Patient selection
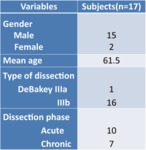
Seventeen consecutive cases with DeBakey type Ⅲ AD that underwent ECG-gated CT angiograms from January 2010 to June 2012 in our institute were enrolled in this evaluation (2 women,
15 men; mean age 61.5 years (range 46 - 77 years)).
The population constists of 2 cases with DeBakey type Ⅲa and 16 cases with DeBakey type Ⅲb. Six cases had a history of open replacements of the aorta (5 with ascending and total arch replacement and 1 with descending aortic replacement )and classification of these cases were based on the post-operative status.
Patient demographics and clinical characteristics are summarized in Table 1.
The extent of the AD was indicated based on “zone classification” ,
which is explained in Fig. 1 .
Retrospective ECG-gated CT
・Image acquisition:
All CT was performed with 32- or 64-detector row CT (Aquilion32 or 64,
Toshiba Medical Systems Company,
Tokyo,
Japan).
The scan parameters adopted are as follows: helical pitch 3.2,
120 kVp,
300mA,
0.5 s rotation time and 1mm collimation.
Under 3 liter/min oxygenation via a nasal tube,
images were acquired after intravenous administration of 100 ml of iodinated contrast medium (Iomeprol 350) at a flow rate of 3 ml/s applying the bolus tracking program implemented on the scanner.
Bolus timing was achieved using an automated triggering technique with a threshold of 200 HU within a region of interest (ROI) placed in the ascending aorta.
Scans were performed from the level of the supraaortic vessels to iliac arteries.
・Image reconstruction and postprocessing
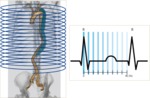
Multiphase image reconstruction was conducted in terms of retrospective ECG–gating,
wherein the images were reconstructed at 10% intervals from 0-90% of the R-R interval (total 10 phases) with a slice thickness of 1mm and 1mm increment.
These 10 volume data sets per patient were transferred to a dedicated workstation for analysis (Fig. 2).
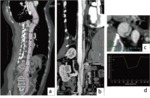
CT Data sets corresponding to the 0% phase of the R-R interval were initially used for semi-automatic centerline extraction of the true lumen of dissected aorta.
CSMPR perpendicular to the centerline and the straightened CPR were then generated (Fig. 3).
After which,
the 3D centerlines were copied to the remaining 9 volume data sets,
and CSMPR and straightened CPR were repeatedly reconstructed according to the centerlines.
Vessel analysis software (AZE,
Tokyo,
Japan) was able to visualize the cyclic motion of the intima either on straightened CPR at any given plane or on CSMPR at any given aortic levels.
Confirmation of the level of entry and re-entry sites was conducted using CSMPR under consensus among two experienced radiologists and one medical student.
Entry or re-entry sites were defined as apparent communication measuingr more than 1mm between the true and false lumen detected in most static phase on CSMPR.
・Quantitative analysis of the true lumen
To assess alteration of the diameter of the true lumen during cardiac cycle,
the semi-automated diameter measurement function on CSMPR of the vessel analysis software was used since this allows the estimation of the mean diameter of the true lumen at given cardiac phase and aortic level.
To assess the rate of the change in diameter of the true lumen during the cardiac cycle,
the ratio of internal diameter change (Rmax) was defined using the following formula,
where Dmax was defined as the maximum value of the diameter during the cardiac cycle and
Dmin was defined as the minimum value of the diameter during the cardiac cycle:
Rmax=(Dmax-Dmin)/Dmax
To assess the kinetics of the intima in the dissection the following parameters were assessed: the average diameter of the true lumen in each cardiac phase calculated at designated levels,
the average of Rmax at each level,
the average of Dmin at each level, and the differences in Rmax between acute (defined as those with less than 30 days from onset)and chronic cases.