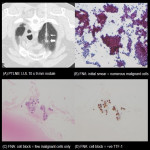
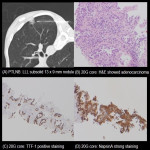
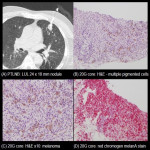
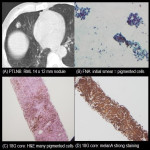
Image guided (CT) PTLNB is standard diagnostic procedure for nodules >= 8 mm with intermediate probabilities of malignancy (BTS guideline for investigation and management of pulmonary nodules 2015) [5].
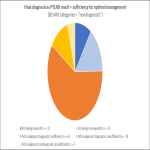
PTLNB pathologic results can be subdivided into five categories:
Diagnostic = specific benign findings, malignant findings;
Non-diagnostic = nonspecific benign pathologic findings, atypical cells, insufficient specimen.
Specific benignity is assigned for results showing distinctive diagnoses such as hamartoma or tuberculosis. Nonspecific benignity is defined when adequate sample without suspicious or malignant features are present, but insufficient for definitive diagnosis (eg granulomatous inflammation, undifferentiated abscess, or organizing pneumonia).
The accuracy of PTLNB is reported to be high. The BTS guideline review derived overall sensitivity/specificity: 90/95% from the published literature [5].
Huang et al reported that accuracy may be less (80-85%) for small nodules <= 15 mm [6].
Nevertheless PTLNB can be technically challenging (particularly for smaller nodules) and results may commonly be inconclusive. The overall utility of the procedure (as opposed to the reported high diagnostic accuracy) depends on how these inconclusive results are interpreted (ie classified as non-malignant). Indeed, the negative predictive value for non-malignant result may be only 50% [7].
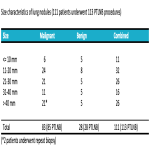
Recent large multicentre study from Korea of 9384 PTLNB procedures (Lee et al) reported overall nondiagnostic biopsy rate of 27.6% (and 35% for nodules <= 20 mm) [1]. Whilst the majority of this limitation was found for benign nodules, these authors found nondiagnostic biopsy rate of 14.4% for nodules with final malignant diagnosis (77.5% of the 9384 biopsies).
In the developing oncologic era of targeted therapies and immunotherapies, access to optimal treatments for primary and secondary lung malignancies increasingly requires comprehensive immunohistochemical (IHC) and additional molecular studies.
Standard IHC stains can be performed on most biopsy specimens, even with small samples taken via fine needle aspiration. However, gene panels and molecular testing are more likely to be successfully performed with larger biopsy samples, which places greater demand for adequate needle tissue sampling. This is particularly relevant for non-small cell lung carcinoma and some metastatic diseases to lung.
If the sample is insufficient for complete IHC and molecular testing, repeat biopsy may be pursued, exposing the patient to further procedural risks, and resulting in delays to initiation of systemic therapy.
Some examples of molecular testing panels required for modern systemic therapies for range of common malignant lung tumour streams is outlined in [Fig 1].
PD-L1, ALK and ROS1 testing requires at least 100 viable (not degenerate) malignant cells in order to be successfully performed.
EGFR and BRAF are tests where DNA/RNA is extracted from viable (not degenerate) cells. If the core biopsy/cell block only has scattered/single cells, only small cell groups or malignant areas, or viable cell loss occurs during slide cut processing, this will also result in insufficient material for analysis.
In practice, when performing lung nodule biopsies where one of the listed tumour streams [Fig 1] is suspected, obtaining larger tissue samples including core biopsy where possible, and with on-site cytopathologist support, may reduce the likelihood of insufficient material and need for repeat biopsies.
Several studies have reported that the negative diagnostic yield of PTLNB for mutational analysis may be of the order of 5-20%, but this is mainly in the setting of therapeutic research trials [2-4].
The factors which limit the diagnostic performance of PTLNB for molecular testing have not been widely studied or emphasised to general procedural radiologists. The impact of nodule morphology and modified biopsy techniques on diagnostic utility remains unclear, particularly as it impacts routine thoracic oncology practice [1].