Endorectal ultrasound (EUS) imaging
Technique
EUS is an good technique for visualizing in vivo a suspicious lesion,
to take biopsies,
and to assess he rectal wall and mesorectal tissue.
Flexible probe allows to evaluate up to 30 cm from anal sphincter,
and to assess iliac lymph nodes.
If a rigid probe is used,
only 8-10 cm can be evaluated.
The most widely used EUS endoscopes are oblique forward viewing echo-endoscopes.
They allow a 360° image on a plane perpendicular to the long axis of the insertion of the endoscopic tube.
Frequencies commonly used range from 7.5 to 10 MHz.
Preparation for the EUS includes voiding the rectum (using enema or an oral lavage preparation).
The anesthetic preparation varies depending on the institution: propofol,
midazolam and even sedative agents are used.
Any suspicious lesion is targeted to determine the depth of infiltration into or through the rectal wall.
Normal anatomy in EUS
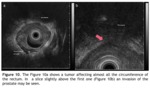
The bowel wall is depicted in five sonographic layers as a result of differences in acoustic impedance (11).
See Figure 5,
which shows an example of normal rectal wall anatomy in EUS.
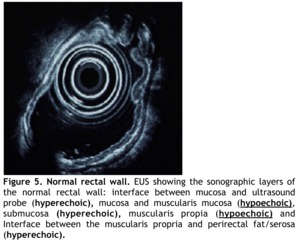
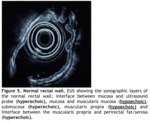
Fig. 5: Figure 5. Normal rectal wall. EUS showing the sonographic layers of the normal rectal wall: interface between mucosa and ultrasound probe (hyperechoic), mucosa and muscularis mucosa (hypoechoic), submucosa (hyperechoic), muscularis propia (hypoechoic) and Interface between the muscularis propria and perirectal fat/serosa (hyperechoic).
References: ECR 2012 / C-1434 Diagnostic management of rectal cancer: MR vs EUS, 10.1594/ecr2012/C-1434 Manuel Gutiérrez Gimeno. Department of Radiology, Hospital Sant Pau, Barcelona, Spain.
Beginning with the lumen,
the first layer is hyperechoic and represents the interface between mucosa and ultrasound probe.
The second layer is hypoechoic,
formed by the mucosa and muscularis mucosa.
The third layer is hyperechoic submucosa.
The fourth is muscularis propia,
a hypoechoic layer.
The fifth layer is hyperechoic and shows the interface between the muscularis propia and perirectal fat.
The anal canal consists of two muscular cylinders (12),
an inner cylinder,
the internal anal sphincter,
and an outer cylinder ,
the external anal sphincter.
By endoscopic ultrasound,
the inner cylinder is represented by an inner hypoechoic ring of tissue and the outer cylinder by a hyperechoic ring (13).
Rectal carcinoma usually appears as a hypoechoic lesion that interrupts the normal five-layer sonographic structure of the rectal wall.
In some cases,
however,
it can appear as a hyperechoic image (12).
High-resolution probes allow marked differentiation between all layers of the rectal wall,
including mucosa and submucosa,
even an in situ carcinoma (Tis) can be depicted.
The main features of the different T stages are:
-
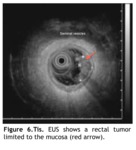
Tis: an exophytic lesion grows towards the lumen but only invades the mucosa (Fig.
6)
-
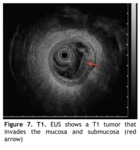
T1 tumor is similar but the lesion invades the submucosa (Fig.
7)
-
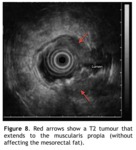
T2: full wall thickness invasion without mesorectal fat invasion (Fig.
8)
-
T3: full wall thickness invasion without mesorectal fat invasion (Fig.
9)
-
T4: invasion of perirectal organs or structures (Fig.
10)
See sidebar for figures.
Despite the high resolution,
EUS has limitations:
-
Differentiating between Tis and T1 is difficult and not always is possible.
-
Misinterpretation of T2 and T3 stages is possible due to:
-
Overstaging a T2 tumor as T3 because the desmoplastic or inflammatory reaction surrounding the tumor is sonographically indistinguishable from malignant tissue (14).
-
Understaging a T3 tumor as T2 due to the impossibility to detect microscopic cancer infiltration (12).
-
No possibility to assess the invasion of the mesorectal fascia.
The accepted morphologic characteristics suggestive of malignant lymph node involvement are:
- node size >5 mm,
- hypoechoic appearance,
- round rather than ovoid or flat,
- peritumoral location (15,16).
Radiotherapy and chemotherapy diminish accuracy in assessing local invasion because post-radiation edema,
inflammation,
fibrosis,
and necrosis alter the normal echogenicity of the rectal wall (18).
Magnetic Resonance Imaging
Technique
Using phased array coils in magnetic resonance imaging,
we can assess the tumour,
the depth of invasion into rectal wall and perirectal fat,
the involvement of mesorectal fascia,
and the involvement or not of the sphincter apparatus.
Rectal MR imaging is based on T2-weighted images.
T1 sequences can provide images of high spatial resolution but they are not good at differentiating tumour and layers of the bowel wall.
Furthermore,
the addition of gadolinium does not seem to improve the diagnostic accuracy for prediction of tumour penetration through the rectal wall or tumour involvement of the mesorectal fascia (19).
The use of bowel preparation,
air insufflation and filling contrast agents varies from centre to centre.
A standard MR protocol should consist of four basic steps,
based in T2-weighted sequences:
-
an axial 5 mm thick T2 weighted fast spin echo (T2W-FSE) sequence of the entire pelvis to localize the primary tumour,
the direction of the rectal wall and possible sites of nodal involvement in the pelvis.
-
three high resolution 3mm thick T2W-FSE sequences (sagittal,
axial and coronal) oriented in an orthogonal plane to the tumour.
T2WI oriented orthogonal to the tumour plane are the best sequences evaluating the deep extension of the tumour.
This protocol allows a thorough evaluation of the extent of the tumour and involvement of lymph nodes (20).
The use of diffusion-weighted imaging (DWI) has now become and standard technique.
This sequence has been included in the most recent guidelines of rectal cancer management (10).
Shortly,
these sequences are based on detecting restriction of the free movement (diffusion) of water protons in tissues.
The sensitivity of the DWI sequence to water motion can be varied by changing the parameter known as the “b value,” which is related with the intrinsic features of the sequence (that will not be discussed here).
Water molecules with a large degree of motion will show signal attenuation with small b values (b = 50–100 s/mm2).
By contrast,
large b values (b = 1,000 s/mm2) are required to attenuate the signal of slow-moving water molecules (20).
DWI has shown to provide additional information on tumour tissue characterisation – hence,
malignancy; it appears to be useful in prediction of treatment (22,23).
In case of N staging,
DWI sequences may aid in detection of lymph nodes,
however,
there is not yet enough evidence supporting the use of this sequence alone in determining whether the lymph nodes are affected (24).
Normal anatomy on MRI
Anatomical features of the rectum and perirectal tissue are easily evaluated using T2-weighted images.
We can distinguish an inner hyperintense layer formed by mucosa and submucosa surrounded by a hypointense layer that represents the muscularis propria.
The muscularis propria is surrounded by the perirectal fat tissue,
which is seen as hyperintense.
The mesorectal fascia is identified as a thin,
low-signal intensity structure that envelops perirectal fat (25).
The anal canal is seen as a cylindrical structure that extends from the insertion of the levator ani muscle in the rectum to the external anal margin.
Images obtained in the coronal plane are specially useful in evaluating sphincter apparatus,
such as for distinguishing between the external sphincter group and puborectal muscle (26).
Rectal cancer on MRI
On MR imaging,
the intensity of a rectal tumour is intermediate in relation to the high signal intensity of the fat tissue and the low signal intensity of the muscular layer (25).
The anterior wall of the upper portion of the high rectum is not surrounded by mesorectal fascia but by the peritoneal reflection.
This is an important point because a neoplasm penetrating the peritoneal reflection is considered a T4 tumour.
A high rectal tumour that invades the perirectal fat above the peritoneal reflection it is also considered as a T4 (27).
In case of low rectum tumours,
their proximity to the sphincter apparatus makes sphincter-sparing surgery not always possible.
Due to the importance of the decision of the type of surgery the radiologist should be meticulous in reporting the exact height of the tumours,
taking as reference the anal verge and puborectal muscle (28).
It is difficult to differentiate stage T1 from stage T2 by MRI due to limited spatial resolution.
Theoretically,
in a T2 tumour the intermediate hypointense layer that represents the muscularis propria is blurred or reduced in thickness.
There is a loss of the interface between the muscularis propria and submucosa while the outer border between the muscularis and the perirectal fat remains intact.
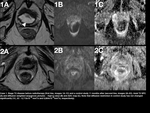
See Figure 11,
where a case of T2 tumour is depicted.
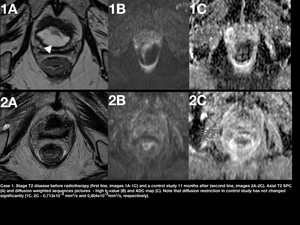
Fig. 11: Figure 11. Case 1. Stage T2 disease before radiotherapy (first line, images 1A-1C) and a control study 11 months after (second line, images 2A-2C). Axial T2 SPC (A) and diffusion weighted sequences pictures - high b-value (B) and ADC map (C). Note that diffusion restriction in control study has not changed significantly (1C, 2C - 0,713x10-3 mm2/s and 0,804x10-3mm2/s, respectively).
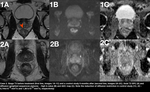
In case of stage T3 tumour (Figure 12),
the muscularis propria is totally disrupted,
with tumour signal extending into the perirectal fat.
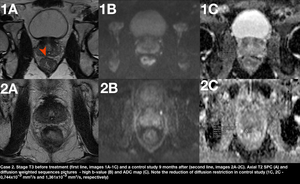
Fig. 12: Figure 12. Case 2. Stage T3 before treatment (first line, images 1A-1C) and a control study 9 months after (second line, images 2A-2C). Axial T2 SPC (A) and diffusion weighted sequences pictures - high b-value (B) and ADC map (C). Note the reduction of diffusion restriction in control study (1C, 2C - 0,744x10-3 mm2/s and 1,361x10-3 mm2/s, respectively). The patient subsequently was reported to be in complete remission.
MR imaging has a special relevance in the study of stage T3 tumours.
As stated previously,
involvement of mesorectal fascia is one of the most important risk factors for local recurrence.
The MERCURY study (29) demonstrated the correlation between MR imaging and histopathology analysis in the assessment of extramural depth of tumour invasion.
MR imaging can therefore be used to assess a positive or negative CRM.
This is extremely important when determining the most appropriate treatment for each patient.
The drawbacks of MR include over-staging of T2 tumour as T3 due to a desmoplastic reaction generated in adjacent normal tissue,
seen as spiculations in the perirectal fat.
Under-staging of T3 tumours as T2 tumours due to microscopic infiltration of perirectal fat is also a possibility.
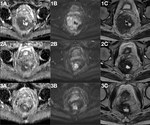
Stage T4 tumours (Figure 13) are characterized by involvement of adjacent organs or perforation of the peritoneum.
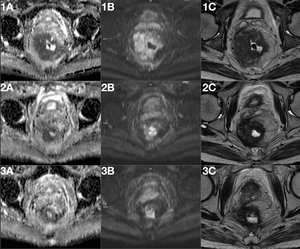
Fig. 13: Figure 13. Case 3. Stage T4 rectal cancer with a partial response to treatment. Baseline imaging (images 1A-1C), during treatment (5 months after baseline, images 2A-2C), after treatment (8 months after baseline, images 3A-3C). Note the diffusion restriction changes in the diffusion weighted images (ADC map, images 1A-3A, high b-value images 1B-3B) and reduction of tumour size in T2 weighted images (images 1C-3C).
Evaluation MR of lymph nodes is difficult and must be interpreted with caution.
Accepted morphologic features suggestive of nodal involvement are a nodal size >5mm and nodes with irregular borders,
mixed signal intensity,
or both (28,30,31).
The accuracy of nodal size as single element of decision is limited because the existence of metastases in nodes less than 5mm is frequent,
up to 30-50% (32).
Table 2 summarizes the main limitations of EUS and MR.

Table 2: Table 2. Summary of the main limitations of EUS and MR.
References: Manuel Gutiérrez Gimeno. Department of Radiology, Hospital General de Catalunya, Barcelona, Spain.
Other techniques
Computed tomography
Abdominal CT has a limited role in the local staging of rectal cancer and its paper is manly the evaluation of distant metastases.
However it still has an important function as a diagnostic tool in patients with a variety of gastrointestinal symptoms.
The accuracy of CT to assess local spreading is variable,
being higher in advanced disease.
The sensitivity is between 80%-95% in patients with advanced disease (32) but decreases considerably when all the T stages are included into the analysis (33).
Both,
CT and MRI have proved good accuracy in the evaluation of local disease so CT is not used as a staging technique.
Nevertheless,
it may be the diagnostic tool in patients with nonspecific symptoms (such as constipation) or in acutely ill patients with an acute complication (obstruction,
perforation).
The CT appearance of rectal carcinoma varies from a focal colonic wall thickening and luminal narrowing (more common) to a large soft tissue mass with central necrosis resembling an abscess (34).
In the evaluation of nodal metastases CT has the same limitation of MRI and US: not all enlarged nodes contain tumour and normal-sized nodes may have microscopic tumour involvement (34).
Although the liver is the main organ to be involved with metastases from colorectal cancer (which are typically hypovascular); due to the dual drainage of the lower rectum (the middle and inferior hemorrhoidal veins drain into the inferior vena cava via the pelvic veins) pulmonary metastases without hepatic metastases may be seen (34).
After surgery,
tumor recurrence usually appears as a soft-tissue mass in or near the surgical site.
This appearance can mimic postoperative fibrosis hindering the diagnosis of local recurrence.
Only a progression in follow-up may suggest the diagnosis (34).
PET-CT has an important value in the diagnosis of local recurrence.
PET
PET,
an imaging technique that is based on visualising metabolic changes within cells,
can be used for detecting increased tumorous activity in case of rectal cancer as well.
Most commonly,
FDG (fluorodeoxyglucose) is used for tracing.
There are some drawbacks for this technique - FDG-PET still lacks spatial resolution,
therefore its use for tumour invasion is limited; PET-CT is associated with high dosis of radiation (35,36).
Moreover,
mucinous tumours are a known pitfall for FDG-PET (34).
PET-CT is more commonly used in systemic disease or advanced phases of treatment,
for example,
for detecting suspected recurrent tumours and distant metastases.
In case of M staging,
FDG-PET is highly sensitive (38),
however it is not commonly included into routine work-up algorithms due to little cost-effectiveness (10).