CAUSES
1.
Mesial temporal sclerosis
2.
Developmental- Diffuse – (poor surgical outcome) Lissencephaly,
Hemimegalancephaly
Focal- (good surgical response) Focal cortical dysplasia,
Heterotopia, Polymicrogyria,
Schizencephaly.
3. Epileptogenic tumors- Ganglioglioma,
Dysembryoplastic neuroepithelial tumor (DNET),
Pleomorphic Xanthoastrocytoma,
Hypothalamic hamartoma,
Desmoplastic infantile ganglioglioma.
4.
Gliosis
5.
Neurocutaneous syndromes – Sturge Weber Syndrome,
Tuberous sclerosis.
6.
Granulomas- Tuberculosis,
Neurocysticercosis.
7.
Inborn errors of metabolism
8.
Hypoxic ischemic encephalopathy
9.
Vascular malformations- Cavernous hemangioma,
Arterio-venous malformations.
10.
Miscellaneous- Rasmussen Encephalitis,
Parenchymal infectious disease.
Periventricular leucomalacia and idiopathic cause.
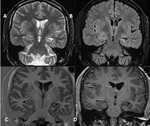
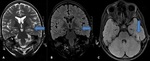
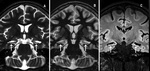
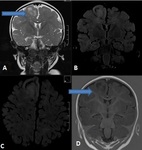
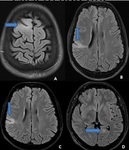
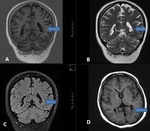
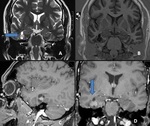
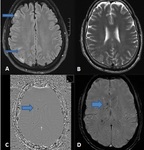
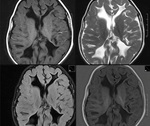
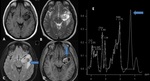
Hippocampal sclerosis (Mesial temporal sclerosis: MTS)(Fig.
2,3,4)
- Mesial temporal sclerosis is the most common cause of intractable epilepsy.
- MTS is the most common cause of partial complex epilepsy in adults.
- Most common etiology in young adult patients undergoing surgery.
- It is histologically characterized by neuronal loss and gliosis.
The hippocampus body is the most affected region.
- Although the cause is not well known it appears that in some predisposed individuals,
some kind of aggressions during the development (such as complex febrile seizures,
brain trauma or infection) can damage the hippocampus causing a mesial temporal sclerosis.
- In 15% of patients another developmental abnormality can be found,
mostly focal cortical dysplasia. This is called dual pathology.
- Surgical removal of visible MRI changes associated with unilateral mesial temporal sclerosis leads to seizure freedom in up to 80% of cases.
- Imaging differentials of hippocampal hyperintensity without volume loss are status epilepticus,
low grade tumors (Astrocytoma,
DNET) and encephalitis.
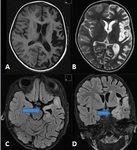
UNILATERAL HIPPOCAMPAL SCLEROSIS
- May diffusely involve the hippocampal complex or may be localized to the anterior region affecting only the head,
and is a less frequent cause of refractory focal TLE.
- In many patients,
the contralateral hippocampal complex may be involved,
though the changes may be asymmetric.
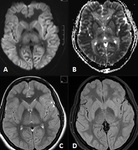
BILATERAL HIPPOCAMPAL SCLEROSIS (Fig 4)
- has been reported in about 10% of adult patients with medically refractory epilepsy
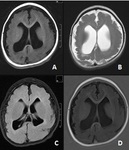
DUAL PATHOLOGY (Fig 3)
- Hippocampal sclerosis may co‑exist with a second pathology,
which may critically impact prognostication of seizure outcome in patients who undergo surgery.
MRI Features of Mesial Temporal Sclerosis
PRIMARY SIGNS:
- A small atrophic unilateral hippocampus.
- Hyperintensity on both T2 W and FLAIR images.
- Loss of the hippocampal internal architecture and that of normal digitations of the head
- Visual assessment of size,
architecture and signal intensity changes is quite sensitive,
with the eye being able to detect asymmetry of 14% or more.
SECONDARY SIGNS:
- Unilateral atrophy of the mamillary body,
fornix columns and the amygdale.
- Increased T2W signal in the anterior temporal lobe white matter with loss of grey-white matter demarcation in the ipsilateral anterior temporal lobe.
- Unilateral dilatation of temporal horn (A less reliable secondary sign)
- Unilateral atrophy of the collateral white matter bundle.
Key Points:
- Mesial temporal sclerosis is the most common cause of intractable epilepsy and also most common etiology in young adult patients undergoing surgery.
- Surgical removal of visible MRI changes associated with unilateral mesial temporal sclerosis leads to seizure freedom in up to 80% of cases.
- Imaging differentials of hippocampal hyperintensity without volume loss are status epilepticus,
low grade tumors (Astrocytoma,
DNET) and encephalitis.
- In 15% of patients another developmental abnormality can be found,
mostly focal cortical dysplasia. This is called dual pathology. MTS may be unilateral and bilateral.
- On MRI,
primary signs of MTS are small atrophic unilateral hippocampus,
hyperintensity on both T2/FLAIR images,
loss of the hippocampal internal architecture and normal digitations of the head region.
- Secondary signs of MTS are unilateral atrophy of the mamillary body,
fornix columns and the amygdale,
increased T2W signal in the anterior temporal lobe white matter with loss of grey-white matter demarcation in the ipsilateral anterior temporal lobe and unilateral dilatation of temporal horn.
Malformations of cortical development (MCD)
- Second cause of anatomic abnormality found in surgical series of patients with chronic epilepsy.
- Development of the cortex can be roughly separated into three steps:
- Cell proliferation and differentiation,
- Cell migration and
- Cortical organization.
- Any abnormality during the process of cortical formation can lead to malformations of corticalDevelopment (MCD).
- About 15% of patients with MCD have seizures refractory to medical treatment and surgery may be the definitive treatment.
In the case of diffuse alterations,
surgical treatment is ineffective,
contrary to focal lesions.
In general,
the degree of damage is related to the extension of the malformation.
- Based on pertinent aspects of central nervous system embryology,
the 2005 classification of MCD classifying them according to the developmental stage during which disruption is considered to be the cause of the anomaly.
There are four groups of malformations.
- Group I comprises disorders with decreased/increased proliferation or the proliferation of abnormal cells,
and includes microcephaly,
focal cortical dysplasia with balloon cells (Type 1),
hemimegalencephaly,
tuberous sclerosis,
dysembryoblastic neuroepithelial tumor and ganglioglioma/ gangliocytoma.
- Group II includes malformations due to abnormal neuronal migration,
such aslissencephalyand heterotopia.
- Group III comprises disorders due to abnormal cortical organization (including late
- Neural migration),
and also includes polymicrogyria,
schizencephaly,
cortical dysplasiawithout balloon cells (Type II) and microdysgenesis.
- Group IV includes all other MCD that are not otherwise classified,
such as mitochondrial disorders and sublobar dysplasia.
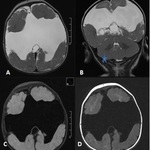
HEMIMEGALOENCEPHALY (HE).
- Hemimegalencephaly is a brain malformation characterized by the presence of an abnormally enlarged and dysplastic cerebral hemisphere.
The contralateral cerebral hemisphere usually appears normal,
except for being compressed or distorted.
- Hemimegalencephaly can occur isolated or associated with other syndromes: Type 1 NF,
tuberous sclerosis,
etc.
- Surgical treatment is recommended: hemispherotomy.
- In imaging,
the main finding iscortical thickening (pachygyria and polymicrogyria) affecting all or part of one hemisphere with ventricular enlargement and ipsilateral frontal horn pointed shape.
- Other findings are changes in the white matter signal intensity by hipermielinisation and hamartomas in the basal ganglia and ipsilateral olfactory tract.
Key Points:
- Hemimegalencephaly is a brain malformation characterized by the presence of an abnormally enlarged and dysplastic cerebral hemisphere.
The contralateral cerebral hemisphere usually appears normal,
except for being compressed or distorted.
- In imaging,
the main finding is cortical thickening (pachygyria and polymicrogyria) affecting all or part of one hemisphere with ventricular enlargement and ipsilateral frontal horn pointed shape.
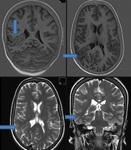
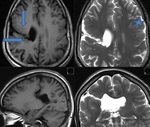
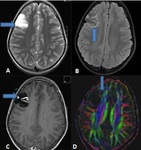
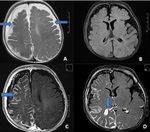
FOCAL CORTICAL DYSPLASIA (FCD) (Fig.
5 & 6)
- Epilepsy associated with FCD is often highly refractory to medication because of the high intrinsic epileptogenicity of these lesions.
- FCD is now recognized as one of the most common causes of seizures in children with intractable epilepsy,
accounting for nearly 80% of all surgically treated cases in children less 3 years of age.
- FCD was first described in 1971 by Taylor et al.
Pathologically are classified by the method recently described by Palmini et al in Types I and II.
- On clinical examination,
patients with type I FCD may or may not present with epilepsy or they may present by just learning disorders or may be asymptomatic.
- Most patients with type II FCD present refractory epilepsy and some require surgical treatment.
- FCD is sometimes associated with other lesions such as hippocampal sclerosis or tumours(DNT,
ganglioglioma).
- Characteristic MRI findings of type II FCDs include the presence of:
- Subtle cortical and subcortical high signal intensity (On FLAIR images).
- Often located at the bottom of a deep sulcus.
- Blurred grey-white matter interface.
- Focal cortical thickening.
- Transmantle sign: Sometimes the hyperintensity is seen extending from the subcortical area to the margin of the ventricle.
This is called the transmantle sign.
This finding represents the arrested neuronal migration.
- The findings of FCD can be subtle or undetectable at MR imaging alone.
However,
MR/FDG-PET fusion imaging and magnetic source imaging can help localize subtle architectural and signal abnormalities seen at MR imaging.
- Interictal FDG PET demonstrates areas of hypometabolic activity in regions of the cortex involved by FCD.
Key Points:
- Epilepsy associated with FCD is often highly refractory to medication and accounting for nearly 80% of all surgically treated cases in children less 3 years of age.
- Patients with type I FCD may or may not present with epilepsy while patients with type II FCD present refractory epilepsy and some require surgical treatment.
- Characteristic MRI findings of type II FCDs include the presence of: Subtle cortical and subcortical high signal intensity (On FLAIR images),
often located at the bottom of a deep sulcus,
blurred grey-white matter interface,
focal cortical thickening and transmantle sign (hyperintensity is seen extending from the subcortical area to the margin of the ventricle.)
- MR/FDG-PET fusion imaging and magnetic source imaging can help localize subtle architectural and signal abnormalities seen at MR imaging.
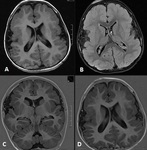
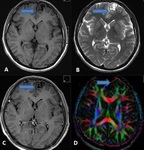
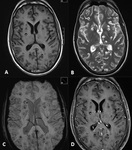
LISSENCEPHALY (Fig 7)
Classic (Type I) Lissencephaly
- Patients with classic lissencephaly may have a smooth brain surface in the complete form,
or more commonly,
they have a smooth surface with some gyral formation along the inferior frontal and temporal lobes in the incomplete form.
- MRI imaging show colpocephaly,
thickened cortex with broad,
flat gyri,
smooth grey-white matter interface,
straight oblique or shallow sylvian fissure,
giving a figure of "eight" or "hourglass" appearance.
Cobblestone (Type II) Lissencephaly (Congenital Muscular Dystrophy)
- Cobblestone lissencephaly is characterized by a nodular brain surface,
ocular anomalies and congenital muscular disorders.
- Cobblestone cortex results from over migration of theneuroblasts and glial cells beyond the external glial limitations into the subarachnoid space.
- MRI finding show thickened cortex has a polymicrogyric appearance association with hypomyelination of underlying white matter.
Key Points:
- Lissencephaly caused by the arrest of neuronal migration developing cerebral cortical thickening and smooth surface.
- Classified into 2 types: Classic (Type I) lissencephaly and Cobblestone (Type II) lissencephaly.
- MRI imaging in type I lissencephaly show colpocephaly,
thickened cortex with broad,
flat gyri,
smooth grey-white matter interface,
straight oblique or shallow sylvian fissure,
giving a figure of "eight" or "hourglass" appearance.
- MRI imaging in type II lissencephaly show thickened cortex has a polymicrogyric appearance association with hypomyelination of underlying white matter.
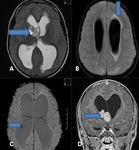
HETEROTOPIA (Fig.
8,9 & 10)
- Heterotopic Grey Matter results from an arrested migration of normal neurons along theradial path between the ventricular walls (ependyma) and the subcortical regions with size,
morphology and location variable.
- Periventricular / subependymal nodular HT (the most common).
Uni o multifocal asymmetric indentation.
- Band HT ("double cortex")/ Laminar.
Symmetric thick band of subcortical gray matter.
- Subcortical nodular HT : Isolated nodules.
Thin surface cortex often and "Mass" of white matter that continues the cortex and the ventricular surface.
- Periventricular and subcortical HT associated.
- CT and MRI show heterotopia similar to the cortex in density and signal intensity respectively.
The ipsilateral hemisphere is small and the lateral ventricle is usually largedue to underdevelopment of white matter.
Key Points:
- Heterotopic Grey Matter results from an arrested migration of normal neurons along the radial path between the ventricular walls (ependyma) and the subcortical regions.
- Broadly into two types: Nodular (Periventricular/ subependymal and subcortical) and Laminar.
- CT and MRI show heterotopia similar to the cortex in density and signal intensity respectively.
- Differential diagnosis of subependymal nodular heterotopia is subependymal nodules (SEN) seen in tuberous sclerosis.
In TS,
the nodules are irregularly shaped and often calcified on CT scan or shows blooming on SWI images.
On MRI images,
SEN are not precisely isointense with cortical grey matter and occasionally shows enhancement after contrast administration.
In contrast,
heterotopia,
parallel cortex in signal on all MR imaging sequences and do not enhance.
POLYMICROGYRIA (Fig.
11)
- Polymicrogyria is a malformation due to an alteration of the cortical development in the late stage of neuronal migration.
- Polymicrogyria (PMG) refers to a cerebral cortex with excessive number of abnormally small gyri that produce a characteristic “lumpy- bumpy” cortical surface,
and is probably one of the most common of the MCDs.
- Polymicrogyria may be associated with various genetic mutations,
metabolic disorders,
and congenital cytomegalovirus (CMV) infections.
- The frequency of epilepsy in PMG is 60% to 85% although seizure onset may not occur until the second decade,
however usually between the ages of 4 and 12.
- Convolutions formed are numerous,
small,
prominent and irregular.
T2-weighted MRIhas two patterns:
- <12 months: cortex with fine and small ripples,
normal thickened
- > 18 months: thick and irregular cortex,
increase of Virchow-Robin spaces and variable cortical invagination.
- There is a predilection for the posteriorperisylvian areas; when bilateral,
use to be syndromic (perisylvian bilateral congenitalsyndrome).
- Using CT and low field strength MRI,
PMG is difficult to discern and may only appear as thickened cortex.
- The only role for CT in the evaluation of PMG is to assess for evidence of calcification which is seen in PMG resulting from congenital CMV infection.
- On imaging,
cortex often appears mildly thickened (6 to 10 mm) on imaging due to cortical over folding rather than true cortical thickening,
T2 signal within the cortex is usually normal.
- Contrast-enhanced T1-weighted MRI: Prominence leptomeningeal veins can be seen soaring through abnormal cortical areas.
Key Points:
- Polymicrogyria (PMG) refers to a cerebral cortex with excessive number of abnormally small gyri that produce a characteristic “lumpy- bumpy” cortical surface,
and is probably one of the most common of the MCDs.
- There is a predilection for the posterior perisylvian areas; when bilateral,
use to be syndromic (perisylvian bilateral congenital syndrome).
- On imaging,
cortex often appears mildly thickened (6 to 10 mm) on imaging due to cortical over folding rather than true cortical thickening,
T2 signal within the cortex is usually normal.
- Contrast-enhanced T1-weighted MRI: Prominence leptomeningeal veins can be seen soaring through abnormal cortical areas.
SCHIZENCEPHALY (SPLIT BRAIN) (Fig.
12 & 13)
- Alteration in cortical organization characterized by the presence of clefts in the cerebral parenchyma,
which extend from the cortical surface to the ventricular system coating with dysplastic gray matter.
- Schizencephaly is a cleft lined with gray matter and connecting the subarachnoid CSF spaces with the ventricular system.
- Cleft filled with CSF and extending medially from the subarachnoid space into the lateral ventricle.
- The wall of the cleft is lined with dysmorphic gray matter.
The gray matter sometimes extends into the lateral ventricle in the form of subependymal heterotopias.
2 TYPES:
- Closed-lip schizencephaly (type 1) is characterized by gray matter–lined lips,
which are in contact with each other.
- Open-lip schizencephaly (type 2) has separated lips and a cleft of CSF extending to the underlying ventricles.
- MRI imaging differential diagnosis of schizencephaly is porencephalic cyst.
Because porencephalic cysts results from insults to otherwise normally developed brain,
the CSF space is lined by gliotic white matter,
not by dysplastic heterotopic cortex.
Key Points:
- Schizencephaly is a cleft lined with gray matter and connecting the subarachnoid CSF spaces with the ventricular system.
- Schizencephaly is of two types: Type I or closed lip schizencephaly characterized by gray matter–lined lips,
which are in contact with each other.
Type II or open lip schizencephaly has separated lips and a cleft of CSF extending to the underlying ventricles.
- MRI imaging differential diagnosis of schizencephaly is porencephalic cyst.
Because porencephalic cysts results from insults to otherwise normally developed brain,
the CSF space is lined by gliotic white matter,
not by dysplastic heterotopic cortex.
EPILEPTOGENIC TUMORS
- Neoplasms often manifest clinically as seizures,
and constitute 2-4% of epileptogenic substrates in the general epilepsy population.
- A particular subset of neoplasms referred to as epilepsy associated developmental tumors is cortical based.
These include -
- Ganglioglioma,
- Gangliocytoma,
- Desmoplastic infantile ganglioglioma (DIG),
- DNET,
and
- Pleomorphic xanthoastrocytoma (PXA).
- These entities represent World Health Organization (WHO) grade I benign/low-grade tumors
- These tumours share the following characteristics:
- They arise in a cortical location.
- Often located in the temporal lobe.
- Closely related to developmental malformations.
- Typically seen in adolescents and young adults.
- Characterized by a benign behavior,
a slow growth,
a sharp delineation and usually show absence of edema.
- Show signs of chronicity,
such as bone remodeling and scalloping of the adjacent skull.
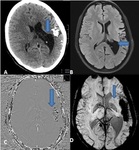
GANGLIOGLIOMA (Fig.
14)
Key Points:
- Ganglioglioma is the most common tumor associated with temporal lobe epilepsy.
- Common age: Children and young adult.
- Location: Majority are supratentorial.
Temporal lobe is most common site followed by frontal lobe.
- On CT scan,
classic appearance of a cyst with partially calcified mural nodule.
Because these tumors are slow growing,
peripherally located ganglioglioma may cause scalloped pressure erosion of the overlying calvarium.
- On MRI imaging,
well defined frontal or temporal lobe mass that is hypointense on T1W and hyperintense on T2 weighted sequences.
Enhancement varies from none to striking.
Nodular,
rim and solid enhancement patterns all occur.
- Calcification is common in ganglioglioma and is an important distinguishing factor from DNET and pleomorphic xanthoastrocytoma
DYSEMBRYOBLATIC NEUROEPITHELIAL TUMORS (DNET) (Fig.
15,
16)
Key Points:
- Dysembryoplastic neuroepithelial tumours (DNET) are benign (WHO Grade I) slow growing tumours arising from either cortical or deep grey matter.
- The vast majority are centred in cortical grey matter,
arise from secondary germinal layers and are frequently associated with cortical dysplasia (up to 80% of cases).
- They characteristically cause intractable partial seizure.
- Swollen gyrus
- Bubbly cystic appearance
- FLAIR image shows mixed signal intensity with bright rim sign
- May be wedge shaped and point towards the ventricle
- Usually no or only little enhancement
- Usually no vasogenic edema or diffusion restriction.
PLEOMORPHIC XANTHOASTROCYTOMA
Key Points:
- Pleomorphic xanthoastrocytoma (PXA) is a rare cause of temporal lobe epilepsy.
- Supratentorial cyst with enhancing mural nodule which abuts the peripheral meninges.
- Peritumoral edema may be seen in PXA,
while it is not a feature of either ganglioglioma or DNET.
- Thickening and enhancement of the adjacent leptomeninges is highly characteristic but is not always present.
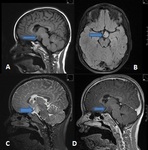
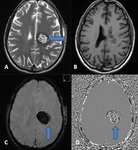
HYPOTHALAMIC HAMARTOMA (Fig.
17)
- Hypothalamic hamartoma is another neoplasm that is typically associated with gelastic epilepsy in childhood.
- It usually has similar signal intensity to gray matter on all pulse sequences and typically does not enhance after administration of intravenous contrast.
- Epileptogenic hypothalamic hamartoma always affects the mammillary bodies and also often the tuber cinereum.
GLIOSIS
- Gliosis is the end state of several injuries that can affect the central nervous system,
mainly: trauma,
infection and stroke.
- The findings are nonspecific at MRI although usually hyper-signal areas often are present in long TR sequences,
associated with loss of volume sings (such as grooves widening and ventricular retraction) and encephalomalacic areas.
NEUROCUTANEOUS SYNDROME
TUBEROUS SCLEROSIS (Fig.
18,
19)
- Autosomal dominant
- Characterized by
cortical tubers,
subependymal nodules,
and
Subependymal giant cell astrocytomas.
White matter abnormalities.
- Tuberous sclerosis was classically described as presenting in childhood with a triad (Vogt triad) of:
Seizures
Mental retardation
Adenoma sebaceum
Cortical tubers:
- Cortical hamartoma or tubers are most characteristic lesion of TS.
Detected on MRI in 95% of patients.
In MRI studies before 6 months of life tubers appear as cortical regions hyperintense on T1 and hypointense on T2,
and after 6 months the signal inverts.
- Enhancement following contrast administration occurs in less than 5% of patients and does not indicate neoplasia because cortical tubers do not undergo malignant degeneration.
FLAIR sequence becomes more positive with age.
- Tuberous sclerosis patients with medically refractory epilepsy usually have multiple tubers.
- Therefore,
it is crucial to identify precisely which of the tubers are responsible for epileptogenic activity.
This is not possible with MR imaging alone.
- More recently,
MR/FDG-PET fusion imaging and magnetic source imaging have shown promise in the improved localization of epileptogenic foci associated with tuberous sclerosis.
White matter lesions:
- Four distinct patterns of white matter lesions on MRI imaging are seen: straight or curvilinear bands that extend from the ventricle through the cerebrum towards the cortex,
wedge shaped lesion,
non-specific tumefactive or conglomerate foci,
cerebellar radial bands.
- These are histologically benign and represent dysplastic white matter with lines of migration disorder.
- MRI signal are same as shows by cortical tubers according to the age and approximately 12% of white matter lesions shows enhancement after contrast administration.
Subependymal Nodules (SEN):
- Subependymal hamartomas are found in 95% of patients with tuberous sclerosis.
- Most common location is near the caudate nucleus along the striothalamic groove of the lateral ventricle just behind the foramen of monro.
Less common sites are within the lateral ventricle include the atria and temporal horns.
- On MRI,
SEN appear as irregular nodules jutting into the CSF filled ventricles.
Signal is variable but is usually hypointense to white matter on T2W because of calcification.
On GRE images,
these nodules show blooming.
Subependymal giant cell astrocytoma (SGCA): (Fig 19)
- SGCA appear to be located at or near the foramen of monro.
- SGCA is frequently calcified so appear heterogenous on MRI.
- Shows intense but inhomogeneous enhancement.
- SGCA enlarged with time but histologically benign.
- Associated with obstructive hydrocephalus and common cause of symptoms in SGCA.
Key Points:
- TS is autosomal dominant disorder characterize by cortical tubers,
subependymal nodules,
Subependymal giant cell astrocytomas,
White matter abnormalities.
- Tuberous sclerosis was classically described as presenting in childhood with a triad (Vogt triad) of Seizures,
Mental retardation,
Adenoma sebaceum.
- Cortical hamartoma or tubers are most characteristic lesion of TS.
Detected on MRI in 95% of patients.
In MRI studies before 6 months of life tubers appear as cortical regions hyperintense on T1 and hypointense on T2,
and after 6 months the signal inverts.
Enhancement following contrast administration occurs in less than 5% of patients.
- Subependymal nodules most commonly located near the caudate nucleus along the striothalamic groove of the lateral ventricle just behind the foramen of monro and On MRI,
SEN appear as irregular nodules jutting into the CSF filled ventricles.
Signal is variable but is usually hypointense to white matter on T2W because of calcification.
On GRE images,
these nodules show blooming.
- SGCA appear to be located at or near the foramen of monro and frequently calcified so appear heterogenous on MRI with intense but inhomogeneous enhancement.
STRUGE WEBER SYNDROME (Fig 20,
21)
Key Points:
- SWS is a rare phakomatosis characterized by facial port wine stains and pial angiomas.
- It is a vascular malformation with capillary venous angiomas in the face (port-wine stain),choroid of the eye and leptomeninges.
- Venous occlusion and ischemia lead to angiomatosis with cortical calcium deposition and atrophy.
- It is clinically characterized by epilepsy,
progressive mental retardation,
and facial telangiectatic nevi.
- Tram-track' subcortical calcification
- Calvarial and regional sinus enlargement may be evident
- Ipsilateral choroid plexus may be enlarged
- Gyriform enhancement after the administration of iodinated contrast material, reflecting pial angiomatosis
- Hemiatrophy of brain.
PERIVENTRICULAR LEUKOMALACIA (Fig 22)
Key Points:
- Is a white matter injury of prematurity affecting the periventricular zones,
and typically results in cavitation and periventricular cyst formation.
- PVL is most common in premature neonates.
- Manifests as cerebral palsy (> 50%),
intellectual disability or visual disturbance.
- Irregular margins of the bodies and trigones of the lateral ventricles,
- Loss of periventricular white matter with cyst formation,
and
- Thinning of the corpus callosum.
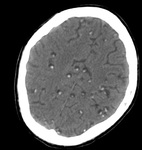
VASCULAR MALFORMATION
Cavernoma (Fig.
23)
Key Points:
- Cavernoma is also known as cavernous malformation or cavernous angioma.
It is a benign low flow vascular malformation with a tendency to bleed.
75 percent occur as solitary sporadic lesions and 10-30 percent occurs as multiple lesions.
- MRI is the modality of choice,
demonstrating a characteristic "popcorn" or "berry" appearance with a rim of signal loss due to hemosiderin,
which demonstrates prominent blooming on susceptibility weighted sequences.
- T1 and T2 signal is varied internally depending on the age of the blood produces and small fluid fluid levels may be evident.
- Gradient Echo or T2* sequences are able to delineate these lesions better than T1 or T2 weighted images.
- In patients with familial or multiple cavernous angiomas GRE T2* sequences are very important in identifying the number of lesions missed by conventional Spin echo sequences.
- The lesions generally do not enhance,
although enhancement is possible.
GRANULOMATOUS INFECTIONS
NEUROCYSTICERCOSIS (NCC) (Fig.
24,
25)
- NCC is caused by the CNS infection with the pork tapeworm Taenia solium which is endemic in most low-income countries where pigs are raised.
- Location – Subarachnoid space – most common location,
parenchyma,
Basal cisterns,
Ventricles.
- Four main stages
- Vesicular: viable parasite with intact membrane and therefore no host reaction.
- Colloidal vesicular: parasite dies within 4-5 years untreated,
or earlier with treatment and the cyst fluid becomes turbid.
As the membrane becomes leaky edema surrounds the cyst.
This is the most symptomatic stage.
- Granular nodular: edema decreases as the cyst retracts further; enhancement persists.
- Nodular calcified: end-stage quiescent calcified cyst remnant; no edema.
IMAGING FEATURES:
- Subarachnoid/intraventricular:
- When in the subarachnoid space/intraventricular,
the cysts typically do not have a visible scolex.
- In the basal cisterns they can be grape-like (racemose).
The cysts are typically 1-2 cm in diameter.
- Usually the cysts are similar in signal intensity to CSF,
although occasionally cyst fluid may somewhat differ.
- In the ventricles,
there is often (79%) 2 associated ventriculitis often leading toaqueductal stenosis and hydrocephalus.
- Parenchymal: Parenchymal cysts usually involve the grey-white matter junction
- Vesicular Stage: On MRI imaging,
cyst shows CSF signal intensity with sometimes hyper intense scolex is seen on T1W image.
Typically,
no enhancement is seen,
however very faint enhancement of the wall and enhancement of the scolex may be seen.
- Colloidal vesicular: Fluid in cyst become turbid in this stage and show hyperintensity on T1W images relative to CSF with surrounding oedema.
Wall of the cyst become thick and shows enhancement on contrast administration.
Scolex can often still be seen as an eccentric focus of enhancement.
- Granular nodular: Edema decrease in this stage and cyst retracts.
Enhancement persist but is less marked.
- Nodular calcified: End-stage quiescent calcified cyst remnant without any edema and no enhancement on post contrast images.
Signal drop out on T2 and T2* sequences.
Key Points:
- NCC is caused by the CNS infection with the pork tapeworm Taenia solium which is endemic in most low-income countries.
- Location – Subarachnoid space – most common location,
parenchyma,
Basal cisterns,
Ventricles.
- Four main stages: Vesicular,
colloidal vesicular,
granular nodular and calcified nodular.
- Different stage show different imaging features,
like in vesicular stage,
cyst shows CSF signal intensity with sometimes hyper intense scolex is seen on T1W image and no enhancement is seen.
Wall enhancement is seen in colloidal vesicular stage and scolex can often still be seen as an eccentric focus of enhancement.
In granular nodular stage,
edema decrease and cyst retracts.
Enhancement persists but is less marked.
In nodular calcified,
end-stage quiescent calcified cyst remnant without any edema and no enhancement on post contrast images.
Signal drop out on T2 and T2* sequences.
TUBERCULOMA (Fig.26)
- Tuberculosis (TB),
caused by Mycobacterium tuberculosis,
accounts for eight million annual,
worldwide deaths.
- Involvement of the central nervous system (CNS) is one of the most serious forms of this infection and is responsible for a high mortality and morbidity.
- Brain tuberculoma,
- A space-occupying mass of granulomatous tissue
- May be single or multiple
- Children develop infratentorial tuberculomas more commonly than do adults.
- Symptoms are often limited to seizures and mass effect.
- IMAGING OF TUBERCULOMAS – Depending on its stage of maturation,
a tuberculomas appearance varies on MRI,
i.e.,
whether
- Noncaseating - hyperintense on T2W and slightly hypointense on T1W images,
and show homogenous enhancement after injection of paramagnetic contrast on T1W images.
- Caseating with a solid center - iso- to hypointense on both T1W and T2W images with an iso- to hyperintense rim on T2W images and a rim enhancement.
- Caseating with a liquid center- When the solid center of the caseating lesion liquefies,
the center appears hyperintense with a hypointense rim on T2W images.
- Diffusion weighted imaging (DWI) shows restriction in tuberculomas with liquid necrosis,
whereas there is no such restriction of diffusion in lesions with solid caseation.
- When combined with spectroscopic imaging,
in vivo spectra are found to be specific for intracranial tuberculomas (lipid peak)
HYPOXIC ENCEPHALOPATHY (Fig 27)
- Can occur at any age and often has devastating neurological sequalae.
- In older children,
it usually occurs due to drowning or asphyxiation whereas cardiac arrest or cerebrovascular disease with secondary hypoxemia is common in adults.
- Hypoxic injury primarily affects the grey matter structures like basal ganglia,
thalami,
cerebral cortex (sensorimotor and visual cortices),
cerebellum and hippocampi.
Parasagittal infarction involving the deep white matter only at border zones of major arterial territories (water shed) can also be seen.
- Diffusion-weighted MR imaging is the earliest imaging modality showing restriction in the basal ganglia,
cerebral cortex in particular,
the perirolandic and occipital cortices usually in the first 24 hours.
Diffusion-weighted imaging abnormalities usually pseudo-normalize by the end of the 1st week.
- In the early subacute period (24 hours-2 weeks) conventional T2-weighted images typically become positive and show hyperintensity of the injured gray matter structures.
T1 hyperintensities indicating cortical laminar necrosis become evident after two weeks.
This hyperintense signal does not represent haemorrhage,
and it is believed to be caused by the accumulation of denatured proteins in dying cells.
This hyperintensity can also be seen within a few days on FLAIR.
Key Points
- Diffusion-weighted MR imaging is the earliest imaging modality to become positive.
- Diffusion restriction seen in cerebral cortex in particular,
the
- perirolandic and occipital cortices,
basal ganglia,
hippocampi and
- cerebellar hemispheres.
Diffusion-weighted imaging abnormalities
- usually pseudonormalize by the end of the 1st week.
- Parasagittal infarction involving the deep white matter only at border zones of major arterial territories (water shed) can also be seen.
Miscellaneous
- There are many other diseases that can cause seizures.
Review all would go beyond the aim of this paper but before the end should note a few:
- Rasmussen's encephalitis (Fig 28): is a chronic inflammatory disease of unknown origin,
usually affecting one brain hemisphere.
Most cases (85% cases) occur in children under the age of 10 years.
- On MRI,
unilateral cortical atrophy with ex-vacuo ventricular dilatation is seen on all pulse sequences and hyperintense signal areas in the affected hemisphere on T2/FLAIR sequences.
Mostly no diffusion restriction is seen,
however sometimes,
restricted diffusion may be seen in altered signal areas.
No significant post-contrast enhancement is seen.
- Parenchymal infectious processes (encephalitis,
abscesses) with or without meningeal involvement: may present with seizures.