Keywords:
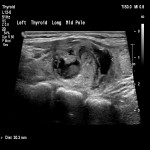
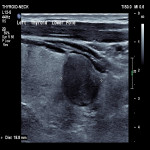
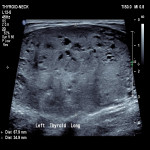
Thyroid / Parathyroids, Ultrasound, Audit and standards, Biopsy, Cancer
Authors:
J. Lee, A. Varma
DOI:
10.26044/ranzcr2022/R-0007
References
1.Huh, S., Lee, H.S., Yoon, J. et al. Diagnostic performances and unnecessary US-FNA rates of various TIRADS after application of equal size thresholds, Scientific Reports, 2020 June 30; Volume 10 (No 1): p10632
2.Sharma, P., Elfatairy, K., Gandhi, D. et al. Diagnostic Performance of ACR-TIRADS in Differentiating Benign From Malignant Thyroid Nodules in Patients Undergoing Fine-Needle Aspiration Biopsy: Comparative Study Based on Five International Guidelines for Management of Thyroid Nodules, Journal of Endocrinology and Metabolism, 2021 July 11; Volume 11(No 3-4), p69-75
3.Tessler, F., Middleton, W., Grant, E. et al. ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White Paper of the ACR TI-RADS Committee, Journal of the American College of Radiology, 2017 March 31; Volume 14 (No 5): p587-595
4.Polyzos, S., Anastasilakis, A., Clinical complications following thyroid fine-needle biopsy: a systematic review, Clinical Endocrinology, 2009 July 6; Volume 71 (No 2): p157-165