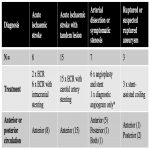
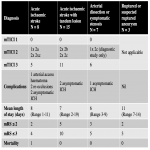
Thromboembolic events are amongst the most common and significant complications of neurointerventional procedures. In the emergent setting, prompt administration of prophylactic antiplatelet agents is thus of utmost importance.
IV ASA can reduce platelet function up to four times faster than oral formations, and also overcomes barriers to oral, nasogastric or rectal antiplatelet administration.[1] As such, IV ASA has become increasingly utilized for a variety of time-critical neurointerventional procedures, most commonly endovascular clot retrieval (ECR) and stent-assisted aneurysm coiling.
IV ASA in the Treatment of Acute Ischaemic Stroke
IV ASA was first proposed as a safe therapeutic agent in acute ischaemic stroke (AIS) in 1998, when it was reported to reduce the risk of microembolic events in AIS without significantly increasing the risk of intracranial haemorrhage (ICH).[1] Amongst the rapid emergence of major ECR trials, a 2015 study across four German stroke centres found that prophylactic IV antiplatelets did not increase the risk of ICH after ECR, when compared with oral antiplatelets.[3] This safety profile was further supported by a 2017 retrospective ECR trial in which IV ASA, when given before or during bridging thrombolysis, did not increase the risk of ICH.[4] As such, IV ASA rapidly became the standard of care for all cases of ECR with carotid artery stenting (CAS) in 2018, when four prospective studies each included 250-1000mg IV ASA just prior to stenting as part of their protocols.[5,6,7,8]
The most rigorous evaluation of IV ASA to date is MR CLEAN-MED, a multicentre randomised clinic trial that aims to assess the effect on functional outcomes of IV ASA and IV heparin (groups: IV ASA, IV heparin, both, neither).[9] Having commenced in 2017, this trial is amid recruitment of 1500 patients, and the results will be important in quantifying the efficacy of IV ASA.
IV ASA in the Treatment of Intracranial Aneurysms
In endovascular coiling of intracranial aneurysms, use of IV ASA has been reported as early as 2006, when administered for all patients in the last two years of a four-year consecutive case series.[10] This study reported significantly fewer thromboembolic complications in the ASA group, without significant difference in aneurysm perforation. More recently, with advances in coil adjuncts, IV ASA has become standard of care for stent-assisted coiling, with a trend towards the provision of even more antithrombotic agents. For example, in a 2020 trial examining 61 patients with ruptured aneurysms treated with stent-assisted coiling, IV ASA was given by protocol on stent deployment, and 25% were also given heparin bolus.[11] Those given only ASA had significantly higher rates (21% vs 12%) of overall complications, such as ischaemia and haemorrhage, with stent thrombosis remaining a common complication (36% of all patients), not associated with the antithrombotic combination.
This study aims to present the modern use of IV ASA within a comprehensive neurovascular centre over a 12-month period, examining the short term technical and clinical outcomes.