The various potential transplant-related complications make pregnancy more risky than in the general population for both the mother and the fetus [1]. In addition, in situations in which an interventional procedure is deemed clinically necessary with no possibility of delaying it until after pregnancy and US cannot be used for guidance, the radiation related risks must be kept in mind. These include spontaneous abortion, teratogenesis, and carcinogenesis, all of which are related to the fetal gestational age, imaging modality used and the fetal absorbed dose [2].
The average fetal effective dose due to the naturally occurring background radiation exposure is estimated to be approximately 0.5-1 mSv for a 9 months pregnancy [3-4]. If x-ray exposure does not involve direct fetal or abdominal exposure then the fetal dose should be negligible [3]; on the contrary, if the abdominal region is involved radiation exposure may be high [5].
As emphasized in the European Directive 2013/59/Euratom, the “as low as reasonably achievable” principle should be applied. This means that a procedure or examination should achieve the clinical aim with the appropriate dose, thus all dose management strategies existing for fluoroscopy and interventional radiology should be used. Fluoroscopy time should be limited, the patient should be placed as close to the detector as possible, the distance from the source of the x-ray maximized, the lower frame rate available on the equipment should be employed, collimation optimized and magnification used only when necessary. Also, a lead apron should be placed between the patient and the table to protect the uterus from external scattered radiation.
A more recent paper describes other reduction strategies than those commonly used: first, removal of the anti-scatter grid during routine biliary procedures performed on adults is an effective method of significantly decreasing patients’ radiation exposure [6]; second, awareness of how the auto exposition control system works and how to make the most of it allows for choice of the correct auto exposition trajectory for the procedures to be performed [7]; third, recording and reviewing patient dose improves patient safety and raises dose awareness in fluoroscopically guided procedures [8-9].
Our aim was to perform a clinically necessary fluoroscopy-guided percutaneous biliary catheter change (BCC) safely in a pregnant liver transplant patient at seventeen weeks gestation by applying all the above dose reduction strategies.
The patient was a 30 year old lady who had received cadaveric liver transplant . Prior to her pregnancy she underwent percutaneous transhepatic biliary drainage (PTBD) in view of stricture of the bilio-enteric anastomosis. During the second trimester of her pregnancy she was found to have a deranged liver profile which was initially monitored clinically but was noted to worsen progressively. In view of the worsening liver function and high risk of obstruction of the biliary catheter it was deemed necessary that she undergo a biliary catheter change under fluoroscopic guidance.
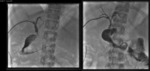
Also, the patient underwent a bilioplasty procedure three months before the biliary catheter change described in this procedure. (Figure 1) In our clinical practice, we currently use a low threshold dose fluoroscopy trajectory when performing interventional procedures and we apply all the possible dose reduction strategies. At the time of the bilioplasty, the patient was unaware that she was 3 weeks pregnant. Nonetheless, a low dose fluoroscopy protocol (50% threshold dose) with 7.5 fluoroscopy frames/sec (the lowest for our angiography suite at the time, the GE Innova 4100) and low image detail level was employed. Magnification and normal image detail level was used only in critical steps of the procedure. The routine recording of patient’s dose indicators, such as Dose Area Product (DAP) and Air Kerma (AK) (3.8 Gy*cm2 and 0.017 Gy respectively for the bilioplasty procedure), allowed the medical physicist to calculate patient’s effective dose (0.68 mSv) and the uterus equivalent dose (0.24 mSv) two weeks after the bilioplasty was performed. (Figure 2)