Study population
Starting from 2008 we treated 151 children (166 eyes) for intraocular retinoblastoma with a total of 782 IAC sessions.
From August 2014 to August 2016,
44 patients (46 eyes with two bilateral retinoblastomas),
16 males and 28 females,
have been treated at our Institution with IAC and included in this prospective study.
Mean age was 35.4 months (range 9-227,
median 28),
mean cranial circumference was 49.6±3.2 cm.
IAC received approval from the local hospital Ethic Committee.
A multi-disciplinary group jointly discussed the opportunity of the treatment and obtained written informed consent from children’s parents or guardians.
Inclusion and exclusion criteria have been previously reported (8).
Standard therapy included a cycle of three treatments at a 3–4-week interval each; the joint evaluation of the multidisciplinary group might lead to the decision for further treatments.
Radiation parameters
IAC was carried out catheterizing directly the OA via the internal carotid artery (ICA).
If an adequate choroidal blush was unattainable,
alternative routes of IAC delivery through the anastomoses with the ECA were searched.
The so-called “balloon-technique” was never employed (10).
All procedures have been performed using Innova 3131 a biplane vascular radiographic imaging system with flat panel (GE Healthcare) using a low-dose pediatric protocol.
The usual rules of radiation safety were respected by all operators (13).
Mean fluoroscopic settings were: 79 kV,
1.1 mA,
15 frames per second with the exception of the phases characterized by an intracranial higher magnification that required 30 frames per second.
Mean subtraction angiography settings were: 76 kV,
162 mA,
2 frames per second.
The dose rate was continuously regulated by the automatic exposure control (including mA,
Kv and spectral filter between 0.2 and 0.3 mmCu).
Whenever possible the operative setting was LOW rather than NORMAL.
Source-to-image receptor distance was: 100 cm. Two different field of view (FOV) sizes were used: 30 and 20 cm.
Operative technique
IAC procedure was divided into 5 phases.
DAP and fluoroscopy time values were recorded for each phase as well as at the end of the whole session.
Phase 1.
Via the femoral sheath and under X-ray fluoroscopic guidance,
a 4F guide catheter was advanced through the aortic arch to the common carotid artery.
This step was characterized by the anteroposterior projection,
a large FOV and by LOW operative setting.
Phase 2.
The catheter was positioned within the ICA supplying the affected eye.
This phase was carried out with an anterior oblique projection at 45°,
a large FOV and with LOW operative settings.
Phase 3.
A lateral arteriogram was performed to verify whether the prominent flow in the OA was from the ICA or the ECA and to visualize the choroido-retinal enhancement and the cerebral vasculature.
This was the phase where the best pathway for drug delivery was chosen and it was carried out in a lateral projection with a small FOV.
The X-ray tube was positioned on the left side of patient’s head and the operative settings were LOW.
Phase 4.
The OA or a branch of the ECA were superselectively catheterized using a flow-directed microcatheter.
After the stability of the microcatheter tip was ensured,
a superselective angiogram was performed to check the patency of the OA and its branches and to look for the choroido-retinal enhancement.
After these mandatory check points,
a 30-min superselective infusion of the drug was carried out by a pulsatile injection.
A control was carried out after the injection of half chemotherapic dose to ensure that the drug was still effectively delivered to the retina.
If the choroidal blush was no longer attainable,
alternative routes were searched and the procedure resumed through a novel pathway as previously reported (9).
This step was characterized by a lateral projection with a small FOV and by NORMAL operative settings.
Phase 5.
The last phase consisted in a lateral arteriogram performed to rule out procedure-related complications such as vasospasm,
embolism or arterial dissection.
This phase was performed in a lateral projection,
with NORMAL operative settings and a small FOV.
Dosimetric measurements
The calculation of the absorbed dose (μGy) was obtained by conversion of the fluorescence signal from thermoluminescence dosimeters (TLD) (Steriotactic and Perioscopic System,
Radiation Products Desing Inc,
 www.rpdinc.com).
www.rpdinc.com).
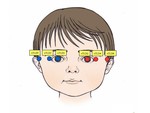
Six surface TLDs (sTLD) were placed in a newborn phototherapy mask and on patient’s eyelids as shown in figure 1.
Twelve deep TLDs (dTLD) were placed inside an anthropomorphic phantom (RSD Alderson Phantoms,
 www.rdsphantoms.com) with a cranial circumference of 58 cm (figure 2) and one (dTLD13) within the neck of the phantom.
www.rdsphantoms.com) with a cranial circumference of 58 cm (figure 2) and one (dTLD13) within the neck of the phantom.
In order to extrapolate the dose values in internal points from ESD as recorded in vivo,
a correlation between the external and internal doses was obtained by irradiating the phantom with the same settings and geometries to simulate the 5 treatment phases (figure 2).
Absorbed doses to the following critical structures were calculated: lenses,
retinas,
optic nerves,
hypophysis and thyroid.
Statistical analysis
DAPs,
fluoroscopy time and absorbed doses data sets were compared using Student’s T test (Microsoft Office Excel 97-2003).
The P value used for significance is 0,01.
The confidence level was 95% and the confidence interval was 6%.
DAP and fluoroscopy time
Forty-four patients underwent 216 IAC procedures (4.9 procedures per patient on average) with infusion of melphalan e topotecan.
Chemotherapic drugs were delivered into the OA catheterized via the ICA 196 times (91% of procedures) or into branches of the ECA in 20 sessions (9% of procedures).
Sessions in which chemotherapic drugs were delivered in part via direct catheterization of the OA and in part via branches of the ECA were considered in the latter group.
The mean DAP was 595±445 cGy·cm2 (median 432) and the mean fluoroscopic time was 540±403 seconds (median 418).
Separating procedures via the OA from those via the ECA it resulted that ECA pathway of drug delivery required higher DAP and longer fluoroscopy times (figure 3).
Assessment of absorbed doses in internal organs
A fixed correlation existed between the doses absorbed by sTLD and by dTLDs.
The dosimeter placed along the X-ray beam entrance pathway in latero-lateral projections (sTLD5) was fully invested by the primary beam in all phases of the procedure with the exception of phase 1 and,
possibly,
of phase 2.
Phase 1 was not included as its contribution could be considered insignificant.
Phase 2,
on the other hand,
was characterized by an irradiation field that just bordered sTLD5 (figure 2).
The relationship between the doses of sTLD5 and dTLDs,
could be subjected to an error as phase 2,
the only phase which employs an oblique projection,
contributed to TDLs irradiation for just 1.59% (via the ECA) and for 3.80% (via the ICA).
dTLD13 (thyroid) was invested by the X-ray beam also in phase 1.
Phase 1 summed to phase 2 contributed to dTLD13 irradiation for just 3.10% (via the ECA) and for 10.26% (via ICA).
With this margin of error,
the relationship between the measurements recorded by sTLD5 and dTLDs could be expressed by a series of coefficients.
Such coefficients,
therefore,
can be considered as correction factors that,
applied to the dose recorded by sTLD5,
allow a good estimation of the absorbed doses by internal organs.
Using these coefficients and with the exception of the left lens,
whose absorbed dose corresponds to that measured directly by sTLD2,
we were able to estimate the dose absorbed by the right lens,
both retinas,
both optic nerves,
hypophysis and thyroid starting from the dose recorded by sTLD5.
Internal organs received higher doses when IAC was carried out through branches of the ECA ranging on average from 10 to 30 mGy depending on the organ.
The high standard deviation of the doses absorbed by some organs was the result of the high variability of fluoroscopy time needed to perform IAC.
For this reason,
we also analysed data calculating the 75th and the 95th percentile values of the absorbed doses.
This analysis shows that the absorbed dose of internal organs was lower than 12.1 mGy in the left retina (the more exposed organ) in 75% of single treatments and lower than 25 mGy in 95% of treatments.
In case of 3 and 6 sessions the left retina of 75% of patients absorbed respectively less than 36.3 and 72.7 mGy whereas the left retina of 95% of patients received less than 75.2 and 150.4 mGy.
Other organs were less exposed.
After 6 sessions,
hypophysis absorbed less than 97.7 mGy in 95% of patients whereas thyroid received less than 62.1 mGy.
(figure 4)
Establishing a direct relationship between DAP and sTLD5
ESD was on average 9.59 mGy (range 0.8 to 165 mGy).
The ratio between DAP (expressed as cGy·cm2) and the dose recorded by sTLD5 (expressed as μGy) was fairly constant (mean = 80±19 cm-2).
The consistency of the ratio is unveiled by the narrow standard deviation and it is better evident plotting the ratio as a function of the frequency of times in which it was recorded (figure 5).