The development and drainage of the pulmonary veins is a complex and incompletely understood process.
A variety of abnormalities form a spectrum of disease ranging from normal insertion of an abnormal number of veins to abnormal insertion of some or all of the pulmonary veins.
In addition,
one or more pulmonary veins may be obstructed,
regardless of normal or abnormal insertion.
Controversy exists about the origin of the primordial pulmonary vein.
Nevertheless,
it is generally accepted that a common pulmonary vein forms in the dorsal mesocardium and is progressively incorporated into the posterior wall of the left atrium.
As the atrium expands and the common vein is absorbed,
the four major branches (two left and two right) achieve their discrete insertions.
The primary diseases that fall within this spectrum include partial anomalous pulmonary venous connection (PAPVR),
sinus venosus defect,
total anomalous pulmonary venous connection (TAPVR),
cor triatriatum and pulmonary vein stenosis.
[1]
Embryology:
Human Lung Stages
|
Stage
|
Human
|
Features
|
|
Embryonic
|
week 4 to 5
|
lung buds originate as an outgrowth from the ventral wall of the foregut where lobar division occurs
|
|
Pseudoglandular
|
week 5 to 17
|
conducting epithelial tubes surrounded by thick mesenchyme are formed,
extensive airway branching
|
|
Canalicular
|
week 16 to 25
|
bronchioles are produced,
increasing number of capillaries in close contact with cuboidal epithelium and the beginning of alveolar epithelium development
|
|
Saccular
|
week 24 to 40
|
alveolar ducts and air sacs are developed
|
|
Alveolar
|
late fetal to 8 years
|
secondary septation occurs,
marked increase of the number and size of capillaries and alveoli [2]
|
Development of the Venous System
Early in the formation of the lungs,
the blood coming from the lung buds drains to the splanchnic plexus,
which connects to the paired common cardinal and umbilicovitelline veins.
The right common cardinal system later evolves into the right sinus venosus,
which,
in turn,
becomes the right superior vena cava and azygos vein.
The left common cardinal vein evolves into the left sinus venosus,
which,
in turn,
becomes the left superior vena cava and coronary sinus.
The umbilicovitelline system becomes the inferior vena cava,
ductus venosus,
and portal vein.
At 25-27 days' gestation,
the developing pulmonary venous plexus retains connections to the right superior vena cava,
left superior vena cava,
and portal system.
No direct communication to the left atrium exists.
At 27-29 days' gestation,
the primitive pulmonary vein appears as an endothelial out-pouching from either the posterior superior left atrial wall or from the central part of the sinus venosus proximal to the primordial lung venous plexus.
Connection between the primitive pulmonary vein and pulmonary venous plexus occurs by 30 days' gestation.
The common pulmonary vein enlarges and incorporates into the left atrium,
and,
normally,
the pulmonary venous part of the splanchnic plexus gradually loses its connection with the cardinal and umbilicovitelline veins.
Failure of the common pulmonary vein to connect with the pulmonary venous plexus leads to persistence of one or more earlier venous connections to the right superior vena cava,
to the left vertical vein/innominate vein,
or to the umbilicovitelline vein/portal vein.
Failure of the septum primum to normally form or abnormal septation of the sinus venosus can lead to direct connection of the pulmonary veins to the right atrium.
Late obstruction of the common pulmonary vein,
after earlier venous channels have disappeared,
can lead to isolated pulmonary vein atresia.
Failure of incorporation of the common pulmonary vein may lead to a left atrial shelf or membrane of  cor triatriatum (ie,
stenosis of the common pulmonary vein).
[3]
cor triatriatum (ie,
stenosis of the common pulmonary vein).
[3]
All pulmonary venous return connects to the systemic venous system.
For this reason,
right atrial and right ventricular enlargement occurs.
If significant pulmonary venous obstruction develops,
right ventricular hypertrophy occurs.
TAPVR occurs alone in two thirds of patients and occurs as part of a group of heart defects (eg,
heterotaxy syndromes) in approximately one third of patients.
An atrial septal defect (ASD) or patent foramen ovale,
considered part of the complex,
serves a vital function in this condition for maintaining left ventricular output.
Because diagnosis of most patients occurs in early infancy,
a ductus arteriosus is frequently found as well.
[4]
Images for this section:
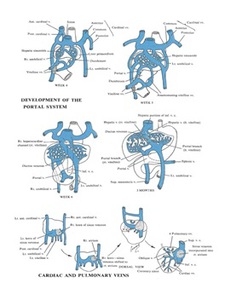
Fig. 1: Development of the Venous System: the Portal System and Pulmonary Veins [5]
References: Edgar R., et al., (2013 Jul 17), The Embryonic Development, Stem Cells, and Regenerative Medicine Research Portal, Retrieved from http://discovery.lifemapsc.com/library/review-of-medical-embryology/chapter-127-development-of-the-venous-system-the-portal-system-and-pulmonary-veins (cited 12.12.2015)
Classification
This anomalous pulmonary venous return can be either partial (PAPVR) or total ( TAPVR).
TAPVR).
Classification system for  TAPVR based on the site of pulmonary venous drainage:
TAPVR based on the site of pulmonary venous drainage:
type I (supracardiac connection): the 4 pulmonary veins drain via a common vein into the right superior vena cava,
left superior vena cava,
or their tributaries.
type II (cardiac connection): the pulmonary veins connect directly to the right heart (eg,
coronary sinus or directly to the right atrium).
type III (infradiaphragmatic connection): the common pulmonary vein travels down anterior to the esophagus through the diaphragm to connect to the portal venous system.
type IV (mixed connections): the right and left pulmonary veins drain to different sites (eg,
left pulmonary veins into the left vertical vein to the left innominate,
right pulmonary veins directly into the right atrium or coronary sinus).
[3]
Classification system for PAPVC based on the site of pulmonary venous drainage:
Four types have been described:
· supra-cardiac
o  persistant left superior vena cava
persistant left superior vena cava
o right superior vena cava (most common)
· cardiac
o right atrium
o innominate vein
· infracardiac
o  portal vein
portal vein
o inferior vena cava ( Scimitar syndrome)
Scimitar syndrome)
· mixed
o a combination of two or more of the above anomalies [6]
PAPVC anatomical classification:
- PAPVC with ASD (80-90% cases): the most common type
The ASD is usually the sinus venosus type.
Approximately 10% have a secundum ASD associated with this anomaly.
The anomalous pulmonary vein,
usually the right upper or middle pulmonary vein can either override the intra atrial septum (anomalous drainage) or can drain separately into the superior vena cava (true anomalous connection).
Usually,
the connection is unobstructed.
- PAPVC with intact atrial septum (isolated PAPVC)
This is a very rare finding and mostly involves the anomalous drainage of the right upper pulmonary vein into the superior vena cava.
Only 3% of patients had PAPVC from the left lung to the innominate vein.
- PAPVC with complex congenital heart disease (heterotaxia)
This is usually seen with heterotaxia syndromes ( polysplenia).
polysplenia).
Left atrial isomerism with a common atrium is observed.
Because of the abnormal positioning of the intra-atrial septum,
the right-sided pulmonary veins anastomose to the anatomically right-sided atrium,
which is the atrium that also receives the inferior vena cava.
About half of the cases may involve some degree of obstruction,
either due to narrowing of a discrete area or due to diffuse hypoplasia of that vein.
This condition is associated with presence of ipsilateral pulmonary arterial hypoplasia.
- Scimitar syndrome (right pulmonary vein to inferior vena cava with lung sequestration)
Scimitar syndrome,
or pulmonary venolobar syndrome,
is a rare  congenital heart defect characterized by anomalous venous return from the right lung (to the systemic venous drainage,
rather than directly to the left atrium). Usually,
the anomaly involves the right lung; the left lung is very rarely involved.
congenital heart defect characterized by anomalous venous return from the right lung (to the systemic venous drainage,
rather than directly to the left atrium). Usually,
the anomaly involves the right lung; the left lung is very rarely involved.
More commonly known as Scimitar syndrome after the curvilinear pattern createdon a chest radiograph by the pulmonary veins that drain to the inferior vena cava.This radiographic density often has the shape of a  scimitar,
a turkish curved sword (scimitar sign).
scimitar,
a turkish curved sword (scimitar sign).
This syndrome is also known as Halasz syndrome,
mirror-image lung syndrome,
hypogenetic lung syndrome,
epibronchial right pulmonary artery syndrome,
vena cava bronchovascular syndrome,
or congenital pulmonary venolobar syndrome.
Anomalies included in this syndrome: a venous anomaly,
an arterial anomaly,
and pulmonary anomaly.
The pulmonary abnormality includes a sequestered lobe of the lung that is separated from the bronchial tree and has nonfunctioning lung tissue.
It is associated with hypoplastic or aplastic right pulmonary artery segments,
hypoplastic or absent bronchi,
hypoplasia of the right lung,
horseshoe lung,
and eventration of the hemidiaphragm.
The arterial anomaly is a collateral arterial vessel (aberrant vessel) with blood supply of the right lower lobe from the abdominal aorta.
Dextropositioning of the heart due to right lung hypoplasia may be observed.
Usually,
the atrial septum is intact.
About 25% cases have other associated congenital heart defects.
[7]
Pulmonary venous obstruction
May occur in all types of anomalous connections:
- supracardiac connections: obstruction may occur at the origin of the ascending (vertical) vein or its attachment to the innominate vein,
or the vertical vein may be obstructed as it crosses between the left pulmonary artery and the left bronchus.
In cardiac connections,
obstruction to pulmonary veins seldom develops but may occur at the junction of the common vein to the coronary sinus.
- infradiaphragmatic connections: severe obstruction almost always inhibits pulmonary venous flow with obstruction of the common pulmonary vein.
This obstruction occurs either as it travels through the diaphragm,
at its junction with the portal vein system,
or as an obstruction of pulmonary venous flow as the ductus venosus closes and pulmonary vein flow is forced to cross the liver portal sinusoid system.
Finally,
in all types,
obstruction may occur because of restrictive atrial septal defect size and because of small left atrial size.
[4]
PARTIAL ANOMALOUS PULMONARY VENOUS CONNECTION
Partial anomalous pulmonary venous connection (PAPVC) represents failure of one or more of the pulmonary veins to be incorporated into the left atrium.
The anomalous veins empty instead into the systemic circulation.
By definition,
at least one pulmonary vein must drain normally into the left atrium.
Prevalence and Epidemiology
The incidence of PAPVC at autopsy it is between 0.6% - 0.7%.
The clinical frequency is less,
indicating that many cases remain asymptomatic.
[1]
Clinical diagnosis of isolated PAPVC is quite rare.
PAPVC occurs in approximately 10% of patients with a proven ASD (atrial septal defect).
PAPVC from the right lung is twice as common as PAPVC from the left lung.
The most common form of PAPVC is one in which a right upper pulmonary vein connects to the right atrium or the superior vena cava.
Mortality/Morbidity
Few data are available because mortality credited to the defect occurs only in adults and the diagnosis has historically been made at autopsy.
Major morbidity,
including arrhythmias,
right-sided cardiac failure and pulmonary vascular disease,
primarily occurs in adults.
Sex
The incidence is higher in the female population.
Age
PAPVC is a congenital defect.
Clinical evidence of this congenital defect may not be apparent until the patient reaches middle age.
[8]
Etiology and Pathophysiology
Anomalous connections can occur both above and below the diaphragm,
generally to an ipsilateral systemic vein.
The right-sided pulmonary veins most often drain into the embryologic derivatives of the right cardinal vein,
usually the inferior vena cava (IVC) or superior vena cava (SVC).
Anomalous left-sided veins typically empty into derivatives of the left cardinal vein,
most often the left innominate vein or the coronary sinus.
Anomalous veins may also connect through remnants of the primitive splanchnic plexus to contralateral systemic vessels,
although this is less common.
Connection of a pulmonary vein (usually the right upper lobe branch) to the posterior SVC at the junction of the right atrium due to a defect of the common wall between the SVC and the right upper lobe pulmonary vein represents a unique process known as a sinus venosus defect.
Drainage of the right pulmonary veins to the IVC may be associated with systemic arterial supply and hypoplasia of the ipsilateral lung with secondary dextroposition of the heart.
This condition,
variably referred to as congenital venolobar syndrome or scimitar syndrome ,
is often placed within the continuum of bronchopulmonary dysplasias.
A horseshoe lung refers to the fusion of the lower lobes across the midline without an intervening fissure.
This anomaly is highly associated with scimitar syndrome,
with up to 80% of patients with horseshoe lung also affected with PAPVC in some series.
[1]
Numerous factors determine the ratio of pulmonary blood flow (Qp) to systemic flow (Qs).
The shunt magnitude,
expressed as the Qp:Qs ratio,
and other factors determine development of symptoms and complications.
The most important factor is the number of pulmonary veins that drain into the systemic circulation.
The more veins that anomalously drain,
the more blood returns to the right side of the heart.
This defect becomes clinically significant when 50% or more of the pulmonary veins anomalously return.
The source of the returning blood plays a role in determining the clinical effect of the defect.
In an individual who is upright,
blood flow to the lungs is primarily directed to the lower and middle lobes.
Therefore,
more blood returns to the systemic venous circulation in individuals in whom the anomalous connection drains into either the right middle and lower lobes or the left lower lobe of the lung.
An associated cardiac defect,
such as an ASD,
may add to the left-to-right shunting.
In scimitar syndrome,
the flow from the PAPVC causes a left-to-right shunt.
Again,
the number of anomalous veins involved determines the symptoms and signs.
The aberrant artery may cause additional left-to-right shunt.
Over many years,
excessive pulmonary venous return to the right side of the heart causes right atrial and ventricular dilation.
The consequences: risk of arrhythmia development,
right-sided  heart failure and development of pulmonary hypertension.
heart failure and development of pulmonary hypertension.
A native PAPVC usually does not have any associated obstruction to venous drainage.
However,
obstruction may occur postoperatively due to baffle obstruction.[9]
Manifestations of Disease
Clinical presentation:
Determining the natural history of this condition was difficult before the era of direct cardiac imaging (ie,
echocardiography,
cardiac catheterization) because the diagnosis was made only postmortem.
Children with PAPVC usually remain asymptomatic and are referred based on an incidentally noted cardiac murmur.
Symptoms may occur in older patients and may be secondary to right-sided volume overload or pulmonary vascular obstructive disease.
The development of complications from PAPVC clearly depends on how many pulmonary veins abnormally return to the right heart.
A single anomalous vein is not usually hemodynamically significant and does not produce any symptoms.
About 10% of patients with an ASD also have PAPVC and may have symptoms of right-sided overload.
Dyspnea may occur in adults (it is rare in children).
A child may experience exercise intolerance as a symptom in cases in which more than 50% of pulmonary veins anomalously drain.
Palpitations may reflect cardiac arrhythmias,
which are almost always supraventricular in origin (due to right atrial dilatation and may present at older age).
Hemoptysis is a rare symptom that reflects either chest infection or the development of pulmonary vascular disease.
Chest pain may be evidence of right-heart ischemia but does not occur in childhood.
More commonly,
chest pain may be a manifestation of recurrent  bronchitis.
bronchitis.
Peripheral edema can occur in adults with cardiac failure.
The severity of symptoms in scimitar syndrome depends on:
· degree of pulmonary hypertension
· severity and frequency of chest infections.
[10]
Physical examination findings are usually more revealing than the history,
but positive findings depend on the volume of abnormal pulmonary venous return to the right heart.
If only a single vein is anomalous,
the physical examination findings may be normal.
In a patient with a larger volume of abnormal veins,
physical examination findings are similar to those of an uncomplicated ASD.
Left parasternal lift reflects right ventricular dilation.
Impulse in the second left intercostal space reflects pulmonary artery dilation.
A soft systolic ejection murmur is heard over the pulmonary area,
reflecting turbulence in the pulmonary trunk due to increased right ventricular ejection volume.
The second heart sound is always widely split but may have normal respiratory variation.
A delay in the pulmonic closure component of the second sound in patients with PAPVC who have an intact atrial septum due to an increased systemic venous return to the right heart.
However,
in patients with PAPVC and ASD,
volume flow into the right heart is always increased,
and respiration further augments that flow.
Therefore,
splitting of S2 proceeds from wide during expiration to wider during inspiration.
This does not occur in patients with a significant ASD,
in whom second heart spitting is wide and fixed.
In the presence of an ASD,
variations in systemic venous return during respiration are counterbalanced by reciprocal changes in flow through the ASD,
maintaining total right ventricular flow more or less constant.
A mid-diastolic murmur due to increased transtricuspid right ventricular filling may be heard over the tricuspid valve area at the lower left sternal border.
Right-sided heart failure signs in adults: hepatomegaly,
jugular venous distension,  ascites,
and peripheral edema.
ascites,
and peripheral edema.
Pulmonary vascular disease may occur in older adults,
although this is rare.
Clinical signs of pulmonary hypertension: right ventricular parasternal lift,
absence of systolic murmur,
narrowly split S2 with a loud pulmonic component,
and,
occasionally,
an early,
high-frequency murmur of pulmonic regurgitation.
Cyanosis does not occur in the presence of an intact atrial septum.[11]
Differential Diagnosis
Top Differential Diagnoses
· Congenital Interruption Pulmonary Artery
· Pulmonary Sequestration
· Wandering Vein
· Fibrosing Mediastinitis
· Chronic Thromboembolic Occlusion
· Swyer-James Syndrome
· Left Superior Vena Cava (SVC)
· Pulmonary Varix
· Left Superior Intercostal Vein
Left Superior Vena Cava (SVC):
2 vessels ventral to the left upper lobe bronchus: Left superior vena cava and left superior pulmonary vein
o Vessels absent with PAPVR
o Drains into coronary sinus,
right sided SVC may be absent
Pulmonary Varix
o Acquired or development dilatation pulmonary vein at
its entrance to left atrium
o Lobulated mass or nodule posterior to the heart
Left Superior Intercostal Vein
o 2-4 intercostal veins and accessory hemiazygos form
the left superior intercostal vein which drains into the left brachiocephalic vein,
enlarges with SVC or left brachiocephalic vein occlusion
o Aortic "nipple" on chest radiograph
Congenital Interruption Pulmonary Artery
o No vertical vein
o Aortic arch on opposite side of interrupted artery
o Normal bronchial branching
o Volume reduction affected lung marked similar to
o marked hypoplasia in scimitar syndrome
Pulmonary Sequestration
o No vertical vein
o Systemic supply to sequestered lung,
usually from
o descending or abdominal aorta
o More common in the left costovertebral angle
o Lung usually dense but may be hyperlucent,
o simulating hypogenetic lung
o Normal venous drainage in intralobar sequestration
Wandering Vein
o Anatomic variant
o Pulmonary vein may normally wander in lung with
o normal drainage int
Fibrosing Mediastinitis
o No vertical vein
o Pulmonary veins may be obstructed
o Endemic geographic area for Histoplasmosis
o Focal hilar or mediastinal mass narrowing nearby
o airways or vessels
o Calcification of mass common (60-90%)
Chronic Thromboembolic Occlusion
o < 1% of patients following acute thromboembolism
o No vertical vein
o Small artery due to organized thrombus
o Normal pulmonary venous anatomy,
size may be
o small from diminished blood flow
Swyer-James Syndrome
o No vertical vein
o Volume loss affected lung
o Normal pulmonary venous anatomy [12]
Treatment
Medical
Medical therapy of partial anomalous pulmonary venous connection (PAPVC) is not indicated for asymptomatic patients.
Heart failure in adults can be managed with diuretics,
cardiac glycosides,
afterload reduction,
and beta blockade.
Arrhythmias should be appropriately treated.
[13]
Surgical/Interventional
All patients who are symptomatic and do not have a contraindication should be treated surgically.
Specifically,
the current consensus holds that if the Qp:Qs (shunt fraction) is greater than 1.5 : 1,
surgical closure is performed.
PAPVC with shunt ratios below 1.5 : 1 are usually well tolerated and can be clinically observed.
The particular repair will vary according to the site of anomalous drainage and the coexistence of any other form of heart disease.
Anomalous left pulmonary veins may be reanastomosed to the left atrial appendage.
Right-sided anomalous veins are often anastomosed to the right atrium and connected to the left atrium with a patch or baffle through a preexisting or surgically created ASD.
In general,
surgical repair of PAPVC is associated with very good outcomes.
[1]
For the PAPVC to the superior vena cava (SVC),
the repair techniques may include internal patch technique,
with or without SVC enlargement,
or the caval division technique with atriocaval anastomosis (Warden technique).
Children with internal patch technique must be observed for obstruction of the SVC with SVC syndrome,
sick sinus syndrome,
obstruction of the pulmonary veins,
and supraventricular tachyarrhythmias.
Patients with scimitar syndrome,
however,
often do poorly and suffer from high degrees of postoperative pulmonary venous stenosis related to baffle obstruction.[14]
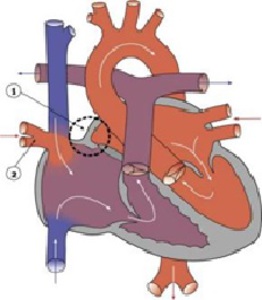
Fig. 2: Partial anomalous pulmonary venous connection (PAPVC)-From:
Hill, M.A. (2015) Embryology Cardiovascular System - Abnormalities. Retrieved December 11, 2015, from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Abnormalities [15]
References: Hill, M.A. (2015) Embryology Cardiovascular System - Abnormalities. Retrieved December 11, 2015, from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Abnormalities
TOTAL ANOMALOUS PULMONARY VENOUS CONNECTION
Drainage of all the pulmonary veins to any location otherthan the left atrium constitutes the entity known as totalanomalous pulmonary venous connection (TAPVC).
Thepulmonary veins may drain to the right atrium,
coronarysinus,
or other systemic veins.
Prevalence and Epidemiology
TAPVC is generally estimated to represent not more than 2.0% of congenital heart disease.
For the most part,
no strong gender prevalence has been noted.
[16]
The curious exception to this is total anomalous connection to the portal system,
which affects males 3,5 times more often than females.
There is a common association of TAPVC with heterotaxy syndrome.
Patients with this association heterotaxy and TAPVC (particularly those complicated by post-repair stenosis) are more likely to have poor outcomes.[1]
Etiology and Pathophysiology
Failure of the normal incorporation of the common pulmonary vein into the dorsal wall of the left atrium with persistent connections of all pulmonary veins to systemic vessels,
right atrium,
or coronary sinus results in total anomalous venous connection.
Various classification schemes for anomalous drainage patterns have been described.
One of the most used is the classification made by Craig and Darling and includes supracardiac drainage (type I),
drainage at the cardiac level (type II),
infracardiac drainage (type III),
and mixed patterns (type IV).
[1]
The most frequently described connection is to the left innominate vein.
Other frequently encountered sites of drainage include the coronary sinus,
the right atrium (secondary to malposition of the septum primum),
the SVC and the portal system.
Patients with TAPVC often have a concurrent ASD,
VSD,
or patent ductus arteriosus (PDA).
Postnatal survival in patients with TAPVC is dependent on the presence of a shunt.
In addition,
many cases of TAPVC demonstrate some degree of pulmonary venous obstruction,
especially the infradiaphragmatic type.
Causes of venous obstruction include intrinsic abnormality of the vessel wall with medial hypertrophy,
compression by adjacent structures,
and narrowing at the level of the diaphragm or ductus venosus.
The presence of venous bstruction portends a worse prognosis.
[16]
Manifestations of Disease
Clinical Presentation
In the newborn with TAPVC,
clinical symptoms and signs can be variable,
depending on the presence and degree of obstruction to the pulmonary veins.
When TAPVC is unobstructed,
symptoms in the newborn infant are variable,
depending on the size of the associated shunt and direction of flow.
Symptoms and signs can range from mild cyanosis and murmur to severe pulmonary overcirculation
with respiratory distress.
Sometimes,
patients are so asymptomatic that the diagnosis is not made until they have reached adolescence or adulthood.
Patients tend to present earlier with more severe symptoms when there is pulmonary venous obstruction.
Tachypnea with gasping and intercostal retractions due to pulmonary edema develop within a few days of birth.
Differential Diagnosis
From Clinical Presentation
An infant with TAPVC without venous obstruction may clinically appear similar to infants with any form of large shunt lesion (VSD,
ASD,
PDA,
atrioventricular canal).
Newborns or infants with TAPVC and venous obstruction are more likely to be cyanotic with severe tachypnea and retractions due to pulmonary edema.
From Imaging Findings
The lack of left-sided heart enlargement on the chest radiograph helps exclude shunt lesions such as VSD,
PDA,
and atrioventricular canal.
Depiction of aberrant pulmonary venous drainage on more advanced imaging modalities essentially eliminates other differential considerations.
The differential considerations are restricted to the type of TAPVC (supracardiac,
cardiac,
infracardiac,
mixed).
[1]
Treatment
Medical
Whereas medical therapy alone is insufficient for the treatment of TAPVC,
therapy should be instituted before surgery to stabilize the patient in terms of metabolic acidosis and volume overload.
Surgical/Interventional
The definitive treatment of TAPVC is surgical.
In the case of supracardiac TAPVC draining to the left innominate vein,
the common vein is anastomosed side-to-side with the left atrium,
and the communication to the systemic veins as well as additional shunts,
such as ASD or VSD,
are closed.
Other types of TAPVC are treated in a similar manner.
Patients with preexisting venous obstruction and coexistent complex heart disease,
especially single ventricle,
have worse outcomes.
Other features that have been correlated with poor outcome include younger age and type II connection.[17]
Pulmonary vein stenosis after repair is a common complication,
particularly in patients with heterotaxy.
Information for the Referring Physician
Detailed descriptions of the course and drainage sites of all pulmonary veins are crucial.
The size of the individual pulmonary veins yields important prognostic information.
Obstruction to venous drainage and signs of right ventricular volume overload or venous congestion on plain films implying infracardiac TAPVC should be clearly stated.
[1]
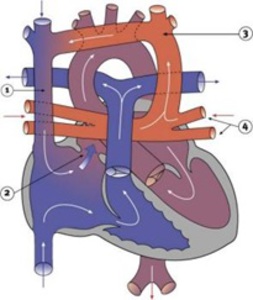
Fig. 3: Total anomalous pulmonary venous connection – From: Hill, M.A. (2015) Embryology Cardiovascular System - Abnormalities. Retrieved December 11, 2015, from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Abnormalities [15]
References: Hill, M.A. (2015) Embryology Cardiovascular System - Abnormalities. Retrieved December 11, 2015, from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Abnormalities
SINUS VENOSUS DEFECT
A sinus venosus septal defect (SVD) most often represents an absence of the common wall between the right upper lobe pulmonary vein and the SVC or right atrium; it is also known as a superior type of SVD.
Much less commonly,
an inferior type of SVD can occur when the common wall between the right atrium and the right lower or middle pulmonary vein is absent,
often associated with malposition of the septum primum.
[1]
Prevalence and Epidemiology
The SVD has been estimated to account for up to 10% of ASDs.[19] The exact prevalence is difficult to measure because of the often subclinical nature of the condition and the difficulty in detection with standard first-line cardiac imaging techniques (plain radiography and echocardiography).
The ratio between female and male is 2:1.
[1]
Etiology and Pathophysiology
The lack of an intact intervening wall between a pulmonary vein and the SVC or right atrium results in “unroofing” of the pulmonary vein,
thereby creating anomalous pulmonary venous drainage to the right atrium.
[1] An interatrial connection posterior and superior to the fossa ovalis is commonly present.
This does not represent an ASD,
but rather it is a connection between the atria formed by the unroofed insertion of the pulmonary vein.
Because of higher left-sided pressures,
flow of blood can pass retrograde from the left atrium through the orifice of the pulmonary
vein and then enter the SVC through the defect and continue to the right atrium,
resulting in a left-to-right shunt.
Additional anomalous connecting pulmonary veins to the SVC superior to the defect as well as other systemic veins may be present.
Manifestations of Disease
Clinical Presentation
The often large interatrial communication of an SVD results in a significant left-to-right shunt with increased pulmonary blood flow; some patients can be relatively
asymptomatic while others may present with congestive heart failure.
Symptoms include dyspnea on exertion,
palpitations,
and angina.
[1] As with conventional ASDs,
SVD may be the cause of an otherwise unexplained stroke due to paradoxic embolization.
Differential Diagnosis
From Clinical Presentation
ASDs and other forms of PAPVC are often difficult to distinguish clinically from SVDs.
From Imaging Findings
The main differential consideration is PAPVC.
Whereas the distinction can often be difficult on transthoracic echocardiography,
transesophageal echocardiography,
cardiac MR,
and CTA should allow exclusion of other entities.
[1]
Treatment
Medical
The definitive treatment of SVD is surgical.
The SVD more often requires surgical repair because the shunt is usually larger with more severe right ventricular enlargement.
Medical therapy aims to optimize cardiopulmonary status in anticipation of operative treatment.
Surgical/Interventional
The traditional means of SVD repair consists of a single patch closure of the defect that baffles the anomalous right upper lobe pulmonary vein back to the left atrium.
This technique can be complicated by narrowing and obstruction of the SVC postoperatively.
A second technique involves placement of a second patch to widen the SVC–right atrial junction to reduce the incidence of postoperative SVC narrowing,[1] but the sutures placed at the SVC–right atrial junction can lead to sinoatrial node dysfunction.
The Warden procedure involves transection of the cranial portion of the SVC above the anomalous venous connection and anastomosis of the SVC to the right atrial appendage.
The connection of the right upper lobe pulmonary vein and the caudal end of the SVC is then patch closed.
Because of the decrease in reported complications of obstruction and nodal dysfunction,
the Warden technique has gained in popularity.
[19]
Information for the Referring Physician
In addition to pulmonary vein anatomy,
the size and location of the SVD should be described.
Right ventricular size and function should be assessed.
The ratio of pulmonary-to-systemic flow (shunt fraction) should also be reported.
COR TRIATRIATUM
When the pulmonary veins drain to an accessory chamber separated by a membrane from the remainder of the left atrium,
the result is termed cor triatriatum.
Prevalence and Epidemiology
The reported incidence of cor triatriatum ranges between 0.1% and 0.4%.
No racial or gender predilections have been described.
[1]
Etiology and Pathophysiology
Like the previously described pulmonary vein anomalies,
there is ample controversy surrounding the embryogenesis of cor triatriatum.
The most commonly held theory is based on incomplete incorporation of the common primordial pulmonary vein into the left atrium.
The common vein becomes a “third atrium” separated from the left atrium by a fibromuscular membrane.
This accessory chamber typically receives all the pulmonary veins and connects to the left atrium through a stenotic orifice (classic cor triatriatum or cor triatriatum sinister).
The common vein may also communicate with the right atrium (cor triatriatum dexter) or the systemic veins (cor triatriatum with PAPVC).
Yet another variation is subtotal cor triatriatum,
in which some of the pulmonary veins drain normally into the left atrium and others drain into an accessory chamber that then empties in any of the patterns previously described.[20]
Manifestations of Disease
Clinical Presentation
Clinical symptoms depend on the degree of stenosis affecting the orifice in the membrane dividing the accessory chamber and the true atrium and the presence or absence of a shunt (drainage into the right atrium or PAPVC).
Symptoms are variable,
ranging from none to pulmonary edema or pulmonary hypertension.
Patients often have respiratory symptoms and recurrent pulmonary infections and are misinterpreted as having primary pulmonary disease.
Differential Diagnosis
From Clinical Presentation
The patient may present with respiratory symptoms,
and primary pulmonary disease may be suspected.
Other causes of pulmonary hypertension,
either idiopathic or secondary to valvular or other congenital heart disease,
should be considered.
From Imaging Findings
The main differential consideration in evaluating a chest radiograph in cor triatriatum is infracardiac total anomalous pulmonary venous connection with obstruction or other severe left-sided heart obstructive lesions,
such as mitral stenosis.
Treatment
Medical
Medical management of pulmonary venous congestion is necessary until the patient is stable for definitive surgical repair.
Surgical/Interventional
Resection of the cor triatriatum membrane with patch closure of any coexistent ASDs is the treatment of choice.
If the patient also has anomalous venous connections,
these may be corrected.
Information for the Referring Physician
Accurate description of the size and drainage pattern of the accessory chamber in addition to clear descriptions of any associated anomalies is required.
If functional information (as in cardiac MRI) has been obtained,
it too should be included in the report.
Procedures
Cardiac catheterization is rarely necessary for precise anatomic diagnosis and hemodynamic evaluation.
Right heart pressures are normal in the pediatric patient.
Oxygen sampling may identify the location of an anomalous vein.
Oxygen saturation in the right atrium that is higher than that found in the SVC strongly indicates PAPVC to the right atrium,
provided that an atrial septal defect (ASD) has been ruled out.
Qp:Qs can be calculated.
Entering the anomalous vein with a catheter and injecting contrast confirms the diagnosis.
Complications of catheterization include bleeding at the vascular entry site,
infection,
decreased pulses distal to an arterial entry site,
and arrhythmia induction.
[1]
Causes
No causes of this condition are known.
No evidence has implicated common teratogens (eg,
drugs,
infections) in the genesis of APVC.
No evidence for a genetic predisposition has been reported.
[1]
Further Outpatient Care
Intermittent follow-up to assess right heart size and pressures and cardiac function and rhythm is necessary in patients who did not undergo surgical treatment.
With a significant shunt,
the pulmonary artery pressures can be elevated,
and pulmonary vascular resistance can increase with age.
Postoperatively,
possible obstruction of the pulmonary veins and superior vena cava (SVC) should be evaluated with echocardiography.
Regular electrocardiography (ECG) and 24-hour ambulatory ECG are also indicated to monitor for atrial arrhythmias.
Further Inpatient Care
Routine postoperative care of the patient who has undergone cardiac surgery for partial anomalous pulmonary venous connection (PAPVC) should be performed.
Pain control should be optimal to reduce the risk of atelectasis.
Anticipate early extubation unless contraindications are recognized; these include excessive chest tube drainage,
hemodynamic instability,
and oversedation.
Encourage early mobilization.
Monitor for atrial flutter,
atrial fibrillation,
and sinus node dysfunction.
Complications
No significant complications develop in pediatric patients who do not undergo surgery for a sinus venosus atrial septal defect (ASD) repair with internal patch technique to include the right upper pulmonary vein in the left atrium.
However,
other types of repair may be associated with complications such as SVC syndrome,
pulmonary venous obstruction,
atrial arrhythmias,
and sick sinus syndrome.
Arrhythmias may occur in adults with unrepaired PAPVC secondary to chronic right atrial enlargement due to volume overload.
Cardiac failure and,
very rarely,
pulmonary hypertension may develop in adults.
Patients with scimitar syndrome may be asymptomatic or may have symptoms related to lung pathology,
including recurrent  pneumonias.
pneumonias.
Prognosis
Prognosis is excellent for patients with PAPVC.
The perioperative mortality rate is comparable to that for ASD repair.
Prognosis becomes more guarded if the lesion is undetected for a long period and if complications,
particularly pulmonary hypertension,
develop.
[1]





