Learning objectives
To review the normal anatomy of the systemic venous return.
To understand the basics of the embryology of the venous system.
To acknowledge and recognize the most common congenital anomalies of the systemic venous return to the right atrium.
Background
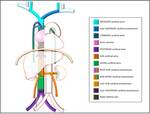
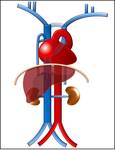
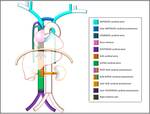
Typically the venous return from the upper half of the body it’s done by the right-sidedsuperior vena cava (SVC) formed by the confluence of the brachiocephalic veins.
The blood from the lower half of the bodydrains typically through the right-sidedinferior vena cava (IVC),
resulting from the confuence of the common iliac veins.
Theazygos-hemiazygos venous system,running up the side of the vertebral column,may connect both systems and provide an alternative path to the right atrium when either of the venae cavae is interrupted.
Anomalies of the...
Findings and procedure details
ANOMALIES FROM THE UPPER HALFBODY VENOUS RETURN
(SVC)
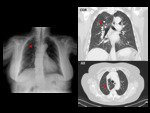
Isolated anomalies of the right SVC are rare,
and consist of low insertion into the right atrium,
drainage to the left atrium,
interruption,
or aneurysmal dilatation.
From the persistence of the left anterior cardinal vein results a double SVC,
and if,
in addition,
the normally persistent right cardinal vein regresses,
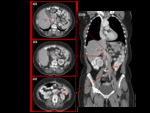
then there is only a left SVC. Fig. 6
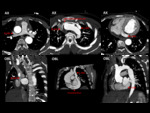
Persistent left SVC / SVC duplication Fig. 4 Fig. 5
A persistent left SVC,
in most cases as...
Conclusion
Radiologists must acknowledge and be aware of the normal venous anatomy and most common congenital anomalies of the systemic venous return,
in order to avoid diagnostic pitfalls,
as although rare these are incidentally found.
Personal information
João Diogo OLIVEIRA, Radiology resident,
Department of Radiology,
Centro Hospitalar Lisboa Ocidental E.P.E.,
Lisbon,
Portugal
Isa MARTINS, Radiologist,
Department of Radiology,
Centro Hospitalar Lisboa Ocidental E.P.E.,
Lisbon,
Portugal
References
Mazzucco A,
Bortolotti U,
Stellin G,
Galucci V,
Anomalies of the systemic venous return: a review,
J Card Surg 1990; 5:122-133.
Terrence C.
Demos1,
Harold V.
Posniak,
Kenneth L.
Pierce,
Mary C.
Olson,
Mark Muscato; Venous Anomalies of the Thorax; AJR:182,
May 2004.
Bass JE,
Redwine MD,
Kramer LA et al.,
Spectrum of congenital anomalies of the inferior vena cava: cross-sectional imaging findings,
Radiographics 2000; 20: 639-652.
Cole TJ,
Henry DA,
Jolles H,
Proto AV.
Normal and abnormal vascular structures that simulate neoplasms on chest...