The International Commission for Radiological Protection (ICRP),
the European Commission (EC),
many professional organisations and regulatory authorities,
realising the above limitations with DRLs,
have started several initiatives to overcome the limitations and to harmonise the further development of DRLs based on a broad cooperation among all stakeholders.
Among these,
the EuroSafe Imaging campaign and the European Society of Radiology (ESR) have contributed to several activities that will be presented together with the work of all others.
The PiDRL project of the EC,
the joint work of a consortium,
has produced guidelines for paediatric DRLs that have recently been submitted to the EC; PiDRL has established close contacts to the ICRP that is working on a new DLR recommendation now under consultation.
1.
DRLs: definitions and methodology
Both the new ICRP consultation draft and the PiDRL project pay attention to harmonisation.
DRLs can be set at local,
national and regional (e.g.
European) levels under the control of defined authorised bodies; they correspond to the 75th percentile of the dose distribution within a representative sample of the patient population.
The 50th percentile - also called achievable dose – can be used for further optimisation but is not a DRL.
A DRL value is considered to be exceeded when the median value of a DRL quantity for a representative patient sample within a healthcare facility is higher than the local,
national,
or regional DRL.
DRL quantities should assess the amount of ionising radiation applied to perform a medical imaging task and should be easily measured or determined (ICRP).
They are specific to the modality used:
- entrance surface air kerma (Ka,e) and kerma-area product (PK,A,
formerly called dose-area product) for radiography (exceptional alternative for mammography: mean glandular dose),,
- kerma-area product (PK,A),
cumulative air-kerma at the patient entrance reference point (Ka,r),
fluoroscopy time and the number of radiographic images for fluoroscopy/intervention,
- volume-weighted computed tomography dose index (CTDIvol) for each sequence and the cumulative dose-length product (DLP) for the entire examination for CT.
- administered activity per kg body weight of a specific radionuclide for nuclear medicine.
Rules for local and national surveys: except for tests of equipment performance,
surveys should be based on patient data,
not phantoms.
National surveys should cover 20-30 facilities (30-50% of facilities in small countries).
Data should represent ³10-20 patients per exami-nation/indication for radiography and CT,
³20-30 for fluoroscopy and ³50 for mammography.
Furthermore,
when automatic data acquisition of DRL quantities is available,
all patients of a specific procedure should be used.
2.
Body size differing from standard size
Standard body size of an adult population often is defined as 70 ± 10 kg.
Such samples are fine to set DRLs but will,
of course,
not cover very obese and slim patients.
More obviously,
paediatric DRLs cannot be based on any standard size.
The scientific literature,
summarised within the PiDRL project,
suggests the following pragmatic approach: While age grouping is fine for head examinations,
examinations of the trunk should be based on weight grouping,
and weight groups are proposed (<5 kg,
5-<15 kg,
30-<50 kg,
50-<80 kg).
Plotting continuous curves correlating exposure versus weight is an alternative approach that is especially useful when the number of patients per group is small.
Determining the effective diameter (and using size-specific dose estimates,
SSDE) or patient equivalent thickness may become the future parameters replacing weight (trunk) and age grouping (head); it will take some time and software development before these can be used in routine data collections of large populations.
3.
Defining DRLs based on clinical questions
Although the ICRP had postulated this approach some time ago,
nearly no implementation has occurred until recently.
Obviously,
coverage of all indications is not realistic,
and one has to concentrate on the most frequent examinations responsible for an important contribution to the medical population dose.
The EuroSafe Imaging campaign has started a project called ‘Is your Imaging EuroSafe?’ ( http://www.eurosafeimaging.org/survey) and decided to concentrate on CT (that represents >50% of the medical populaton dose in most European countries):
http://www.eurosafeimaging.org/survey) and decided to concentrate on CT (that represents >50% of the medical populaton dose in most European countries):
DRLs for ten important indications of CT of the trunk:
- CT head: acute head trauma
- CT head: acute stroke
- CT chest: pulmonary embolus
- CT chest: rule out pulmonary metastases of extrathoracic cancer
- CT chest: HRCT for diffuse parenchymal disease
- CT abdomen: liver metastases
- CT abdomen: urinary calculus
- CT abdomen: appendicitis
- CT Colonography
- Cardiac CT: Calcium coronary scoring
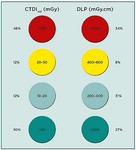
Preliminary results of the survey on CT in suspected pulmonary embolus are shown in Figs.
3 and 4: For 89% of the participants,
this was indeed a routine protocol,
and 87% used dose modulation.
Fig.
4 demonstrates a considerable spread of institutional values both for the CTDIvol and the DLP. The use of dose modulation reduced both exposure quantities, whereas the influence of iterative reconstruction was less clear.
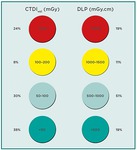
Fig.
5 shows the distribution of the same exposure quantities for CT evaluation of acute stroke.
While absolute values for CTDIvol and DLP were higher,
the spread was similarly wide.
Obviously,
this first attempt to define indication-specific DRLs has many limitations,
as sources of variation were not strictly controlled: patient factors (age,
gender,
size),
disease factors (prevalence,
case mix,
general diagnosis vs.
staging vs.
follow-up),
and -most importantly- imaging factors (representivity of sample,
protocol,
equipment,
calibration).
The EuroSafe Imaging campaign is currently lancing a new initiative to overcome the most important limitations: EuroSafe Imaging Stars is a project designed to identify and recognise imaging facilities across Europe that embody best practice in radiation protection (Fig.6).
By partnering with institutions that are committed to putting the principles advocated and concepts developed by the European Society of Radiology into practice,
EuroSafe Imaging aims to bridge the gap from raising awareness and advocacy to impacting the reality of clinical practice.
The group of EuroSafe Imaging Stars likely might be used to define more representative and more precise European indication-based DRLs for the above list of CT examinations.
More detailed information and standardised conditions will allow for an analysis of the importance and magnitude of the different sources of variation.
As dose is always related to image quality,
the minimal image quality needed to answer the specific clinical task will be defined and hormonised.
Furthermore,
as many EuroSafe Imaging Stars will likely have an automatic dose management system in their department,
dose registries will soon simplify the collection of national and European indication-based DRLs,
including their regular reviews and updates.