Congress:
EuroSafe Imaging 2021
Keywords:
Management, Radiation physics, Radioprotection / Radiation dose, Nuclear medicine conventional, Dosimetry, Radiation safety, Radiation therapy / Oncology, Quality assurance
Authors:
A. G. Umolu, N. Mahmood, J. Clark, M. Hierath, A. Brady, G. Frija, D. Howlett
DOI:
10.26044/esi2021/ESI-13019
Results or findings
- The project will seek to evaluate current clinical audit practice and existing guidance across European member states
- QuADRANT will also aim to identify perceived barriers to audit participation and provide recommendation to improve uptake of good clinical audit practices to enhance patient experience and outcomes
SPECIFIC QuDRANT OBJECTIVES
-
-
- Review the status of implementation of clinical audits in the member states
- Identify good practices in Member States available guidance and resources for clinical audits, national, European and international level
- Provide further guidance and recommendation on improving the implemantation and integration of clinical audits into national healthcare systems
- Identify potential for further coordinated EU action on quality and safety of radiology, radiotherapy and nuclear medicine.
- QuADRANT comprises five work packages, including two conferences and a pan-European survey
- With input from European professional societies and national/European/international organizations (including IAEA, WHO, HERCA)
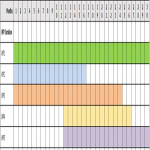
Project Timeline
Work Packages (WP)
-
- WP1:
- Objective: Project management and implementation (Month 1-30)
- Co-ordinator: ESR
- Led by D.C Howlett (Project lead), A. Brady (Project co-lead) and the ESR project Office: Mr J Clark, Ms M Hierath
- WP2: Webinar workshops (14th to 16th December 2020)
- To discuss the current state of play of clinical audit to the EU
- Discuss legal requirements and benefits of clinical audit
- Identify good practices, existing guidance
- Involving key players and healthcare/administrative representatives from member states
- WP3: This involves:
- Pre-survey to collect details/information of persons/bodies involved in clinical audit in the EU and 4 other countries (United Kingdom, Switzerland, Iceland, Norway) which has been completed
- Main survey (post WP2) to gather detailed information on clinical audit practice/ process across Europe- to launch in early 2021
- To include literature review plus expert interviews
- WP4: A workshop (late 2021)
- To present WP3 survey results (plus WP2 output)
- Discuss and present a way forward with member state representatives and professional experts
- WP5: A report for the European Commission, based on output from WP2, 3, 4, literature search interviews
- Collection of best practices
- Recommendations on improving implementation/integration of clinical audit into national healthcare systems, ultimately enhancing patients experiences and outcomes.