General features:
-Most common invasive gynecologic malignancy
-May appear as polypoid endometrial mass,
or diffuse endometrial thickening with or without myometrial invasion
-Most common symptom is abnormal genital bleeding
Prognostic factors:
-Histologic tumor grade
-Staging (especially,
Depth of myometrial invasion)
-Lymph node involvement
Therapeutic approach:
-Primary surgical treatment
-From conservative,
minimally invasive therapy: hormonal therapy,
to aggressive,
multidisciplinary therapy depending on tumor stage,
and patient’s age
Classification:
Classified into two pathogenic groups: Type I and Type II,
which show different clinical behaviors,
imaging manifestations,
and therapeutic outcomes (Table.
1)

Table 1: What are Type I and Type II tumors?
Type I tumors are low-grade,
affect younger women,
associate with hyperestrogenism,
or with endometrial hyperplasia,
whereas Type II tumors are high-grade,
occur in postmenopausal,
have aggressive behavior,
may arise from atrophic endometrium or endometrial polyp.
Typical Type I tumor (Fig.
1)
Typical Type II tumor (Fig.
2)
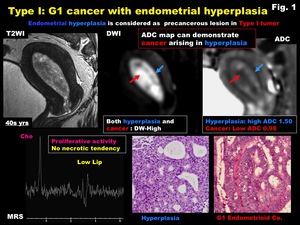
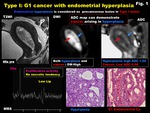
Fig. 1: Type I: G1 cancer with endometrial hyperplasia
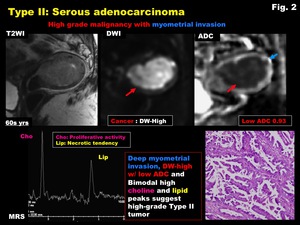
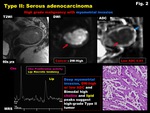
Fig. 2: Type II: Serous adenocarcinoma
Diagnosis and staging of endometrial carcinoma
-Endometrial carcinoma is usually diagnosed by endometrial biopsy,
and dilation and curettage (D&C) is performed when endometrial biopsy does not yield sufficient diagnostic material.
-Endometrial carcinoma is surgically staged using the FIGO cancer staging system (Table.
2).
Carcinosarcoma is classified as uterine sarcoma in WHO classification (2003),
however,
is recently considered to be epithelial origin and staged according to the classification of endometrial carcinoma.

Table 2: Staging: FIGO 2008
MR imaging manifestations:
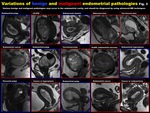
Various benign and malignant pathologies may occur in the endometrial cavity (Fig.
3),
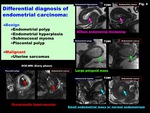
and both benign and malignant tumors may mimic endometrial carcinoma on MR imaging (Fig.
4).
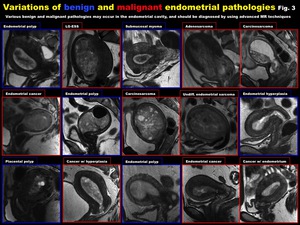
Fig. 3: Variations of benign and malignant endometrial pathologies
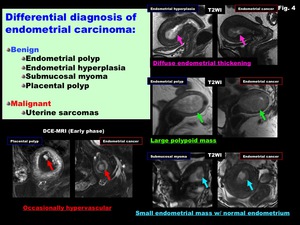
Fig. 4: Differential diagnosis of endometrial carcinoma
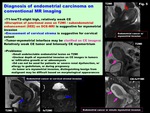
Diagnosis on conventional MR imaging (Fig.
5)
-T1-low/T2-slight high,
relatively weak CE
-Disruption of junctional zone on T2WI,
and Subendometrial enhancement (SEE) on DCE-MRI are suggestive for myometrial invasion
-Encasement of cervical stroma is suggestive for cervical extent
-Tumor-myometrial interface may be clarified on CE images: Relatively weak CE tumor and Intensely CE myomertrium
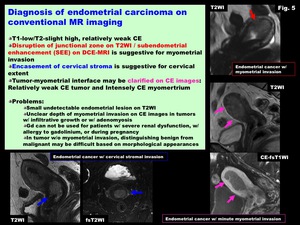
Fig. 5: Diagnosis of endometrial carcinoma on conventional MR imaging
However,
some problems may be encountered in evaluating endometrial carcinoma on conventional MR imaging such as: 1) Small undetectable endometrial lesion on T2WI,
2) Unclear depth of myometrial invasion on CE images in tumors with infiltrative growth or with adenomyosis,
3) Gd can not be used for patients with severe renal dysfunction,
with allergy to gadolinium,
or during pregnancy,
4) In tumor without myometrial invasion,
distinguishing benign from malignant may be difficult based on morphological appearances,
and endometrial carcinoma should be diagnosed by using advanced MR techniques.
Diagnosis by using advanced MR techniques
Advanced MR techniques is helpful as “problem-solving” MR techniques in diagnosing endometrial carcinoma
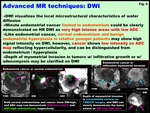
-Diffusion-weighted imaging (DWI) with ADC measurement
DWI visualizes the local microstructural characteristics of water diffusion.
In oncologic imaging,
various malignant tumors may show high signal intensity on DWI due to their high cellularity and long T2 relaxation time.
Apparent diffusion coefficient (ADC) measurement yields quantitative information regarding tissue structure based on the molecular motion of water.
Malignant lesions with increased cellularity show low ADC values,
whereas relative hypocellular benign pathologies and normal structures tend to show higher ADC values (Fig.
6,
7).
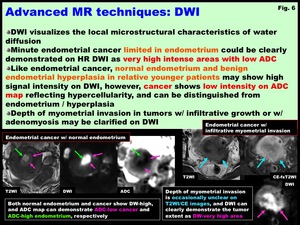
Fig. 6: Advanced MR techniques: DWI
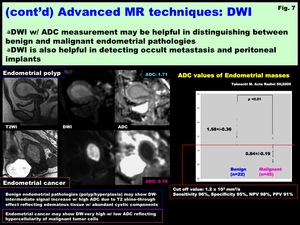
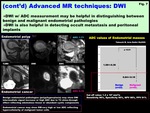
Fig. 7: (cont’d) Advanced MR techniques: DWI
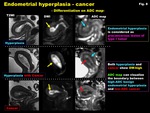
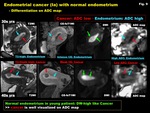
Like endometrial carcinoma,
normal endometrium and benign endometrial hyperplasia in relative younger patients may show high signal intensity on DWI,
however,
cancer shows low intensity on ADC map reflecting hypercellularity,
and can be distinguished from normal endometrium and endometrial hyperplasia (Fig.
8,
9).
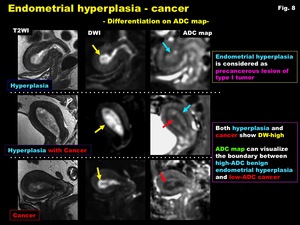
Fig. 8: Endometrial hyperplasia - cancer
-Differentiation on ADC map-
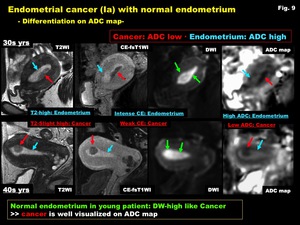
Fig. 9: Endometrial cancer (Ia) with normal endometrium
-Differentiation on ADC map-
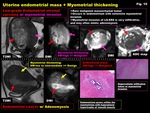
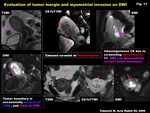
Depth of myometrial invasion in tumors with infiltrative growth or with adenomyosis may be clarified on DWI (Fig.
10,
11).
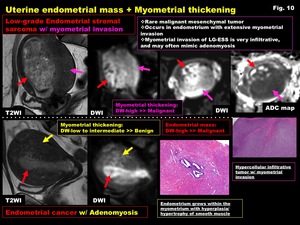
Fig. 10: Uterine endometrial mass + Myometrial thickening
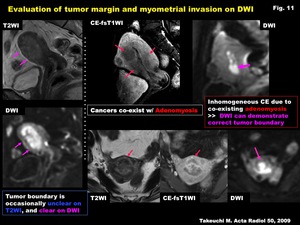
Fig. 11: Evaluation of tumor margin and myometrial invasion on DWI
DWI with ADC measurement may be helpful in distinguishing between benign and malignant endometrial pathologies (Fig.
7)
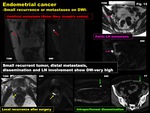
DWI is also helpful in detecting occult metastasis and peritoneal implants (Fig.
12)
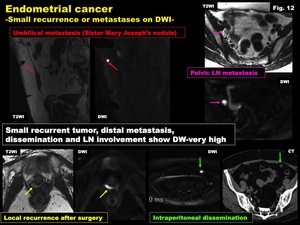
Fig. 12: Endometrial cancer
-Small recurrence or metastases on DWI-
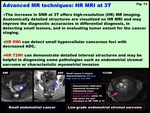
-High-resolution (HR) MRI at 3T
The increase in SNR at 3T offers high-resolution (HR) MR imaging.
Anatomically detailed structures are visualized on HR MRI and may improve the diagnostic accuracies in differential diagnosis,
in detecting small lesions,
and in evaluating tumor extent for the cancer staging.
HR T2WI can demonstrate detailed internal structures and may be helpful in diagnosing some pathologies such as endometrial stromal sarcoma with characteristic myometrial invasion (Fig.
13)
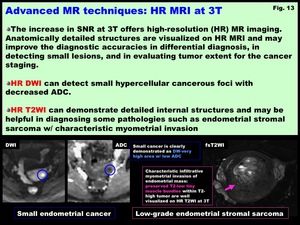
Fig. 13: Advanced MR techniques: HR MRI at 3T
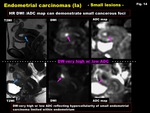
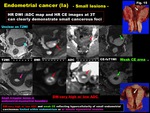
HR DWI can detect small hypercellular cancerous foci with decreased ADC.
Minute endometrial carcinoma limited in endometrium could be clearly demonstrated on high resolution (HR) DWI as very high intense areas with low ADC (Fig.
14,
15).
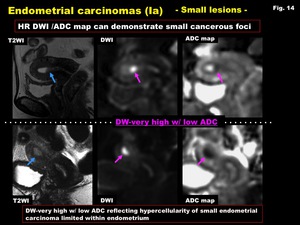
Fig. 14: Endometrial carcinomas (Ia)
-Small lesions-
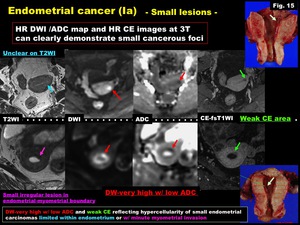
Fig. 15: Endometrial cancer (Ia)
-Small lesions-
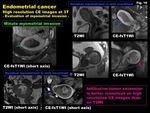
HR CE images are helpful in demonstrating minute myometrial invasion,
or infiltrative tumor extension of endometrial carcinomas.
Residual myometrium may be better visualized on HR CE images than on T2WI (Fig.
16).
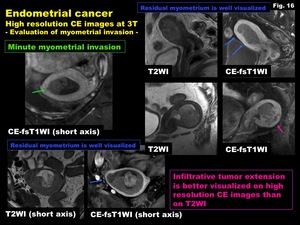
Fig. 16: Endometrial cancer
High resolution CE images at 3T
-Evaluation of myometrial invasion-
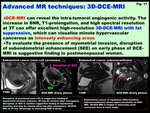
-3D-Dynamic contrast-enhanced imaging (DCE-MRI)
DCE-MRI is performed with intravenous administration of contrast medium
Two types of DCE-MRI are in clinical use.
Dynamic multiphase contrast-enhanced MRI for the assessment of vascular characteristics of tumors,
and perfusion imaging for the assessment of tumor microenvironment: perfusion and permeability indices
The term “DCE-MRI” usually means the former method,
by using 3D gradient echo T1WI,
images are acquired at different time intervals after intravenous bolus injection of Gd at multiple phases of dynamic contrast medium enhancement.
The arterial phase acquisition provides qualitative tissue contrast deriving from the vascularity differences of tissues.
The arterial phase images can improve the diagnostic capability in tumor detection,
tumor staging (especially in evaluating the depth of myometrial invasion by endometrial carcinoma),
and the tumor characterization.
DCE-MRI can reveal the intra-tumoral angiogenic activity.
The increase in SNR,
T1-prolongation,
and high spectral resolution at 3T can offer excellent high-resolution 3D-DCE-MRI with fat suppression,
which can visualize minute hypervascular cancerous as intensely enhancing areas.
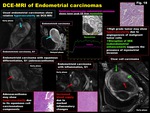
To evaluate the presence of myometrial invasion,
disruption of subendometrial enhancement (SEE) on early phase of DCE-MRI is suggestive finding in postmenopausal women (Fig.
17,
18).
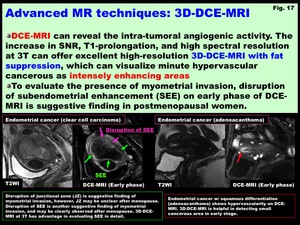
Fig. 17: Advanced MR techniques: 3D-DCE-MRI
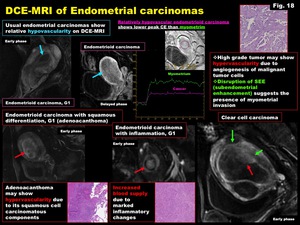
Fig. 18: DCE-MRI of Endometrial carcinomas
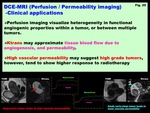
Perfusion / Permeability imaging:
Perfusion can be assessed by semiquantitative method analyzing changes in the signal intensity,
and quantitative method utilizing pharmacokinetic modeling techniques.
Because the parameters provided by signal intensity are relative values,
semiquantitative data are not objective.
In quantitative analysis concentration-time curves are generated by fitting the time-intensity curve of Gd into pharmacokinematic model.
Bolus-injected Gd passes through a capillary bed,
rapidly passes into the extravascular-extracellular space (the leakage space): Ve at a rate determined by the permeability of the microvessels,
and their surface area and blood flow.
The transfer constant (Ktrans) describes the transendothelial transport of Gd by diffusion,
and is mainly influenced by vascular permeability,
the leakage space (Ve) as a percentage of unit volume of tissue,
and the rate constant (Kep=Ktrans/Ve) (Fig.
19)
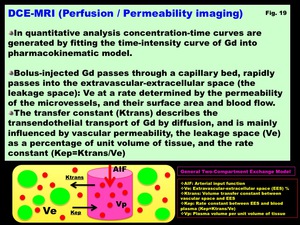
Fig. 19: DCE-MRI (Perfusion / Permeability imaging)
Perfusion imaging provides biomarkers and predictors of response to therapy.
Evaluation of MR perfusion parameters in pre-treatment and early treatment response at 2 weeks in patients with cervical cancer (Harry VN.
Gynecol Oncol 116; 2010),
and ovarian cancer (Sala E.
Radiology 263; 2012) may predict treatment response more readily than that of delayed assessment of changes in tumor size.
Perfusion imaging visualizes heterogeneity in functional angiogenic properties within a tumor (i.e.
cervical cancer with necrosis),
or between multiple tumors (i.e.
multiple peritoneal implants – omental masses of advanced ovarian cancer).
Ktrans may approximate tissue blood flow due to angiogenesis,
and permeability.
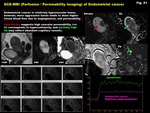
High vascular permeability may suggest high grade tumors,
however,
tend to show higher response to radiotherapy (Fig.
20,
21)
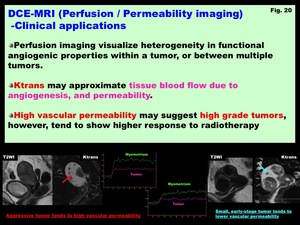
Fig. 20: DCE-MRI (Perfusion / Permeability imaging)
-Clinical applications-
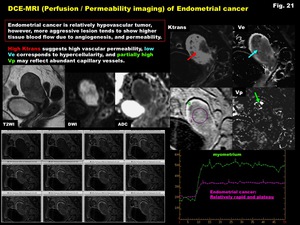
Fig. 21: DCE-MRI (Perfusion / Permeability imaging) of Endometrial cancer
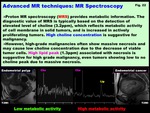
-MR Spectroscopy (MRS)
Proton MR spectroscopy (MRS) provides metabolic information,
which is useful for the differentiation of benign and malignant tumors in the brain,
prostate and other various organs.
The diagnostic value of MRS is typically based on the detection of elevated level of choline,
which is a biomarker of cancer tissue and is increased in actively proliferating tumors.
Choline is used for a biomarker for treatment response,
and is expected as an early predictor for treatment response in patients with gynecologic cancers.
In the female pelvis other metabolites such as creatine,
lipid,
lactate,
N-acetyl compound are useful in making differential diagnosis,
especially in the estimation of histological types of gynecologic tumors.
3T-MRI can offer high-quality MRS because of superior spectral separation and increased signal-to-noise ratio.
Metabolites:
-Choline: Cho (3.2 ppm) peak reflects metabolic activity of cell membrane in solid tumors.
High grade malignant tumors tend to show higher choline peaks (Takeuchi et al.
Eur Radiol 2011).
However,
high-grade malignant tumors with massive necrosis may cause low Cho concentration due to the decrease of viable tumor cells.
-Lipid: Lip (1.3 ppm) peak may arise from triglycerides and cholesterol esters in neutral lipid droplets,
and have been observed in various cancers and are considered to be important biomarker for malignant tumors (Delikatny et al.
NMR Biomed 2011).
Lip peaks may be observed in both viable and necrotic tumor areas resulting from considerable amount of cellular death due to rapid cellular turnover,
and be useful in evaluating heterogeneous mass containing areas of viable tumor cells,
cysts,
necrosis or hemorrhage such as high-grade malignancy (Fig.
22,
23).
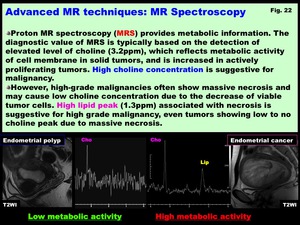
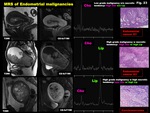
Fig. 22: Advanced MR techniques: MR Spectroscopy
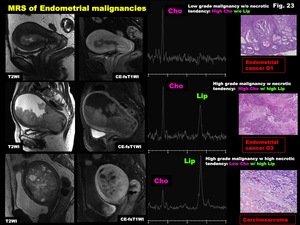
Fig. 23: MRS of Endometrial malignancies
Imaging manifestations of Histological subtypes
Type I tumor vs Type II tumor: (Table.
1)
Type I tumors are low-grade,
affect younger women,
associate with hyperestrogenism,
or with endometrial hyperplasia (Fig.
1),
whereas Type II tumors are high-grade,
occur in postmenopausal,
have aggressive behavior,
may arise from atrophic endometrium or endometrial polyp (Fig.
2)
Type I tumors:
-Low grade endometrioid carcinoma (G1-2) (Fig.
24)
-Type I endometrioid carcinomas are relatively low grade (G1-2),
affect younger women,
estrogen dependent tumor (may be associated with unopposed estrogen exposure or PCO),
arise from endometrial hyperplasia
-Commonly lower stage with no or minute myometrial invasion
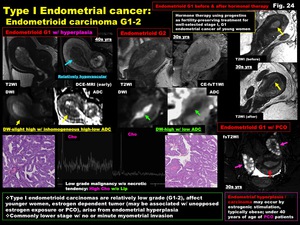
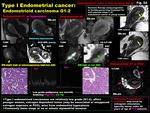
Fig. 24: Type I Endometrial cancer:
Endometrioid carcinoma G1-2
-Mucinous adenocarcinoma (Fig.
25)
-Mucinous adenocarcinoma is uncommon,
relatively low grade
-May contain mucin-pooling dilated cystic glands observed as tiny T2-high cysts in the tumor
-Adenoacanthoma (Fig.
25)
-Endometrioid carcinoma with squamous differentiation contains Adenoacanthoma and Adenosquamous carcinoma
-Adenoacanthoma is relatively low grade tumor but shows hypervascularity like high grade tumor due to its sqamous cell carcinomatous components
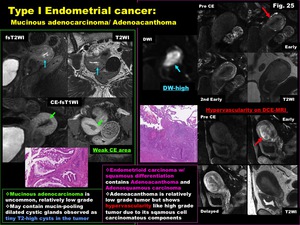
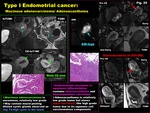
Fig. 25: Type I Endometrial cancer:
Mucinous adenocarcinoma/ Adenoacanthoma
Type II tumors:
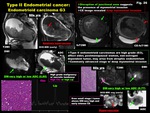
-High grade endometrioid carcinoma (G3) (Fig.
26)
-Type II endometrioid carcinomas are high grade (G3),
affect older,
postmenopausal women,
non-estrogen dependent tumor,
may arise from atrophic endometrium
-Commonly advanced stage with deep myometrial invasion
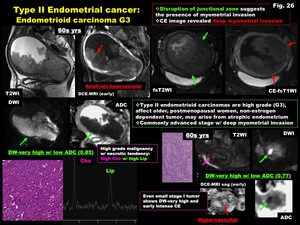
Fig. 26: Type II Endometrial cancer:
Endometrioid carcinoma G3
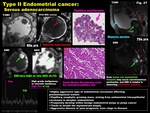
-Serous adenocarcinoma (Fig.
27)
-Highly aggressive type of endometrial carcinoma affecting postmenopausal women
-Papillary,
exophytic growing mass,
arising from endometrial intraepithelial carcinoma in atrophic endometrium
-Frequently develop within benign endometrial polyp as polyp cancer
-Tends to invade the myometrium deeply
-Aggressive disease with poor prognosis,
even stage Ia disease
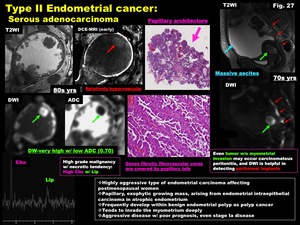
Fig. 27: Type II Endometrial cancer:
Serous adenocarcinoma
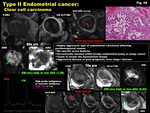
-Clear cell carcinoma (Fig.
28)
-Highly aggressive type of endometrial carcinoma affecting postmenopausal women
-No specific gross features
-Occasionally develop within benign endometrial polyp as polyp cancer
-Tends to invade the myometrium deeply
-Aggressive disease w/ poor prognosis,
even stage I disease
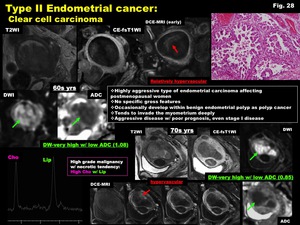
Fig. 28: Type II Endometrial cancer:
Clear cell carcinoma
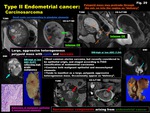
-Carcinosarcoma (Fig.
29)
-Most common uterine sarcoma,
but recently considered to be epithelial origin,
and staged according to FIGO classification of endometrial carcinoma
-Contains both malignant epithelial and mesenchymal elements
-Tends to manifest as a large,
polypoid,
aggressive heterogeneous mass.
Occasionally appear as “delivery”.
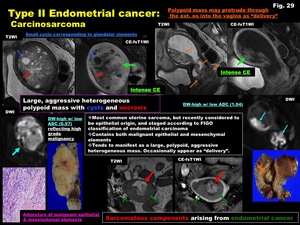
Fig. 29: Type II Endometrial cancer:
Carcinosarcoma
Carcinogenesis of endometrial carcinomas
Precursor of type I tumor:
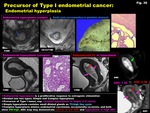
-Endometrial hyperplasia (Fig.
30)
-Endometrial hyperplasia is a proliferative response to estrogenic stimulation
-Divided into two types: Simple and Complex hyperplasia
-Precursor of Type I tumor,
especially complex hyperplasia with atypia (1/4 cases)
Simple hyperplasia contains small dilated glands as T2-high tiny cysts
Complex hyperplasia mimics endometrial carcinoma,
occasionally co-exists,
and both show DW-high.
ADC map may demonstrate cancer with low ADC and hyperplasia with high ADC
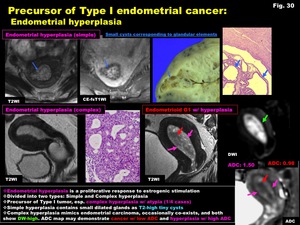
Fig. 30: Precursor of Type I endometrial cancer:
Endometrial hyperplasia
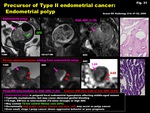
Precursor of type II tumor:
-Endometrial polyp (Fig.
31)
-Endometrial polyp is polypoid focal endometrial hyperplasia affecting middle-aged women
-Typically asymptomatic,
but may cause abnormal genital bleeding
-T2-high,
DW-low to intermediate (T2-shine through) with high ADC
-May contain T2-low central fibrous core (CFC)
-Occasionally high grade cancers (serous and clear cell) may occur as polyp cancer
-Even small,
stage I polyp cancer shows aggressive behavior with poor prognosis
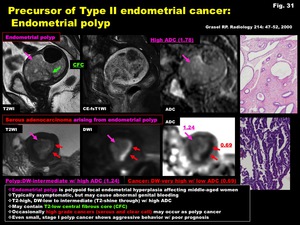
Fig. 31: Precursor of Type II endometrial cancer:
Endometrial polyp
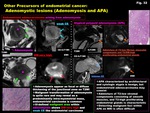
Other Precursors of endometrial carcinoma: Adenomyotic lesions
-Adenomyosis (Fig.
32)
-Adenomyosis appear as focal or diffuse thickening of the junctional zone on T2WI
-Malignant transformation of adenomyosis is quite rare and may reveal as a predominantly intra-myometrial mass,
endometrioid carcinoma is common
-Ill-defined malignant area within adenomyosis shows DW-very high and weak CE like endometrial carcinoma
-Atypical polypoid adenomyoma: APA (Fig.
32)
-APA characterized by architectural and cytologic atypia is benign,
but endometrioid adenocarcinoma may coexist
-Admixture of T2-low stromal components consisting of smooth muscles,
and T2-high proliferating endometrial glands is characteristic
-Detecting malignant foci within APA on MRI is often difficult
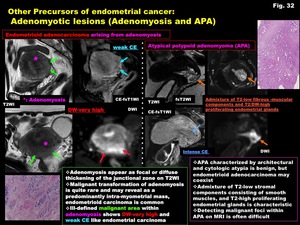
Fig. 32: Other Precursors of endometrial cancer:
Adenomyotic lesions (Adenomyosis and APA)
Take Home Points:
-DWI with ADC measurement and MRS may be helpful in distinguishing benign from malignant endometrial pathologies
-High on DWI with low ADC due to hypercellularity; high Cho and Lip on MRS reflecting metabolic activity and necrosis/apoptosis,
suggest high-grade tumors.
-ADC map can clearly demonstrate low-ADC cancerous area arising in high-ADC precancerous lesions
-3D-DCE-MRI and DWI can demonstrate depth of myometrial invasion correctly even in tumors with infiltrative growth or with co-existing adenomyosis.
-Small endometrial lesion can be demonstrated on HR DWI
-3D-DCE-MRI may demonstrate small hypervascular lesions,
and evaluate SEE with myometrial invasion
-DWI is also helpful in detecting occult metastasis and peritoneal implants