IPF can be classified based on imaging findings on HRCT as outlined by the ATS/ERS/JRS/ALAT group [3]. Based on this, IPF can be categorised as:
- Usual interstitial pneumonia (UIP)
- Probable UIP
- Indeterminate for UIP
- Alternative diagnosis
There is moderate interobserver agreeance, even amongst thoracic radiologists, on how the ILDs are categorised. HRCT of biopsy-proven UIP also has atypical features in up to 30% of cases, so this guide is still being actively refined [3].
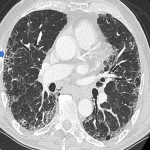
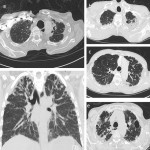
The UIP pattern (Figure 1) is characterised by honeycombing and traction bronchiectasis. There may be ground-glass opacity superimposed on a fine reticular pattern, although lone ground-glass opacity is not typical of UIP. The distribution of UIP is typically subpleural with basal predominance [3].
Probable UIP refers to the pattern described above in the absence of honeycombing [3].
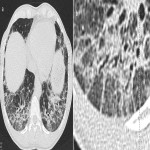
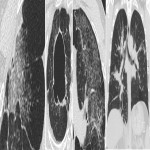
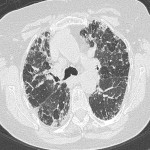
Indeterminate UIP (Figure 2) refers to the presence of fibrosis without features suggestive of UIP such as honeycombing or traction bronchiectasis. There may be subtle reticulation [3].
The category of alternative diagnosis should be used when there are features more suggestive of another aetiology of ILD on HRCT, for example pleural plaques which suggest asbestosis, or a dilated oesophagus which suggests connective tissue disease [3].
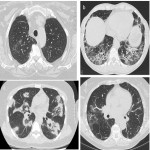
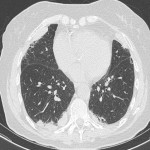
Organising pneumonia (OP) is a pattern of alveolar and bronchial inflammation which can occur in response to a variety of insults, including those of infective and non-infective aetiology. Cryptogenic organising pneumonia (COP) is the specific term assigned when no cause is identified. COP is a form of IIP mostly seen in patients in the fifth and sixth decade of life. Characteristic radiological features include multifocal peribronchial and subpleural consolidation with air bronchograms and ground-glass opacities, with or without fibrosis. These findings evolve over weeks (Figure 3). Most cases respond to steroid therapy [5].
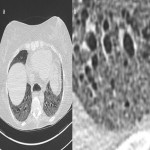
Non-specific interstitial pneumonia (NSIP) is another common pattern of ILD and can be further subcategorized into fibrotic or cellular NSIP, depending on the degree of associated fibrosis or volume loss. NSIP has a slight female predominance and it typically occurs in younger patients compared to IPF [6]. The important distinguishing factor from UIP is the temporal and spatial homogeneity of changes. Radiologically, it is characterized by extensive ground-glass opacity in inter and intra-lobular spaces. Subpleural sparing is a feature. There is often a basal predominance (Figure 4) [6].
Connective tissue diseases can also present with varying patterns of ILD. A common appearance of these is an NSIP pattern [6]. Features such as oesophageal dilation can support underlying connective tissue disease (Figure 5) [3].
Pleuroparenchymal fibroelastosis (PPFE) is a form of IIP characterised by extensive fibrosis involving the upper lobe with collagen and elastin in the subpleural region, resulting in narrowing of the anteroposterior diameter of the chest (Figure 6). PPFE is idiopathic, but has a genetic predisposition and associations with infection with mycobacterium avium complex or aspergillus and bone marrow transplant have been proposed. The prognosis can vary widely [7].
Granulomatous ILD is most commonly caused by sarcoidosis (Figure 7), which mainly occurs in the third decade of life and has mild female predominance. This is a non-caseating, multisystem granulomatous process of unknown aetiology thought to be secondary to immune dysregulation in genetically susceptible individuals. In the chest, sarcoidosis can affect the mediastinum, lung parenchyma or a combination of both. The findings on HRCT include bilateral hilar lymphadenopathy (which can be calcified), consolidation or ground glass opacities, perilymphatic micronodules and fibrotic changes [8].
Exposure-related ILD encompasses pneumoconioses, such as asbestosis and silicosis, and hypersensitivity pneumonitis, for example farmer’s lung [9]. The mainstay of diagnosis is history of exposure to a causative agent. The different diseases within this category each have characteristic features on HRCT, for example perilymphatic and centrilobular nodules in silicosis, and pleural abnormalities such as thickening and plaques in asbestosis (Figure 8). Hypersensitivity pneumonitis (Figure 9) is associated with centrilobular ground-glass nodules in acute stages and mosaic attenuation, reticular abnormality and honeycombing as the disease progresses.
Management of ILD
The management of ILD is referred to the respiratory specialist and multidisciplinary team, to determine if there is in fact ILD and, if there is, if it can be categorised further. From here, management may include antifibrotics or steroids, sampling for a more definitive diagnosis, or watchful waiting.
Despite radiology input, biopsy and multidisciplinary team discussions, 10% of ILDs remain unclassifiable. This is fine, as the most important feature here is the clinical progression of the disease. ILD can have five types of clinical behaviour, each with its own treatment goal and surveillance strategy [10].
- Reversible and self-limited: the cause should be removed and these should have short-term observation with HRCT at 3-6 months to confirm regression.
- Reversible with risk of progression: consider long-term therapy and monitor with both short-term observation to check for treatment response and long-term observation to monitor for progression.
- Stable disease: aim to maintain stability and monitor long-term for progression.
- Progressive disease with potential for stabilisation: aim to stabilise with therapy and monitor long-term to assess disease course.
- Progressive disease despite therapy: aim to slow the progression of the disease and monitor long-term to assess the disease course and the need for organ transplant or palliation.
For radiologists, the role is mainly to stratify the dominant patten to predict the disease course and exclude potential causes such as connective tissue disease or exposure-associated ILD. It is crucial to work as a team, with periodic assessment of patients, to make sure the working diagnosis and treatment plan changes as the clinical picture unfolds.