Keywords:
Radiation therapy / Oncology, Experimental, Breast, Radiotherapy techniques
Authors:
H. Kleiven1, N. Tejani2, T. Squire1, G. Buchanan1, S. Powell1, L. Castle-Burns1, S. Bray1, S. Watson1, M. Burnell1, V. Coleman1, N. Davis1, J. Spence1, H. Elsaleh1; 1ACT/AU, 2VIC/AU
DOI:
10.1594/ranzcr2018/R-0037
Methods and materials
The trial was approved by The Canberra Hospital Human Research Ethics committee.
The Dermalab Combo is a probe-based platform capable of quantitative assessment of multiple skin parameters in a non-invasive manner including:
-
skin erythema index
-
skin hydration
-
dermal thickness
-
elasticity
-
water permeability
Acute reliability of captured parameters was assessed by repeat measurements on the forearm of a single volunteer,
and natural variation longitudinally over a two-week period on two pre-defined areas of the untreated breast of a female volunteer.
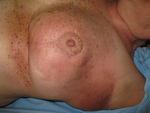
Our target study group was patients planned for adjuvant breast radiotherapy,
where the main side effect is skin toxicity.
Dermalab Combo assessment was performed on the upper breast,
lower breast and -if applicable- boost site,
at baseline,
twice weekly during treatment and 6 weeks after radiotherapy completion.
We concurrently captured RTOG skin toxicity grading and patient behaviour related to use of creams.
Analysis and statistics were performed using R.