor difficult.
Nonuniform,
asymmetric,
and focal enlargement,
and the presence of a mass with postcontrast enhancement,
usually indicates tumor recurrence.
Radiation fibrosis often shows low SI on both T1W and T2W images [12].
T2 fat-suppressed and STIR sequences help to differentiate radiation fibrosis from tumor [6].
Traumatic brachial plexus injuries
There are two distinctive populations affected by traumatic brachial plexopathy.
The first is neonates who have sustained a traction injury due to shoulder dystocia during vaginal delivery.
The second population affected by traumatic brachial plexopathy is young men in the second and third decades.
These patients sustain blunt force injury after a fall from a height or a motorcycle or motor vehicle crash and penetrating injury from a gunshot [13].
It is important to differentiate between preganglionic and postganglionic injuries,
as the prognosis is better in postganglionic injuries,
where surgical repair in the form of nerve grafting is possible. MRI of preganglionic injuries can show nerve root avulsions,
with or without pseudomeningoceles [14].
Pseudomeningoceles are cerebrospinal fluid collections due to a dural tear (Fig.
15,
16). The presence of a pseudomeningocele is highly indicative (seen in 80% of avulsions) for a preganglionic lesion,
but is not pathognomonic [15].
Signal abnormalities are observed in the spinal cord in approximately 20% of patients with preganglionic injuries,
such as edema,
haemorrhage and myelomalacia.
Enhancement of intradural nerve roots and root stumps suggests functional impairment of nerve roots despite morphologic continuity.
Features of denervation/atrophy in the paraspinal muscles is an accurate indirect sign of root avulsion injury [13].
MRI of postganglionic injuries show thickened nerves with low signal intensity on T1-weighted images and increased signal intensity on T2-weighted images (Fig.
17). Nerve contiguity can be assessed; there may be discontinuity with distal nerve contraction [14].
Increased T2 signal and enhancement of intact nerves suggests damage or impairment despite contiguity.
Direct compression of the brachial plexus by a haematoma,
fracture fragment or callus formation may also cause brachial plexopathy [14].