MRI,
CT and Doppler ultrasound can be useful in detecting these pathologies by identifying the most common signs.
Sonography usually shows PVT as a hyperechoic lesion within the lumen of the portal vein.
However,
clots exhibit variable echogenicity and may,
if recently formed,
be hypoechoic or anechoic.
PVT eliminates the venous flow signal normally obtained from the lumen of the portal vein during either pulsed duplex or color flow Doppler imaging (Fig.
3) [3].
CTPV is seen as a mass of tubular anechoic collaterals within an enlarged hyperechoic porta hepatis.
Normal portal venous landmarks are obliterated.
Doppler sonograms of these collaterals show a slightly turbulent low-velocity venous signal,
with little or absent respiratory and cardiac variation [3].
Vessels constituting the CTPV have a hepatopetal flow and this fact supports the theory that cavernous transformation forms as a bypass route (or porto-portal collateral channels) between the splanchnic veins around the obstructed portal vein and intrahepatic portal veins.
Blood flow in the hepatic artery increases,
probably as a response to decreased blood flow in the intrahepatic portal veins.
The estimation of arterial flow volume by Doppler techniques is difficult,
because vessel beam angles and vessel caliber cannot be readily calculated.
The diagnosis of increased hepatic arterial Doppler shifts is therefore a subjective parameter.
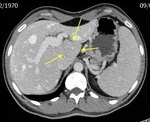
Non-contrast CT scans are usually unable to demonstrate the thrombus,
with the exception of some acute cases where the thrombus is hyperdense (Fig.
4).
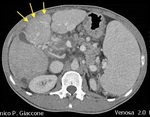
The occlusion of a branch of the portal vein by a thrombus can manifest as transient hepatic attenuation differences (THAD) in the arterial/early portal phase,
showing increased enhancement (i.e.
perfusion) of the lobe or segment previously supplied by the vein due to hepatic arterial compensatory flow.
When blockade takes place at the level of the portal trunk (before the sinusoids),
as in PVT,
or before the central lobular vein (into sinusoids),
as in cirrhosis,
portal flow remains adequate for the central zones of the liver,
but not for the peripheral ones.
The arterial response,
based on the activation of the peribiliary plexus,
produces enhancement of the peripheral subcapsular hepatic parenchyma,
with relative hypodensity of the central perihilar area.
The consequent pattern on CT scan is called a “central–peripheral phenomenon” (Fig.
5) [4].
The atrophy-hypertrophy complex is frequently observed in patients with CTPV.
Some findings,
such as hypertrophy of the caudate lobe,
may mimic chronic liver disease or signs of portal hypertension,
but left lateral segment atrophy and a normal or enlarged segment IV are distinctive findings of cavernous transformation.
Morphologic changes in the liver reveal that the central part of it (ie,
segment IV and caudate lobe) becomes enlarged,
whereas the peripheral part (ie,
left lateral segment and/or right liver lobe) becomes smaller.
The reason why the right liver lobe and the left lateral segment become atrophic with CTPV cannot be elucidated on the basis of our current study findings.
It can be supposed that their location,
far from the CTPV,
leads to compromised portal venous flow.
Similarly,
it can be supposed that the reason why segment IV and the caudate lobe become hypertrophic with CTPV is related to their location next to the cavernous transformation,
which results in maintained portal inflow (Fig.
6,
Fig.
7) [5].
On CT imaging,
three quantitative axis measurements may be obtained: measurement of the caudate lobe and the caudate lobe–to–right lobe ratio (CL/RL),
the transverse diameter of liver segment IV and the longitudinal axis of the spleen [5].
Portal biliopathy is defined as biliary changes in patients with an extrahepatic portal vein obstruction causing CTPV.
The mechanisms of biliary abnormalities in extrahepatic portal vein obstruction are both extrinsic compression by collaterals and peribiliary fibrosis resulting from ischemic or inflammatory changes underlying portal thrombosis.
In cases of reversible biliary change after shunt surgery,
mechanical compression by collaterals is the mechanism behind biliary abnormalities in extrahepatic portal vein obstruction.
In cases of persistent biliary abnormalities,
ischemia or fibrous scarring of the bile duct is considered the major cause of biliary abnormalities.
Various biliary abnormalities are considered in portal bioliopaty,
such as wall thickening of the duct and delayed and prolonged enhancement of the thickened wall; these conditions are classified as varicoid,
fibrotic or mixed type on the basis of the presence or absence of structure and on their pathogenesis.
In the varicoid type,
the irregular contour of the bile duct was primarily caused by multiple smooth extrinsic compressions of the cavernoma.
In the fibrotic type,
MR imaging shows localized strictures with proximal duct dilatation.
The structures were primarily caused by fibrous scarring related to chronic inflammation and ischemic injury (Fig.
8,
Fig.
9,
Fig.
10,
Fig.
11) [4].
MR cholangiography,
coupled with dynamic 3D gradient-echo imaging,
offers the advantages of being a non-invasive technique,
useful for providing all the diagnostic components of portal biliopathy (including extrahepatic portal vein obstruction,
portal cavernoma,
gallbladder varices and bile duct abnormalities) and delineating the extent and severity of bile duct structure.
Spin-echo MR images usually show PVT as an area of abnormal signal within the lumen of the portal vein.
PVT usually appears isointense on T1-weighted images,
but it may be hyperintense if recently formed.
PVT typically has a more intense signal on T2-weighted images.
On gradient-echo MR images,
PVT is usually seen as an area of diminished intravascular signal with an intensity similar to that of adjacent stationary soft tissues.
Gradient-echo images can show PVT more clearly,
but may provide less anatomic resolution and liver-lesions contrast (Fig.
12) [3].
Portal branch occlusion and segmental cholestasis are two causes of focal parenchyma fibrosis and atrophy associated with liver capsular retraction.
From a technical point of view,
the most efficient sequence for the detection of hepatic capsular retraction in magnetic resonance imaging (MRI) is a T2-weighted (T2W) spin-echo sequence,
preferably without fat saturation,
to benefit a better contrast between intra-abdominal fat and hepatic parenchyma.
T1-weighted (T1W) sequences before and after gadolinium injection play a key role in the differential diagnosis of the capsular retraction (Fig.
13) [6].