AUTOINMUNE DEMYELINATION
Inmune-mediated events can cause myelin loss and oligodendrocyte death leading to axonal degeneration,
this complex phatology remains unclear: some investigations theorice about genetically susceptible individuals and if environmental factors can contribute to triggered the disease [1][2].
Neuromyelitis optica (NMO):
The mean age at presentation is in the fourth decade of life,
with a stronger female predilection (F:M ratio 3:1) and more frequently found in caucasian patients.The classic presentarion is optic neuritis with WM nonspecific lesions and longitudinally extensive myelitis,
with a relapsing course in 90% of patients[1][2][3].
CT:
NECT: initial CT normal.
MRI:
T1: hypointense patchy lesions in WM.
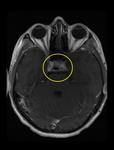
T1 C+: open ring enhacement in acute spinal cord lesions (Fig.
1).
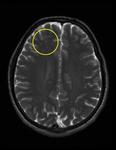
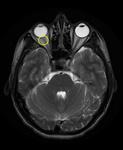
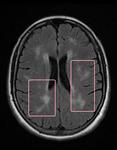
T2: hyperintense “bright spotty lesions” in WM (Fig.
2-3).
STIR: hyperintense optic nerve and spinal cord.
Acute disseminated encephalomyelitis (ADEM):
Demylination of WM para or post infectious or vaccination.
The patients are usually pediatric age range and the gender predominance is slight male[1][2][4].
CT:
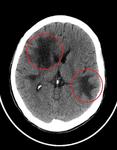
NECT: low density WM lesions (Fig.
4).
CECT: ring-enhacing WM lesions.
MRI:
T1 C+: punctate,
ring,
arc or peripheral enhancement (Fig.
5).
T2: hyperintensities in thalami,
brainsteam and posterior fossa with surrounding edema (Fig.
6-8).
DWI: variably,
only peripherical restriction.
INFECTIVE DEMYELINATION
Immunocompromised patiens are more likely to develop these diseases,
the depletion of cellular immunity helps to acquire or reactive infections that damage brain cells: the infectious agent affects olygodendrocytes and causes demyelination[1][2].
HIV encephalopaty:
Is common that patients with HIV developed a neurological clinical syndrome,
between 50-60% of all AIDS patients have neurological manifestations[1][2][5].
CT:
NECT: global atrophy and patchy WM hypodensitity progressive to confluent.
MRI:
T2: deep WM and periventricular symmetric hyperintensity,
can affect posterior fossa too,
whithout mass effect or enhancement (Fig 9-12).
Progressive Multifocal Leukoencephalopathy:
This demyelinating disease is an opportunistic infection caused by the JC virus.
More than 85% of the adult population has antibodies against JC virus with asymptomatic infection,
the most common predisposition to develop a symptomatic state is the inmmunosupression [1][2][6].
CT:
NECT: WM assymetrical multifocal low attenuations,
periventricular and subcortical (Fig 13).
MRI:
T1: hypointense focal WM regions (Fig.
14-15).
T2: hyperintense focal WM regions (Fig.
16-17).
DWI: peripheral patchy diffusion restriction (Fig.
18-19).
VASCULAR-INFLAMMATORY DEMYELINATION
Vasculopathy is very prevalent and one of the major indications for neuroimaging,
the spectrum of disease is huge: we can classify these affections in atherosclerosis and arteriolosclerosis (include different modalities of stroke,
the most serious and disabling manifestations of vessel disease) and nonatheromatous vascular diseases (cause stroke or stroke-like lesions) [1][2].
Small-vessel disease:
It is a very common disease,
in elderly patients almost a phisiological state.
The pathology is multifactorial,
most common in hypertension,
diabetes,
smoking...
all predisposing factors to develop sclerosis of arterioles[1][2][4].
CT:
NECT: WM periventricular regions of low atenuation.
MRI:
T2: confluent hyperintensity,
symetric and bilatarel affection of the WM with a vascular distribution (Fig.
20-22).
Subacute cerebral infarction:
Cerebral stroke is the first cause of acquired disability worldwide,
and the second cause of death.The cerebral vessels obstruction of blood flow ends up with focal brain necrosis.
Between 1 to 3 weeks following the schemic evente,
the infarction is demnominated subacute [1][2][7].
CT:
NECT: low attenuation foci with mass effect (cytotoxic edema) and a vascular distribution involving gray and WM (Fig.
23).
CECT: patchy enhancement,
hemorrhagic foci can mimic this found.
MRI:
T1: hypointense regions of edema with mass effect (Fig.
24).
T2/FLAIR: hyperintense regions of edema with mass effect (Fig.
25-26).
DWI: elevated diffusion restriction (Fig.
27).
Cerebral amyloid angiopathy:
Cerebrovascular disease caused by the deposition of cerebral β-amyloid in the tunica media and adventitia of small and medium-sized vessels of leptomeninges,
cerebral cortex and subcortex.
Exist both forms: sporadic (young ages) and hereditary (found in 30% of normal elderly autopsy).
Is also common in patients with Alzheimer disease and correlated with Down syndrome [1][2][8].
CT:
NECT: subaracnoid hemorrhage and WM diffuse low density (Fig.
28).
MRI:
T1: lobar cerebral hemorrhage.
T2: acute hematomas and coalescent WM hyperintensity (Fig.
29).
T2* GRE: small focal lesions “black dots” (Fig.
30).
Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephaplopathy (CADASIL):
CADASIL is a cerebral autosomal dominant arteriopathy,
a mutation in diferent NOTCH3 exons.
This disease cuases subcortical infarcts and leukoencephalopathy,
the reason is the accumulation of granular osmiophilic material in arteries and arterioles [1][2][9].
CT:
NECT: subcortical low density lesions (Fig.
31).
MRI:
T1: hypointense large lesions in WM and small confluent lesions in the cortex.
T2: WM hyperintensities well-delineated (stroke) and diffuse (leukoariosis) (Fig.
32-35).
TOXIC-METABOLIC DEMYELINATION
A lot of toxins can affect the brain (accidentally encountered,
drug abuse or administered in a controlled medical setting,
and with acute or long term effects),
majoritity cause bilateral symmetric lesions that involve the deep gray nuclei with varying white matter involvent [1][2].
Posterior reversible encephalopathy syndrome (PRES):
The pathophysiology of this disease remais unknown,
but the most accepted explanation is disruption of the blood brain barrier due to a disorder of vascular autoregulation of the posterior circulation,
results in vasogenic edema in the parieto-occipital lobars [1][2][10].
CT:
NECT: hypodense parieto-occipital patchy bilateral regions (Fig.
36).
MRI:
T1: bilateral symmetric hypointense cortical and subcortical WM involvement.
T2: bilateral symmetric hyperintense cortical and subcortical WM involvement (Fig.
37-39).
T2* GRE: hypointense signal if hemorrhage.
Wernicke-Korsakoff Syndrome:
Commonly seen in alcoholics (90% of the patients in developed countries) due to vitamin B1 deficiency and the chronic toxic effects of alcohol abuse [1][2][11].
CT:
NECT: normal,
atrophy in alcoholics.
MRI:
T1 C+:enhancement of mamillary bodies.T2: hyperintensity around third ventricle,
periqueductal area,
mamillary bodies and dorsomedial thalami (Fig.
40-42).
Cerebral radiation necrosis:
Radiation induced injuries which appears months to years after a radiation treatment: it can produce vasogenic edema,
fibrosis,
necrosis and WM damage [1][2][12].
CT:
NECT: hypodense regions due to edema and symmetric WM lesions (Fig.
43).
MRI:
T1 C+: “soap bubble” or “Swiss cheese” enhancement in WM (Fig.
43-44).
T2: WM hyperintensiy (edema and demyelination) (Fig.
45-48).