Congress:
EuroSafe Imaging 2017
Keywords:
Breast, Action 11 - Dialogue with industry, Action 3 - Image quality assessment based on clinical indications, Action 2 - Clinical diagnostic reference levels (DRLs), Action 5 - Exchange and collaboration with medical industry, Action 4 - Quality of radiological equipment, Action 3 - Optimisation, diagnostic reference levels, image quality, Mammography, Physics, Workforce
Authors:
G. Sugrue, P. Gilligan, R. Ennis, N. Phelan, M. Lewis, L. Rainford, F. Flanagan, E. R. Stanley, M. F. J. Ryan
DOI:
10.1594/esi2017/ESI-0037
Background/Introduction
Breast cancer is the most common cancer affecting European women today and is the one of the leading causes of cancer mortality [i].
The premise for breast cancer screening is early detection and earlier treatment
[i].
The premise for breast cancer screening is early detection and earlier treatment [ii] and is founded on a large evidence base.
The establishment of a breast cancer screening programme was the precursor to a wider Irish cancer screening programme
[ii] and is founded on a large evidence base.
The establishment of a breast cancer screening programme was the precursor to a wider Irish cancer screening programme [iii] including cervical and colorectal cancer screening.
[iii] including cervical and colorectal cancer screening.
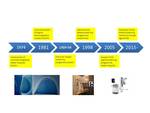
Mammography was first introduced into Ireland at the Mater Hospital in 1974 ( Fig. 1 ,
Fig. 2 ).
In 1989 Professor JT Ennis established a pilot breast screening programme.
This initial programme was part of a larger European project with the European Reference Organisation for Quality Assured Breast Screening and Diagnostic Services (EUREF) which promoted high quality mammography screening and breast care in Europe.
This enabled Ireland to gain expertise in high quality breast imaging.
Similar initiatives involving other breast imaging techniques were adopted in recent years,
involving PET-CT,
MRI and breast tomosynthesis.
In tandem with this,
a review of cancer services in 2009 by the Irish state led to the consolidation of public mammography services to eight specialist academic centers.
Incidents around poor quality mammography imaging contributed to the thinking behind this strategy [iv]. As part of the EU directives,
a screening programme involving radiation can only be carried out with the knowledge and approval of the Minister for Health.
[iv]. As part of the EU directives,
a screening programme involving radiation can only be carried out with the knowledge and approval of the Minister for Health.
Currently,
the number of public mammography machines in Ireland is 14.3/1,000,000 (Figure 3).
Since 2010,
there have been no analogue mammography machines in clinical use in Ireland.
Justification for mammography is carried out according to IREFER [v] and national screening guidelines
[v] and national screening guidelines
 [vi].
Currently,
MRI plays a limited role in routine breast screening.
BreastCheck,
the Irish national breast cancer screening programme,
was formally introduced in February 2000.
Breast screening continues to expand; currently women aged 50-64 are invited for breast screening,
and this is to be expanded to women aged 50-69 by 2021.
BreastCheck is now in its sixteenth year of screening and has provided almost 1.37 million mammograms to over 480,000 women and detected more than 8,500 cancersvi.
Since the inception of breast imaging services in Ireland,
a number of the techniques have been employed,
ranging from conventional x-ray,
xeromammography,
analogue and digital mammography. Optimisation of breast screening is multifactorial,
and the main contributors are outlined in Fig. 3 .
[vi].
Currently,
MRI plays a limited role in routine breast screening.
BreastCheck,
the Irish national breast cancer screening programme,
was formally introduced in February 2000.
Breast screening continues to expand; currently women aged 50-64 are invited for breast screening,
and this is to be expanded to women aged 50-69 by 2021.
BreastCheck is now in its sixteenth year of screening and has provided almost 1.37 million mammograms to over 480,000 women and detected more than 8,500 cancersvi.
Since the inception of breast imaging services in Ireland,
a number of the techniques have been employed,
ranging from conventional x-ray,
xeromammography,
analogue and digital mammography. Optimisation of breast screening is multifactorial,
and the main contributors are outlined in Fig. 3 .