Keywords:
Thyroid / Parathyroids, Ultrasound, Audit and standards, Biopsy, Cancer
Authors:
J. Lee, A. Varma
DOI:
10.26044/ranzcr2022/R-0007
Methods and materials
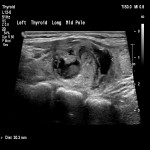
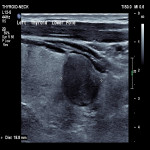
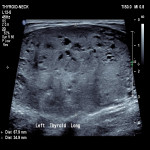
ACR TI-RADS is a classification system that standardises thyroid ultrasound reporting while also predicting the rate of malignancy using multiple ultrasound parameters. These parameters include composition, echogenicity, shape, margins and echogenic foci. These parameters are assigned a score and the total score along with the size of the nodule determines the classification and associated recommendation. These recommendations include no follow up, follow up interval and FNA.
Ultrasound guided FNA is minimally invasive, economical and relatively safe. It is the most commonly used method to differentiate benign from malignant thyroid nodules. The Bethesda System for Reporting Thyroid Cytopathology has also been developed to standardise and classify cytology findings with the associated risk of malignancy.
This study is a single-centre retrospective study conducted from August 2019 till August 2021 that evaluated 79 thyroid nodules in 74 patients. The nodules were classified according to TI-RADS and compared with cytological results from subsequent FNA. Periprocedural complications were also recorded.