Purpose
·To evaluate role of 2-deoxy-2-(18Fluorine) fluoro-D-glucose fluorodeoxyglucose (FDG) PET-CT in staging lung nodules<2cm (presumed T1a cancers).
·To assess the role of FDG PET-CT in discerning benign from malignant nodules and its prognostic impact on survival.
Materials and Methods
Retrospective database analysis of adult patients with pulmonary nodules<2cm undergoing FDG PET-CT from January 2011 to December 2012 at a tertiary care Institution in the UK.
Data collected included demographics,
nodule morphology,
size,
maximum standardised uptake value (SUVmax),
and test characteristics of FDG PET-CT.
Patients had confirmed histological diagnosis of malignancy or progression on serial imaging as a surrogate.
Median (range) follow-up was 25 (18-28) months.
Results
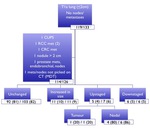
Out of a total of 133 nodules,
126 nodules were found to be appropriate for inclusion.
Follow up data for a median period of up to 25 months was also collected.
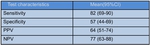
PET-CT had a sensitivity of 82%,
specificity of 57%,
PPV of 64% and NPV of 77% in this cohort.
Diagnosis was confirmed by a combination of imaging features,
interim progression on follow-up imaging and/or histopathology in 60/126 cases.
The additive impact of PET-CT on staging in this cohort was limited and it did not...
Conclusions
FDG PET-CT did not significantly alter staging in patients with lung nodules<2 cm in our retrospective cohort.
PET-CT had a sensitivity of 82%,
but the specificity was low.
Further prospective multi-centre studies are required to validate these findings.