Congress:
EuroSafe Imaging 2016
Keywords:
Action 10 - Improved communication and collaboration of health professionals in radiation protection, Action 8 - EuroSafe Imaging Stars network and data collection, Action 3 - Optimisation, diagnostic reference levels, image quality, Action 2 - Clinical diagnostic reference levels (DRLs), Action 10 - EuroSafe Imaging Stars, Action 13 - Stakeholder engagement and collaboration
Authors:
D. Feldbergs, E. Rutka, K. Kupcs, A. Veiss, M. Radzina
DOI:
10.1594/esi2016/ESI-0038
Background/Introduction
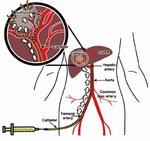
Transarterial chemoembolisation (TACE) is a minimally invasive procedure performed in interventional radiology to restrict a tumor’s blood supply.
This type of treatment affects cancer in two ways: first - the chemical substance is delivered in high concentration close to the tumor,
without exposing the patient's entire body to the toxic drug effect; second - TACE stops the blood supply to the affected site and the tumor no longer benefit for development of the necessary nutrients and oxygen.
At the same time it blocks the chemical substance escape from the tumor mass.
Annually in Latvia in P.
Stradins Clinical University Hospital Surgical Clinic we diagnose approximately 120 patients with hepatocellular carcinoma and cholangiocarcinoma.
Transarterial chemoembolisation is one of the therapies used when the surgery and chemotherapy is not possible.
Chemoembolisation of hepatic lesions in Latvia are performed since 2011,
therefore it is urgent to find out what is the dose of radiation during embolisation or it does not cause significant side effects to the patient,
as well as the amount of radiation compared with other therapeutic interventional radiology procedures.
Study hypothesis: Within the limits of appropriate radiation safety measures during chemoembolisation of liver lesions,
the permissible radiation dose is not exceeded.
The aim of study: evaluate the patients received radiation doses during procedure time of chemoembolisation of liver lesions.