The pre-existing bone disease may persist after renal transplantation and is characterized by findings of secondary hyperparathyroidism,
namely bone reabsorption,
periosteal reaction and brown tumours (osteitis fibrosa cystica - figure 1).[1]
In 95% of patients,
skeletal features of hyperparathyroidismare foremost identified in the hands.[5] Bone reabsorption can be categorized by location in subperiosteal,
intracortical,
trabecular,
endosteal,
subchondral and subligamentous.[1],[5]
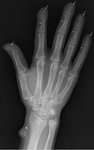
Subperiosteal reabsorption presents as a lacelike irregularity of the cortical margins.[1],[5] It has a virtually pathognomonic predilection for the radial aspects of the middle phalanges of the middle and index fingers in the hands and for the distal phalangeal tuffs,
a finding consistent with acro-osteolysis (figure 2).[5] These irregularities of the cortical surface can additionally produce a false appearance of periosteal reaction (pseudoperiostitis).[5]
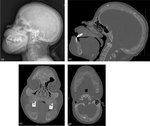
Bone reabsorption in the skull has a salt and pepper appearance,
characterized by innumerable punctate lucent and radiodense foci (figure 3).[1],[5] Subchondral resorption can involve any joint,
leading to a widened and irregular appearance.
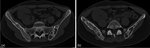
When the sacroiliac joint is affected,
subchondral resorption is more prominent at the iliac side and may even simulate an inflammatory or infectious arthritis (figure 4).[5]
Brown tumours are typically solitary well-defined lytic lesions,
that may cause endosteal scalloping and osseous expansion,
most frequently found in facial bones,
ribs,
pelvis and femur; yet brown tumours may also present as multifocal lesions and associated with a large soft-tissue component (figures 5 and 6).[1],[5]
The aforementioned post-operative disturbances of mineral bone metabolism contribute to exacerbate osteopenia and osteoporosis,
shown as a diffuse decrease in bone radiodensity,
affecting more frequently the axial skeleton (figure 7) and increasing the risk of pathological fractures (figure 8).[1],[5] ROD also encompasses osteosclerosis,
which may be the only manifestation (figure 9).[1],[5] Increased radiodensity is usually uniform and has also predilection for the axial skeleton.
The spine can display a pattern known as rugger jersey spine: a striped appearance secondary to alternating bands of sclerosis along the endplates and areas of central lucency (figure 10).[5]
It may even trigger osteomalacia,
which is characterized by Looser zones: transverse areas of lucency frequently bilateral and symmetric,
affecting both cortical and trabecular bone.
Looser zones,
also known as pseudofractures,
can be found at sites of stress,
namely the medial aspect of the femoral neck or the pubic rami,
but also at unusual sites,
such as the lateral aspect of the femoral shaft,
the ischium,
the iliac wing and the lateral scapula.[5]
Soft-tissue manifestations are more pronounced after long-term haemodialysis and renal transplantation and include multifocal crystal deposits of calcium and/or urate.
Vascular calcifications involve intimal and medial layers without prominent luminal implication and are initially found in dorsalis pedis artery.
Periarticular calcifications are symmetric and can extend into the adjacent joint,
causing chondrocalcinosis or even bursitis and synovitis (figure 11).[5] Visceral calcifications usually are not radiographically evident and can be life-threatening in heart,
lungs,
stomach and kidneys.[5]
Amyloid arthropathy is characterized by soft-tissue mineralization (β2-microglobulin deposits),
extrinsic large bone erosions and joint space narrowing,
which can ultimately lead to chronic destructive arthropathy.[1],[5]
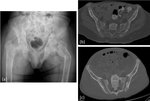
Osteonecrosis (manly femoral head necrosis) may be detected in conventional X-ray,
but MR is the most sensitive modality in the diagnosis (figure 12).[1],[3]
Musculoskeletal infections (osteomyelitis or septic arthritis) can also occur as a complication of renal transplantation.[1]