Patients
Patients with suspected malignancy of the upper aerodigestive tract or a cancer of unknown primary with cervical lymphadenopathy were scheduled to undergo 18F-FDG-PET computed tomography (PET/CT) for staging and treatment planning and thereafter,
without additional radiopharmaceutical administration,
an integrated simultaneous PET/MRI study.
Patients were retrospectively included in the current study if they fulfilled the following inclusion criteria: (a) if a de-novo or recurring HNSCC of the upper aerodigestive tract was histopathologically proven either by biopsy or by resection within 2 weeks after imaging,
(b) if a histopathological report including a tumour grading was available,
(c) if a dedicated simultaneous PET/MRI of the neck including T1w-DCE and DWI sequences was performed with sufficient image quality not distorted by motion artefacts,
(d) if no diagnostic or therapeutic intervention was performed on the tumour and (e) if a tumour was delineable in the imaging studies.
PET-imaging
Fasting of at least 6 hours prior to the investigation was ensured.
After application of 18F-FDG dependent on body weight and an uptake time of 90 minutes (Lasix was given 5 minutes after admission of the radiotracer) a whole-body PET/CT was performed.
Immediately afterwards,
patients were transferred to the PET/MRI where whole-body and dedicated neck images where acquired without radiotracer reapplication.
Integrated PET/MRI tomograph
All examinations were performed on a combined simultaneous whole-body hybrid PET/MRI scanner (Siemens Biograph mMR; Siemens Healthcare,
Erlangen,
Germany).
Detailed descriptions of the technical aspects were described in previous publications [4,5].
Simultaneous PET/MRI imaging protocol
Patients were placed in supine position with their arms beside the trunk.
PET/MRI was conducted in two steps.
After a whole body PET/MRI,
a dedicated PET/MRI of the neck using a combined head & neck coil was performed,
which included following sequences:
- a PET of the head & neck neck with 10 minutes of acquisition time
- an axial DWI-EPI sequence with b-values of 0 and 800 (TR/TE 8620/73 ms,
slice thickness 4 mm,
voxel size 3.2 x 2.6 x 4.0 mm)
- a dynamic T1-weighted contrast-enhanced sequence during the administration of 0.1 mmol Gadobutrol per kg of bodyweight (Gadovist®,
Bayer Healthcare,
Leverkusen,
Germany) at a rate of 3 ml per second and flushing with 10 ml of normal saline using a power injector.
This T1w-DCE sequence consisted of 40 subsequent scans à 6 seconds (40 slices per scan) with a TR/TE of 2.47/0.97 ms,
a slice thickness of 5 mm,
a flip angle of 8° and a voxel size of 1.2 x 1.0 x 5.0 mm.
Contrast application was started after the fifth scan
- a coronal T2-weighted TIRM
- an axial T2-weighted TSE sequence with fat suppression
- an axial T1-weighted turbo spin echo (TSE) without contrast medium followd by an axial and a coronal fat saturated T1-weighted TSE sequence after contrast application
Image analysis
For all tumours,
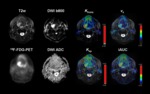
mean and maximum standardized uptake values (SUV) were analyzed in the PET dataset of the neck with a nuclear medicine physician plotting an isocontour VOI around the tumour (SUVmax threshold 40%).
T1w-DCE images were processed with a commercially available software module for tissue perfusion estimation (Tissue 4D,
Siemens Medical Systems,
Erlangen,
Germany) as described previously [6].
The pharmacokinetic parameters Ktrans,
kep,
ve and iAUC were calculated – scaling the arterial input function (AIF) in relation to the gadolinium dose and modelling it by a bi-exponential function – using the two-compartment model by Tofts and Kermode [7].
For each patient,
these four parameter maps were projected onto the T2-weighted fat-suppressed TSE sequences and the tumour was manually delineated on each slice,
resulting in mean values of Ktrans,
kep,
ve and iAUC averaged over the complete tumour.
DWI images were transferred to a desktop computer with Mac OS X (Apple,
Cupertino,
California,
USA) and an open-source freeware 4D DICOM viewer (OsiriX,
Pixmeo,
Geneva Switzerland).
ROI’s were manually drawn on the ADC maps along the contours of the tumour on each slice in conjunction with the complete MRI and PET datasets; mean and minimal ADC values (ADCmean and ADCmin) were then averaged for the whole tumour volume.
For an example see figure 1.
Statistical analysis
Statistical analysis and graphics creation was performed with SPSS 20 (IBM SPSS Statistics,
Armonk,
New York,
USA).
Values are presented as mean ± standard deviation (SD).
Mean value comparison was carried out using the Mann-Whitney-U test.
Spearman’s non-parametric rank sum correlation coefficients were calculated between DCE parameters,
SUVmax,
SUVmean,
ADCmean and ADCmin.
0 - 0.2 was defined as poor,
0.2 - 0.4 as weak,
0.4 - 0.6 as moderate,
0.6 - 0.8 as strong and 0.8 - 1.0 as very strong correlation.
Significance level was set at p ≤ 0.05.