CLINICAL ISSUES
Most of the times the AIVC is asymptomatic and is diagnosed incidentally by imaging.
The most common clinical symptoms are related to venous insufficiency of lower extremities and/or idiopathic deep venous thrombosis.
A recent report states that 5% of the patients with idiopathic deep venous thrombosis who are under 30 years old have an absent IVC.
An increase of the renal venous pressure could lead to haematuria.
Varicocele in men or pelvic congestion syndrome in women could be other causes for clinical symptoms.
IMAGING
Usually the most evident morphological changes seen with imaging techniques are related to the redistribution of the venous flow and the development of collateral pathways.
In fact,
aside from the main cause,
imaging findings and clinical issues are very similar to those in patients with chronic IVC occlusion.
Common collateral pathways
The common collateral pathways in disturbance in drainage of IVC (incl.
congenital absence) are:
- Deep pathway (most common) – It involves the ascending lumbar veins (mainly) Fig. 8 Fig. 10 Fig. 3 ,
external an internal vertebral plexuses.
They anastomose with the azygos Fig. 9 and hemiazygos veins and so the blood finds its way to the superior vena cava and the right atrium.

Fig. 7: Deep collateral pathway.
References: Georgi Valchev, MD
- Intermediate pathway (rare) – it could develop in cases with absence of the infrarenal part of the IVC when the renal and suprarenal portions are preserved.
It that case the blood could follow a return through the internal iliac veins to the uterine / prostatic plexuses and then through the ovarian / pampiniform plexus to the left gonadal vein which flows into the left renal vein.
If the absence of the infrarenal IVC is below the point of joining of the right gonadal vein,
blood return could be done through the right ovarian / testicular also.

Fig. 11: Intermediate collateral pathways.
References: Georgi Valchev, MD
- Superficial pathway (common) – Blood returns from the external iliac veins to the inferior epigastric veins.
The flow continues to the superior epigastric veins and then through the internal mammary to the brachiocephalic veins and eventually the superior vena cava.

Fig. 12: Superficial collateral pathway.
References: Georgi Valchev, MD
- Portal pathway (less common) – Venous blood from the internal iliac veins returns to the haemorrhoidal plexus and so through the superior rectal vein it reaches the inferior mesenteric,
then the lienal vein and the porta.

Fig. 14: Portal collateral pathway.
References: Georgi Valchev, MD
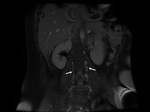
The imaging method of choice is contrast-enhanced computed tomography (CECT).
Fig. 15 Fig. 16 Fig. 17 The venous phase is essential.
All of the described changes can be assessed in a well-performed examination.
Multiplanar reformations could be very useful and aid to the diagnosis.
Magnetic-resonance imaging (MRI) is a good method for more reliable distinguishing of aberrant vessels from masses by using flow voids or flow-related enhancement.
Despite that,
other findings are similar to CT.
Fig. 18
Ultrasonographic findings could prove very helpful.
Since deep venous thrombosis is one of the more common related conditions,
often ultrasound (US) is the first imaging technique used.
Great suspicion of a present IVC anomaly should be raised especially with younger patients.
Being a retroperitoneal structure the IVC is sometimes hard to visualise throughout its entire course.
The hepatic segment is easier to examine and draining of the hepatic veins directly into the right atrium shows absence of that segment.
Dilated collateral veins is another finding related to the condition.
Prenatal US could be useful in finding IVC anomalies but specifying the exact type is hard,
because visualising the IVC throughout its entire course presents a significant challenge.
The abnormalities can be detected during fetal echocardiography.
The absence or interruption of the IVC is related with a dilated superior vena cava and azygos vein.
In the four-chamber view an enlarged azygos vein can be seen posterior to the descending aorta as a “double vessel sign”.
In the “three vessel view” along with the aorta and pulmonary artery,
the superior vena cava is seen so dilation could be found.
The point where the azygos vein joins the superior vena cava could also be demonstrated.
Venography is the most accurate imaging technique.
It is able to show all paths of venous drainage.
However,
its use is limited and less invasive methods as CECT and MRI are preferred.
From the possible related changes in the venous flow,
the one that could be demonstrated on a conventional X-ray is the dilation of the azygos vein.
Fig. 19
DIFFERENTIAL DIAGNOSIS
The main differential is hypoplasia of IVC.
The clinical findings (if present) are usually the same and are related to venous insufficiency and deep venous thrombosis.
The collateral venous pathways follow the same principles as with absence of the IVC.
Fig. 20
The most common cause that could lead to similar changes is the chronic obstruction of the IVC.
This can be due to thrombosis or extrinsic compression/invasion (primary tumours like sarcoma or leiomyomatosis are rare).
The infrarenal part is more often involved in thrombosis.
In a thrombosis occurring early in life the vein is very small but still distinguishable on a well performed CECT or MRI.
There are usually signs of chronic thrombosis like calcifications.
Fig. 21
Dilated paravertebral collateral veins may mimic retroperitoneal lymphadenopathy.
Fig. 22 And from a practical point of view that differential is more important than the aforementioned.
One of the concerns is that a wrong diagnosis could lead to an unnecessary intervention like fine needle biopsy with potentially serious consequences.
TREATMENT
If there is no clinical significance and there is sufficient flow through the collateral network,
no treatment is required.
Thrombosis is the most common related condition that needs to be treated.
A surgical approach is also possible with a prosthetic bypass from the external iliac to the intrathoracic azygos vein.