MOLECULAR AND PHYSICOCHEMICAL PROPERTIES
Molecular Structure - Gadobenate dimeglumine comprises a linear, ionic chelate of the paramagnetic Gd3+ ion (Gd-BOPTA), salified with two molecules of meglumine. The structure resembles that of gadopentetate dimeglumine (Magnevist®, Gd-DTPA), apart from the addition of a hydrophobic benzyloxymethyl substituent (Figure 1). This substituent confers on gadobenate dimeglumine two properties that differentiate it from gadopentetate dimeglumine and the other conventional Gd contrast agents: markedly higher r1 and r2 relaxivity, and a dual route of elimination through both the renal and hepatobiliary pathways [1,4].
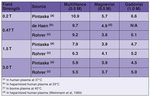
The benzyloxymethyl substituent also confers slightly increased stability compared to gadopentetate dimeglumine and other linear Gd chelates, as evidenced by the conditional stability constants (Table 1) [5]. While excess chelate is added to the formulation of some Gd chelates, such as gadodiamide and gadoversetamide, to minimize the presence of free Gd3+ ion in the formulations of these less stable agents [6-8], no excess chelate is added to the formulation of gadobenate dimeglumine. The release of free Gd3+ ion has been postulated as the reason for the increased retention of Gd3+ ion in bone after administration of gadodiamide [9,10] and may also be related to the increased transmetallation observed with these agents [11,12]. The presence of excess chelate or the in vivo release of free Gd3+ ion have both been suggested as potential causes of the spurious hypocalcemia observed to occur during routine laboratory assays for serum calcium, a phenomenon not seen with gadobenate dimeglumine [13-16].
Relaxivity - The weak and transient interaction of gadobenate dimeglumine with serum proteins results in a slowing of the tumbling rate of the molecule, which in turn causes a shortening of both the T1 and T2 relaxation times and concomitant changes in both the r1 and r2 relaxivities of gadobenate dimeglumine compared to other Gd agents [1,17]. The higher r1 relaxivity of gadobenate dimeglumine noted in early studies at 0.47T [1] has recently been shown to hold true at all commercially available magnetic field strengths: the r1 and r2 relaxation rates for gadobenate dimeglumine at 0.2T, 1.5T, and 3.0T were 107%–131% and 91%–244% higher than for gadopentetate dimeglumine, respectively (Tables 2 and 3) [1,2,18,19].
Clinical studies aimed at comparing gadobenate dimeglumine with conventional Gd contrast agents have consistently demonstrated improved contrast enhancement with gadobenate dimeglumine due to the higher r1 relaxivity of this agent. An experimental study conducted recently to confirm differences in relaxivity between Gd contrast agents and to explore image sequence optimization at 1.5T and 3T revealed that the highest signal intensities were obtained with gadobenate dimeglumine and that the effect of the greater r1 relaxivity at both 1.5T and 3T is improved with shorter TR and TE values [20]. These differences may explain the improved imaging performance of gadobenate dimeglumine in numerous intraindividual crossover trials [21-33].
Pharmacokinetics - Although mostly cleared via the kidneys, gadobenate dimeglumine is also paritially taken up by functioning hepatocytes (3% to 5% of the injected dose) and excreted via the hepatobiliary pathway. As a results, gadobenate dimeglumine may also be used not only for conventional dynamic phase imaging of the liver, but also for delayed liver imaging in a manner similar to that of the purely liver-specific agents (eg, mangafodipir, Mn-DPDP, Teslascan™], ferumoxides [Endorem®, Ferridix™] and gadotexate [Primovist™]).
IMAGING OF THE CENTRAL NERVOUS SYSTEM
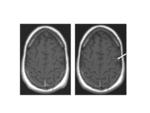
Conventional T1-Weighted Imaging - Accurate diagnosis and characterization of primary tumors and metastatic lesions of the CNS is essential if the appropriate treatment option is to be selected. The use of Gd-enhanced MR is routine for neurooncologic patient evaluation, and provides information complementary to noncontrast MR imaging. For patients with intraaxial glial tumors, patient management decisions are based largely on findings related to the extent of lesion contrast enhancement and what these findings indicate regarding the potential for tumor resection. Similarly, treatment options for patients with metastatic lesions are typically selected based on the size, number, and location of lesions depicted on contrast-enhanced MR images.
Gd contrast agents improve lesion conspicuity relative to surrounding brain parenchyma. Interindividual parallel group or crossover comparative studies among the conventional Gd contrast agents have revealed no differences in the extent of lesion signal enhancement or diagnostic performance [34-38], a finding consistent with the similar r1 relaxivity values of these agents. Moreover, several studies have demonstrated the necessity for double and triple doses of conventional agent in order to achieve sufficient improvement in diagnostic efficacy for detection of small or poorly-enhancing lesions [39-44].
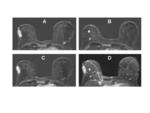
Compared with conventional Gd agents, the higher-relaxivity gadobenate dimeglumine has demonstrated improved efficacy at equivalent doses and equivalent efficacy at lower doses for the detection and characterization of CNS lesions [45-47]. However, the most compelling evidence for improved performance comes from intraindividual crossover studies, in which patients are randomized to receive one contrast agent then another in two identical MR studies typically performed within two weeks. In an early retrospective analysis of patients from a multicenter, multinational, phase II study, patients receiving comparator agents (gadopentetate dimeglumine, gadodiamide, or gadoterate meglumine) were then imaged in an identical manner within 30 days with gadobenate dimeglumine [21]. Sensitivity for lesion detection with gadobenate dimeglumine (93%–100%) was markedly superior to that of the pooled comparator-enhanced examinations (65%–73%). The increase in the lesion-to-brain contrast of the main lesion was consistently greater with gadobenate dimeglumine than with comparator agents relative to unenhanced images (143% vs 127%).
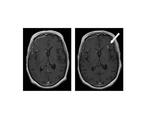
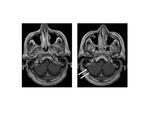
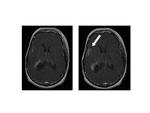
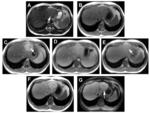
Four independent, prospective, intraindividual crossover studies have been performed using essentially identical imaging protocols and image evaluation endpoints. Overall, a total of 314 patients with brain or spine lesions underwent two MR exams within 2 days to 2 weeks with equal 0.1 mmol/kg doses of both gadobenate dimeglumine (N=314) and a comparator agent (gadopentetate dimeglumine [N=178], gadodiamide [N=113], or gadoterate meglumine [N=23]) [22-25,48]. T1w SE and T2w fast SE images were obtained before, and T1w SE images after contrast injection. Blinded experts assessed the two sets of postcontrast images for both qualitative (global contrast enhancement, lesion delineation, internal lesion morphology and structure, tumor vascularization, and global image preference) and quantitative (contrast-to-noise ratio [CNR]; percent lesion enhancement) efficacy parameters. In all cases, significant (p<0.05) reader preference was noted for gadobenate dimeglumine-enhanced images (Figures 2-5). Similarly, quantitative assessment revealed significantly (p<0.05) greater lesion enhancement and CNR with gadobenate dimeglumine [22-25]. Analysis of a subset of 84 patients with intraaxial enhancing brain lesions from one study [24] revealed significantly (p<0.0001, all readers) better diagnostic information and imaging performance with gadobenate dimeglumine for all visualization endpoints [49].
As in adults, gadobenate dimeglumine has demonstrated utility for detection and characterization of CNS lesions in pediatric patients [50]. Specifically, a parallel group study of 63 children with tumors of the brain or spine revealed significantly better pre- to postdose changes in border delineation and contrast enhancement with 0.1 mmol/kg gadobenate dimeglumine compared with 0.1 mmol/kg gadopentetate dimeglumine.
Increased diagnostic information can lead to improved patient management. The ability to use lower doses of gadobenate dimeglumine with equivalent efficacy, or higher doses when the clinical situation demands, may help optimize the diagnostic workup of individual patients, potentially reducing imaging costs while decreasing hospital stays and avoiding inappropriate surgery.
Brain Perfusion Imaging - MR brain perfusion imaging is frequently used for evaluation of cerebrovascular disease and brain tumors, and for monitoring patients following radiotherapy. Perfusion imaging characterizes blood flow through the brain, providing physiologic information relevant to the underlying pathology. Perfusion maps are generated using T2w imaging sequences after the rapid injection of a contrast agent bolus. The susceptibility effect of the passing agent causes a decrease in the signal intensity in the tissue, which varies between normal tissue and lesion. Typically, a 20% drop in T2 signal is required to provide perfusion maps of adequate diagnostic quality. To achieve this signal loss with conventional Gd contrast agents, increased doses (0.15 or 0.2 mmol/kg) are often required. With its higher r2 relaxivity [2,20], gadobenate dimeglumine might be expected to provide a sufficient drop in signal, and thus good-quality perfusion maps, with just a single 0.1 mmol/kg dose. In a recent comparison of gadobutrol, a 1 M Gd contrast agent, and gadobenate dimeglumine for brain perfusion, both allowed the acquisition of high-quality perfusion maps at 1.5T using standard 0.1 mmol/kg doses [51]. The susceptibility effect was comparable for both agents and was stronger than that for conventional Gd contrast agents.
MAGNETIC RESONANCE ANGIOGRAPHY
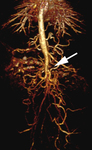
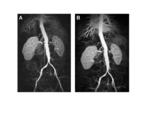
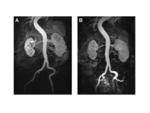
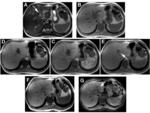
As technical advances improve the performance of MRA, the preference for this noninvasive imaging study over conventional catheter angiography continues to grow. Despite the development of intravascular “blood pool” contrast agents, the majority of clinical MRA examinations continue to utilize conventional first-pass Gd contrast agents. However, adequate vessel enhancement frequently requires administration of double or triple doses of these agents. MRA dosing studies with gadobenate dimeglumine have suggested that imaging accuracy is highest with a dose of 0.1 mmol/kg gadobenate dimeglumine, compared with lower (0.025, 0.05 mmol/kg) or even with higher doses (0.2 mmol/kg) [52]. Because of its increased r1 relaxivity in blood, gadobenate dimeglumine is well-suited for this application (Figures 6-8).
Early phase I studies showed that signal intensity peaks and the area under the enhancement curve are higher and of longer duration with gadobenate dimeglumine than with gadopentetate dimeglumine [26]. Consistent with these findings, improved performance with gadobenate dimeglumine has been noted in intraindividual MRA studies in the carotid, abdominal, renal, pelvic, and peripheral arteries [26-30]. Several intraindividual comparisons of gadobenate dimeglumine with gadopentetate dimeglumine have revealed superior arterial contrast enhancement and vessel conspicuity with gadobenate dimeglumine, particularly in smaller vessels [26-30]. The improved conspicuity of smaller vessels may be a reflection of the greater relative signal intensity enhancement at lower concentrations of gadobenate dimeglumine, as might be expected in smaller vessels and more distal peripheral vasculature [20,53]. Moreover, intraindividual comparisons between 0.1 mmol/kg gadobenate dimeglumine and 0.2 mmol/kg gadopentetate dimeglumine have demonstrated equal [29] or better [30] quality MRA images with the lower, standard dose of gadobenate dimeglumine (Figures 9,10).
In two Phase III clinical trials evaluating gadobenate dimeglumine for MRA of the iliofemoral [54] and renal [55] arteries, the sensitivity, specificity, and accuracy values for the detection of significant stenoocclusive disease were similar for the two vascular territories, ranging between 54–81%, 90–95%, and 85–88%, respectively, for the detection of significant iliofemoral disease [54] and between 60%–84%, 89%–95%, and 80%–87%, respectively, for the detection of significant renal disease [55], relative to digital subtraction angiography (DSA) as the reference standard.
Overall, the results of the various clinical studies for MRA suggest advantages for 0.1 mmol/kg gadobenate dimeglumine in terms of performance, cost, and, potentially, patient safety. The future of MRA is likely to benefit from improvements in hardware and acquisition sequences. It is likely the increased r1 relaxivity will prove increasingly beneficial for MRA protocols for which maximal achievable signal is of fundamental importance.
DYNAMIC MR OF THE BREAST
The goal of breast imaging is to accurately define the presence, type, and extent of disease, and subsequently to ensure patients receive the appropriate treatment, ie, mastectomy or breast-conserving surgery (lumpectomy, quadrantectomy), with or without preoperative neoadjuvant chemotherapy. Although conventional mammography and ultrasound are currently the most widely used techniques for breast cancer screening, contrast-enhanced MR has shown considerable clinical utility, particularly in women with dense breasts, and is recommended by the American Cancer Society for imaging women with an inherited high risk of breast malignancy [56,57].
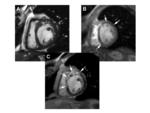
Gadobenate dimeglumine significantly improves the detection and characterization of breast lesions [31,58,59] (Figures 11,12). Dosing studies indicate that the sensitivity for lesion detection is comparable for 0.1 and 0.2 mmol/kg gadobenate dimeglumine, with the highest specificity obtained with a 0.1 mmol/kg dose [58]. In an intraindividual crossover study of 26 women, gadobenate dimeglumine depicted significantly (p=0.003) more lesions compared to gadopentetate dimeglumine, and detected lesions were significantly (p<0.001) more conspicuous with gadobenate dimeglumine [31]. Overall, the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and overall accuracy for identification of malignant lesions were higher with gadobenate dimeglumine (94.7%, 100%, 100%, 80.0%, and 95.6%) than with gadopentetate dimeglumine (76.3%, 100%, 100%, 47.1%, and 80.4%).
More recently, gadobenate dimeglumine has demonstrated notable utility for evaluation of the contralateral breast in patients with confirmed lesions in one breast [60]. Such patients are known to have up to a five times higher risk compared to women in the general population [61]. In 118 patients with unilateral breast cancer and negative findings on physical examination, conventional mammography, and ultrasound, gadobenate dimeglumine-enhanced MR imaging revealed contralateral lesions in 28 (24%) patients [60] of which 24 were detected in patients with dense breasts. In this study, the sensitivity, specificity, accuracy, PPV, and NPV for depiction of malignant contralateral lesions were 100%, 94%, 95%, 79%, and 100%, respectively.
Vascular asymmetry has also been shown to be associated with ipsilateral invasive breast cancer with a high degree of sensitivity (88%) and specificity (82%). Therefore, sensitivity for malignancy may be improved by incorporating vascular mapping into the examination. A comparative study between gadobenate dimeglumine and gadopentetate dimeglumine for the evaluation of breast vascularity demonstrated that a gadobenate dimeglumine dose of just 0.05 mmol/kg is sufficient for the evaluation of one-side increased breast vascularity associated with ipsilateral invasive breast cancer, and that a dose of 0.1 mmol/kg is significantly superior to a dose of 0.1 mmol/kg gadopentetate dimeglumine [59].
LIVER MRI
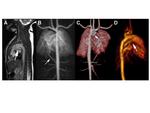
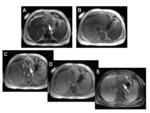
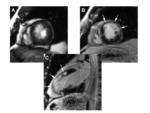
Despite the development of liver-specific contrast agents based on iron oxide particles or manganese, Gd-based MR contrast agents remain the most widely utilized for the majority of liver imaging studies [62]. Unenhanced T1w and T2w imaging, followed by T1w imaging during the immediate arterial, portal-venous, and equilibrium phases of contrast enhancement after administration of an extracellular Gd contrast agent remains the standard imaging protocol for MR evaluation of the liver. The unique pharmacokinetic properties of gadobenate dimeglumine (ie, uptake by functioning hepatocytes and partial elimination through the hepatobiliary system) allow it to be used both for dynamic imaging of the liver in an identical manner to that of the conventional agents, and for delayed liver imaging when liver-to-lesion contrast is based on the differential functional uptake of gadobenate dimeglumine [4,63-66]. (Figures 13-15).
In an intraindividual crossover comparison of 0.05 mmol/kg gadobenate dimeglumine with 0.1 mmol/kg gadopentetate dimeglumine, Schneider et al demonstrated equivalence between the two study groups for dynamic liver imaging despite the lower dose of gadobenate dimeglumine, and superiority of gadobenate dimeglumine for delayed imaging [33]. Greater lesion-to-liver contrast was noted for more patients on delayed images after gadobenate dimeglumine, and more correct diagnoses of histologically-confirmed lesions were made with the complete gadobenate dimeglumine image set than the complete gadopentetate dimeglumine image set [33].
Numerous studies have highlighted the value of delayed imaging with gadobenate dimeglumine [64,66-71]. Not only does delayed hepatobiliary phase imaging with gadobenate dimeglumine benefit the detection of metastatic lesions [71] and the accurate identification of hypervascular malignant lesions [62,70], it has also proven invaluable for the improved characterization of equivocal or atypical lesions on conventional dynamic imaging, particularly focal nodular hyperplasia (FNH) [64,70]. A specific and unique attribute of gadobenate dimeglumine for hepatobiliary phase imaging is in the accurate differentiation of FNH from hepatic adenoma (HA) [66]. Whereas both lesions typically present similar enhancement features on dynamic phase imaging, FNH invariably appears iso- or hyperintense on delayed phase images after gadobenate dimeglumine (reflecting the presence of a functioning albeit malformed biliary system) while HA always appears hypointense (due to the complete absence of biliary excretion).
The additional diagnostic information on delayed imaging, combined with the possibility of using a lower overall dose to obtain similar diagnostic information on dynamic imaging, offers a distinct clinical advantage for gadobenate dimeglumine-enhanced liver MR imaging. Importantly, gadobenate dimeglumine displays a safety profile comparable to the conventional Gd contrast agents in patients with impaired renal function or hepatic function [3,73,74].
CARDIAC MR
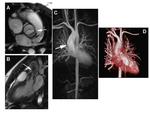
Technical advances have markedly improved the clinical utility of contrast-enhanced MR of the heart [75]. Perfusion imaging for cardiac functionality, as well as evaluation of delayed hyperenhancement (10 to 20 minutes postcontrast injection) for identification of infarcted areas and assessment of myocardial viability and potential for reperfusion, are today standard MR applications [75-77]. As with other applications, the greater r1 relaxivity of gadobenate dimeglumine has the potential to benefit all cardiac MR applications (Figures 16,17).
In a study of 24 patients with acute MI, improved signal intensity and CNR was achieved following administration of a 0.05 mmol/kg dose of gadobenate dimeglumine compared to a 0.1 mmol/kg dose [78]. Dynamic contrast-enhanced MR with 0.05 mmol/kg gadobenate dimeglumine in a larger cohort of 103 patients with acute MI demonstrated that this dose was well-tolerated and efficacious, with high sensitivity (98%) and specificity (72%) for demonstrating infarct [79]. A more recent intraindividual comparison in which patients received two cardiac MR examinations within 2 days, one with 0.05 mmol/kg gadobenate dimeglumine and the other with 0.1 mmol/kg gadopentetate dimeglumine, was performed in 23 patients with clinically proven MI [32]. No statistically significant difference in CNR was found in the 61 infarcts assessed, suggesting equivalent efficacy between the double dose of gadopentetate dimeglumine and the single dose of gadobenate dimeglumine [32]. Lower doses seem to afford at least equivalent efficacy, with the potential for cost savings and enhanced safety in this patient population.
SAFETY
Adverse Events - Just 413 (14%) of 3,092 subjects who received gadobenate dimeglumine in 79 clinical trials reported one or more adverse events (AE) which were considered definitely or potentially related to gadobenate dimeglumine [3]. This incidence was similar to that observed with placebo (17%) or with active controls (8%). No increase in the AE rate was observed in children, and no worsening of renal or liver function was observed in those with hepatic or renal impairment. Moreover, a retrospective analysis of ECG parameters in more than 1000 patients and healthy volunteers revealed no detrimental effect on cardiac electrophysiology. The lack of an effect on cardiac electrophysiology confirmed earlier findings from a dedicated intraindividual crossover comparison with placebo [80]. The favorable safety profile of gadobenate dimeglumine was confirmed by postmarketing safety surveillance data in which an AE reporting rate of 0.05% was observed after more than 1.5 million administrations of gadobenate dimeglumine.
A subsequent analysis of AE rates in subjects who received both gadobenate dimeglumine and gadopentetate dimeglumine revealed no difference between the agents [81]. Among patients enrolled in intraindividual crossover studies the AE rate was 8.0% for gadobenate dimeglumine and 8.5% for gadopentetate dimeglumine (p=0.84). The findings for other safety parameters, including laboratory tests and vital signs, were also similar for the two agents.
Nephrogenic Systemic Fibrosis - Patients with renal impairment undergoing contrast-enhanced MR imaging require special consideration in light of recent findings related to nephrogenic systemic fibrosis (NSF). Administration of Gd contrast agents has been implicated as a risk factor in the development of NSF [82-84]. Furthermore, repeated or higher than recommended doses of Gd contrast agent, as well as a greater degree of renal impairment at the time of Gd exposure, seem to increase the risk of development of NSF. The true prevalence of this condition is unknown; however, in one retrospective study of 370 patients with severe renal insufficiency receiving gadodiamide, the estimated risk for development of NSF was 4% [85].
Postmarketing reports have identified NSF following both single and multiple administrations of Gd contrast agents. In these reports, the specific agent was not always identified; however, when a specific agent was identified, the most common agent was gadodiamide followed by gadopentetate dimeglumine then gadoversetamide. NSF has also developed following sequential administrations of gadobenate dimeglumine or gadoteridol followed by gadodiamide [86].
Currently, guidelines for minimizing the risk of developing NSF in patients with reduced renal function for which contrast-enhanced MR imaging is indicated [87,88] recommend caution when administering Gd contrast agents in patients at increased risk, ie, those with moderate-to-severe renal dysfunction.

![Fig. 2: Commonly Used Gadolinium Contrast Agents and Their Physicochemical Properties [5]](https://epos.myesr.org/posterimage/esr/ecr2008/28780/media/230482?maxheight=150&maxwidth=150)