I.
Various Tools for Assessment and Prediction of PH
1.
Gold standard methods
Measurement of hepatic venous pressure gradient (HVPG) and upper GI endoscopy are considered the gold standards for PH assessment in patients with cirrhosis.
| Hepatic venous pressure gradient (HVPG) |
HVPG is calculated by subtracting the free hepatic venous pressure (FHVP) from the wedged hepatic venous pressure (WHVP) and these values are obtained by hepatic vein catheterization[2].
- FHVP is determined by direct measurement of hepatic venous pressure
- WHVP is typically obtained by balloon occlusion of the hepatic vein,
though it can also be estimated by wedging the catheter in the end tributaries of a hepatic vein.
HVPG is an accurate and reproducible way of measuring portal pressure in patients with cirrhosis of any etiology.
Also,
it is the best surrogate indicator of prognosis in patients with cirrhosis.
The higher the HVPG,
the greater risk of varices,
clinical decompensation and development of hepatocellular carcinoma[3].
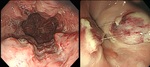
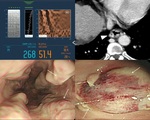
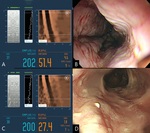
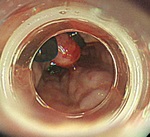
Upper GI endoscopy is the best way of assessing the presence of esophageal varices.
It detects indicators of increased bleeding risk,
the red color signs and "wale" marks (Fig.
3).
Both HVPG measurements and upper GI endoscopy are safe but invasive diagnostic techniques that carry patient discomfort,
increase the burden for medical providers and increase the cost of medical care.
Because of this,
there is a clinical need for non-invasive ways of assessing portal hypertensive patients that could substitute for the need of hemodynamic measurements and endoscopy.
2.
Non-invasive tools; ultrasonography (US) based
Ultrasonography is a non-invasive,
economical,
and effective method for assessing portal hypertension[4].
Let me introduce three methods for assessement and prediction of PH:
- B-mode / spectral and color doppler study
- Transient elastography (TE)
- Shear wave elastography (SWE)
| B-mode / spectral and color doppler study |
1.
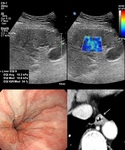
The diameter of the main portal vein (MPV)
The normal diameter of MPV ranges from 6.4mm to 12.1mm.
The mean diameter of MPV in patients with liver cirrhosis is about 12mm,
but it varies depending on respiration (an increase of 20-100% in diameter during deep inspiration,
upto 13-16mm).
How to measure the diameter of PV[5]:
- Patient's condition: in supine position,
quiet repiration,
and fasting ≥ 4 hours
- measrement point: where the PV crosses the IVC
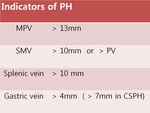
If the diameter of MPV is 13mm or more,
the specificity to PH is 100%,
and the sensitivity 45-50%.
The sensitivity may be increased upto 81% by measuring splenic and superior mesenteric vein diameters.
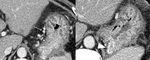
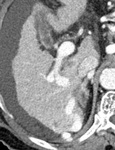
Other indicators of PH includes splenic vein,
SMV,
and gastric vein (Fig.
4,5).
The differential diagnosis of dilated portal vein includes:
- Portal hypertensive splenomegaly
- Acute portal vein thrombosis (PVT)
- Postprandial increase in portal vein diameter.
2.
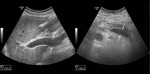
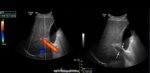
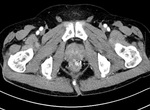
Portal flow direction and velocicy
The normal direction of portal venous flow is hepatopetal (toward the liver) during the entire cardiac cycle and the mean velocity ranges from 15 to 18cm/s.
However,
as portal venous pressure increases,
the direction of flow reverses (hepatofugal; away from the liver),
and blood flow velocity decreases (<16.5cm/s) (Fig.
6).
The differential diagnosis of hepatofugal portal veous flow includes:
- Eating
- Use of drgus that increase portal flow
- Budd-Chiari syndrome (BCS)
- Side-to-side portocaval shunts
- Surgical or spontaneous splenorenal shunts with cirrhosis
- Tricuspid regurgitation (tricuspid flow reversal)
3.
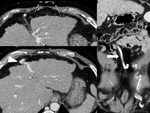
Changes in hepatic vein (HV) and hepatic artery (HA)[6]
The normal waveform within the HV is triphasic but it changes into monophasic waveform in liver cirrhosis.
In addition,
resistive index (RI) and flow velocity of HA increase.
If the RI of HA is 0.78 or more,
it can be regarded as PH,
and the specificity is high but the sensitivity is low.
| Transient Elastography (TE) |
TE is a relatively new sonographic technique for evaluating of liver stiffness.
Several studies have demonstrated that there is a direct correlation between liver stiffness and HVPG or esophageal varices.
1. Assessment of PH
The first study in which PH was assessed using TE was done in 2006 with 124 patients with hepatitis C virus (HCV). There was a close direct correlation between liver stiffness and HVPG (Pearson correlation,
0.84; P < 0.001) and the Area Under the Receiver Operating Characteristic (AUROC) for CSPH was 0.94[7].
2.
Assessment of esophageal varices
There are also studies regarding diagnostic performance of TE for the presence of esophageal varices.
Liver stiffness values obtained by TE directly correlated with the presence of esophageal varices and AUROC ranged from 0.74 to 0.85 with cut-off values from 13.9-21.5 kPa with a sensiticity and specificity of 76-100% and 32-85%[8].
(Fig.
7,
8).
There was a meta-analysis of diagnostic accuracy in valuation of portal hypertension in chronic liver disease using TE,
and the results demonstrated TE as a good screening tool for significant portal hypertension,
but only moderate diagnostic utility for the prediction of esophageal varices[9].
| Shear Wave Elastography (SWE) |
SWE uses measurement of acoustically generated tissue shear wave propagation speeds to derive estimates of liver stiffness,
with the advantage of simultaneous anatomic B-mode US imaging.
This allows selection of a liver parenchymal region of interest devoid of blood vessels or focal lesions for analysis[10].
There are two types of SWE,
point SWE (ARFI; acoustic radiation force impulse) and real-time SWE (RT-SWE).
There was a study regarding diagnostic performance of liver stiffness by using ARFI in 135 patients with hepatitis C virus.
In the study,
AUROC values for the presence of esophageal varices and high risk esophageal varices was 0.890 and 0.868 with cut-off values 2.05m/s and 2.39m/s[10].
Two other studies using RT-SWE also reported the direct correlation between the liver stiffness measurement and PH and the cut-off values were 15.2 kPa and 15.4 kPa[11,
12] (Fig.
9).
Another study regarding RT-SWE as a monitoring tool of PH was done in patients with cirrhosis and portal hypertension.
It demonstrated strong correlation between ΔLiver Stiffness (LS) and ΔHVPG,
and AUROC of ΔLS for predicting clinical improvement and clinical aggravation were 0.794 and 0.925,
respectively.
That is, estimating ΔLS by using SWE may be useful in patients with cirrhosis and portal hypertension[13].
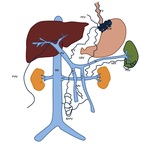
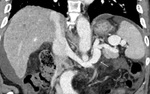
II. Varices in Portal Hypertension
The portosystemic collateral channels that can develop in portal hypertension are numerous,
widespread,
and varied in appearance (Fig.
10).
On CT scans,
varices appear as well-defined round,
tubular,
or serpentine structures that are smooth,
have homogeneous attenuation,
and enhance with contrast material to the same degree as adjacent vessels.
The most common portosystemic collateral channels are coronary venous collateral vessels in the lesser omentum,
seen in 80% of cases[14].
1.
Gastric & paragastric varices (Fig.
11).
Coronary varices,
left gastric varices,
and paragastric varices are the same.
Retrogastric varices and gastric varices are used to mean the same thing.
- Afferent vein: left gastric vein,
posterior gastric vein,
short gastric vein
- Efferent vein: paraesophageal varices,
gastrorenal shunt
2.
Esophageal & paraesophageal varices (Fig.
12).
Backflow of the left gastric vein (coronary vein) results in paraesophageal varices and esophageal varices.
- Afferent vein: left gastric vein
- Efferent vein: Azygos vein,
IVC,
pulmonary or brachiocephalic veins
3.
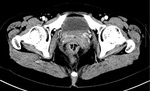
Rectal & pararectal varices (Fig.
13).
- Afferent vein: superior rectal vein of IMV
- Efferent vein: recto-genital shunt,
inter-rectal shunt,
middle and inferior rectal veins,
internal iliac vein
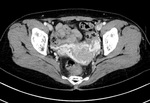
4.
Paraumbilical veins & abdomnial wall collaterals (Fig.
14).
- Afferent vein: umbilical portion of the left portal vein
- Efferent vein: inferior epigastric vein,
iliac vein inferiorly,
and substernal / internal mammary veins,
intercostal veins,
azygos veins superiorly
5.
Splenorenal shunt (Fig.
15).
The reflux flow from the splenic veins enters the renal vein,
then into the lumbar veins and into the IVC.
Splenorenal shunt is suggestive of high risk of hepatic encephalopahty.
6.
Other collaterals
- Right posterior portal branch-IVC collateral (Fig.
16)
- Utero-vaginal collaterals (Fig.
17)
- Adnexal collaterals (Fig.
18)
III.
Management of Complications of Portal Hypertension
1.
Pharmacologic therapy for PH[15]
- Beta-blockers (e.g.
propranolol,
nadolol)
- Statin (e.g.
simvastatin)
- Low-sodium diet and diuretics in cases with liver cirrhosis and asictes
2.
Endoscopic procedures for varices
- Endoscopic variceal ligation (EVL): 1st line therapy for esophageal varices
- Sclerotherapy: 1st line theraphy for gastric varices
3.
Interventional procedures
- Transjugular intrahepatic portosystemic shunt (TIPS)
- Balloon-occluded retrograde transvenous obliteration (BRTO)
- Plug-assised retrograde transvenous obliteration (PARTO)
Interventional procedures are recommened for selected high-risk patients[16]:
- failed the pharmacologic and/or endoscopic therapy
- had a single episode of clinically significant rebleeding (transfusion requirement > 2units of blood within 24hours,
a systolic blood pressure < 100mmHg or a postural change of > 20mmHg and/or pulse rate > 100bpm)
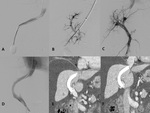
| Transjugular Intrahepatic Portosystemic Shunt (TIPS) |
TIPS is an established procedure that has proven benefit in the treatment of patients who have complications of portal hypertension,
such as variceal bleeding and ascites.
TIPS is a percutaneous imaging-guided procedure in which a channel is constructed within the liver with the intent of reducing portal pressure by diverting blood from the portal to the systemic circulation.
TIPS reduces the portosystemic pressure gradient by functioning as a side-to-side portacaval shunt[17] (Fig.
20).
But the TIPS procedures may lead to a number of potentially significant adverse events[18].
- Technical complications: transcapsular puncture (up to 33%),
hemobilia,
stent migration
- Hepatic encephalopathy due to diversion of portal venous flow,
which diminishes metabolic filtering effect of the hepatic parenchyma (in 30-46% of patients)
- Deterioration of hepatic fuction
- Hepatorenal syndrome
- TIPS stenosis and occlusion: most common at the hepatic venous end
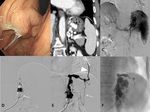
| Balloon-Occluded Retrograde Transvenous Obliteration (BRTO)[19] |
BRTO is an endovascular technique that was refined in Japan as a therapeutic adjunct or alternavite to TIPS in the management of gastric varices. It is also an effective therapy for sclerosis of de novo portosystemic shunts complicated by hepatic encephalopathy.
A BRTO procedure involves occlusion of outflow veins of the portosystemic shunt,
such as a gastrorenal shunt,
using an occlusion balloon followed by the injection of a sclerosing agent directly into the varix endovascularly (Fig.
21).
Critical to the procedure is the stagnation of the sclerosant within the varix or shunt without reflux into either the portal or systemic vasculature,
which might result in serious complications.
Complications of BRTO includes:
- Balloon rupture during the procedure,
resulting in pulmonary edema,
treatment failure,
recurrent variceal bleeding
- Complications associated with the use of liquid and foam sclerosants,
including pulmonary edema,
cardiogenic shock,
renal failure etc.
- Worsening of esophageal varices due to enhanced portal flow to the liver following BRTO (up to 40% of patients)
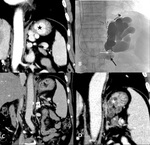
| Plug-Assised Retrograde Transvenous Obliteration (PARTO)[20] |
PARTO is a modified BRTO technique where a balloon occlusion catheter and sclerosants were replaced with a vascular plug/coils and gelatin sponge to minimize some of the complications and logistical issues associated with the balloon catheter (Fig.
22).
Compared with BRTO,
PARTO has several advantages:
- Prevents procedure-related complications
- Decreases the procedure time as it does not require an indwelling balloon catheter and sclerosing agents
- Does not require selective embolization of efferent veins in most cases as it does not require sclerosing agents