Keywords:
CNS, Neuroradiology brain, Oncology, MR, Diagnostic procedure, Cancer, Metastases
Authors:
A. Lasocki1, L. Caspersz1, G. McArthur2; 1Melbourne, VIC/AU, 2Parkville, VIC/AU
DOI:
10.26044/ranzcr2021/R-0383
Methods and materials
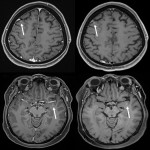
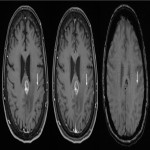
Institutional ethics committee approval was obtained. Patients were identified prospectively on the basis of an upcoming brain MRI booking and a history of metastatic melanoma. Only patients with either no known active IMM or low-volume IMM (no IMM >10mm in long axis), based on the preceding MRI report, were included. This included patients with successfully treated IMM. Verbal consent was obtained. A cohort of 100 examinations was planned. All MRI examinations were performed on a 3-Tesla MRI (MAGNETOM Skyra, Siemens, Erlangen, Germany) utilizing a standardized protocol, including pre- and post-contrast 3D-GRE (MPRAGE with water excitation), post-contrast 3D-TSE (T1-SPACE, with fat saturation), axial T2WI, axial FLAIR, DWI and Susceptibility-Weighted Imaging. Post-contrast T1-SPACE was performed after post-contrast MPRAGE.
Two MRI reads of each study examination were performed, at least two months apart to minimize recall bias. First, T1-SPACE images were assessed after MPRAGE, noting additional findings identified by T1-SPACE. MPRAGE images were subsequently reviewed, to determine whether the additional findings demonstrated by T1-SPACE were considered consistent with additional metastases, convincingly FP (most commonly normal vessels) or remained equivocal. When reported by other radiologists, the imaging report was subsequently reviewed after interpretation for quality assurance purposes. The order of reviewing the sequences was reversed for the second assessment.