Ultrasound
Ultrasound is a quick,
widely available and inexpensive method for characterising small indeterminate lesions identified on portal venous phase CT,
for example a small lesion demonstrating attenuation values between 20-40 HU which most probably represents a hyperattenuating cyst.
Focused ultrasound is particularly useful for the elderly co-morbid patient who cannot undergo contrast CT due to renal impairment,
or if the patient is unable to tolerate an MRI scan due to claustrophobia.
A lesion can be safely dismissed as a simple cyst if it is (figure 1):
- Well-circumscribed
- Anechoic
- Avascular
- Demonstrates posterior acoustic enhancement
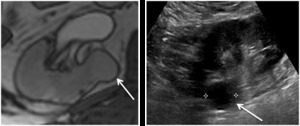
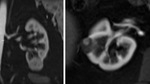
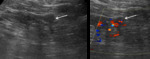
Fig. 1: Left - Axial T2 weighted image showing an incidental lesion arising medially from the left renal cortex.
Right - Subsequent focused US confirms a well circumscribed, anechoic lesion with posterior acoustic enhancement consistent with a simple cyst
Care must however be taken since papillary RCC can often demonstrate low internal echoes,
therefore appearing quite hypoechoic on ultrasound.
Solid renal masses are conspicuous only when their echogenicity is different from the adjacent renal parenchyma,
there is distortion of normal renal contour or there is abnormal vascularity on colour doppler (figure 2).

Fig. 2: Small partly exophytic RCC on ultrasound (left). Internal vascularity demonstrated on colour Doppler (right).
Whilst highly echogenic lesions are likely to represent AMLs,
they cannot be entirely differentiated from RCC on ultrasound,
therefore it is essential to confirm the presence of macroscopic fat on unenhanced CT or MRI.
Following the initial characterisation study, it is acceptable for small lesions < 2cm to be followed up by ultrasound alone (figure 3).

Fig. 3: Hyperechoic 1.8 cm interpolar lesion on ultrasound (left). Unenhanced CT demonstrates macroscopic fat (-85 HU, centre). Follow up 12 month ultrasound demonstrates no interval growth (right).
Contrast enhanced ultrasound (CEUS) with microbubble injection is an emerging tool in small renal mass characterisation and serves to identify abnormal vascularity. Although CEUS cannot differentiate malignant from benign renal masses,
for instance RCC from oncocytoma,
CEUS can reliably differentiate simple cystic lesions from solid vascularised lesions (figure 4).6 CEUS is particularly useful for assessing septal vascularity within complex cystic lesions and problem solving in cases where CT/MRI assessment of small renal lesions proves indeterminate.

Fig. 4: Mildly hyperechoic renal lesion on US (left). Avid enhancement following microbubble injection (right).
References: Dr Cherian George, University Hospital of North Midlands
Contrast enhanced CT
Multi-phase CT is the gold standard for characterisation of the small renal mass. In order to evaluate contrast enhancement,
unenhanced image acquisition should be followed by a nephrographic phase study (90–120s delay).7 A triple phase study with unenhanced,
corticomedullary and nephrographic phases will certainly enable more accurate renal lesion characterisation and differentiation of tumour subtypes,
however at the expense of additional radiation dose.
- The nephrographic phase is the optimum phase to characterise renal lesions since there is maximal and homogeneous enhancement of the normal renal parenchyma; this therefore allows the detection of renal masses which enhance differently.
An increase in attenuation (or enhancement gradient) of a lesion between unenhanced and nephrographic phase reflects the presence of solid vascularised tissue.
It is however recognised that cysts can artefactually demonstrate pseudoenhancement (up to 10 HU),
the degree of which is influenced by factors such as cyst size,
close location to renal parenchyma,
beam hardening artefact and errors in image reconstruction algorithms.8
A renal lesion that demonstrates an enhancement gradient:
- <10 HU = insignificant (pseudoenhancement)
- 10-20 HU = indeterminate
- ≥20 HU = definitive contrast enhancement (figure 5)

Fig. 5: Portal venous phase CT in a 42 year old male with abdominal pain reveals an incidental 1.2 cm left renal lesion (40 HU, left). Subsequent multiphase renal CT demonstrates enhancement gradient of 24 HU, consistent with a primary renal tumour (centre and right). Excision revealed a type I papillary RCC.
Distinct CT imaging features can allow differentiation of the various histological RCC subtypes.
For instance:
- Clear cell RCC typically appears more heterogenous and hypervascular,
with a greater tendency for cystic change and tumour necrosis.9
- Papillary RCC appears more hyperattenuating relative to renal parenchyma on unenhanced CT.
Papillary RCC also appears more homogenous with relatively reduced vascularity,
therefore demonstrating lower enhancement gradients,
more likely to fall within the indeterminate range (figure 6).

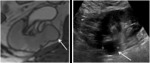
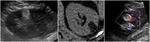
Fig. 6: Enhancement characteristics of individual tumour subtypes. Papillary RCC enhances the least
[ref - 11. Zhang J, Lefkowitz RA et al. Solid renal cortical tumors: differentiation with CT. Radiology. 2007 Aug;244(2):494-504.]
In view of this heterogeneity of imaging appearances,
the size and placement of the ‘region of interest’ (ROI) circle within a renal lesion may influence enhancement gradient measurement and therefore the ability to reliably diagnose RCC. A recent study revealed smaller ROI circles (~0.5 cm2) placed peripherally within a renal lesion had higher accuracy in differentiating RCC and papillary RCC from cysts.10
Benign lesions such as oncocytomas and AMLs of course also demonstrate variable contrast enhancement due to their inherent vascularity.
- The classic central stellate scar in oncocytoma is only seen in a small proportion of cases and oncocytoma cannot be reliably distinguished from RCC on imaging alone.
- Macroscopic fat within a renal lesion is diagnostic of an AML,
however approximately 5% of AMLs do not demonstrate macroscopic fat on CT or MRI,
otherwise known as fat poor AMLs.
- In the presence of calcification,
RCC can often demonstrate macroscopic fat due to metaplastic bone formation,
however in the absence of calcification,
macroscopic fat within RCC is extremely rare (figure 7).12

Fig. 7: Chromophobe RCC containing a small island of macroscopic fat.
Hyperdense cysts contain colloid or haemorrhagic material and therefore appear ‘bright’ on pre-contrast imaging,
with attenuation values characteristically >70 HU.
Typical appearances of hyperdense cysts are described below13:
- >70 HU on unenhanced CT
- < 3 cm in diameter
- Homogenous appearance
- Round
- Thin-walled
- Enhancement gradient < 10 HU

Fig. 8: Hyperdense Cyst. Attenuation value 75HU on unenhanced CT (left). No significant contrast enhancement on nephrographic phase, 80 HU (right).
Due to the variable composition of cyst contents (haemorrhagic material vs colloid),
attenuation values of hyperattenuating cysts on CT can be less than 70 HU; hence without an unenhanced phase to measure contrast enhancement,
it is difficult to differentiate a hyperattenuating cyst from a solid enhancing lesion on portal venous phase CT alone.
Incidental lesions with soft tissue attenuation on portal venous phase CT (20-100) therefore require careful comparison with previous available imaging or further work up with focused ultrasound,
CT or MRI (figure 9).
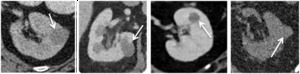
Fig. 9: From left to right:
1) Hyperattenuating cyst identified on lung cancer staging CT (42 HU), portal venous phase. No enhancement on subsequent multi-phase CT.
2) Hyperattenuating cyst identified on lymphoma staging CT (38 HU), portal venous phase.
3) Hyperattenuating lesion identified on CT Colonogram (42 HU), portal venous phase. Further MRI characterisation revealed a benign cyst.
4) Hyperattenuating cyst identified on CT KUB for renal colic (38 HU), unenhanced phase.
MRI
MRI is often very useful when:
- Renal CT characterisation is equivocal
- Patient cannot undergo contrast CT due to renal impairment or iodinated contrast allergy
- In young patients where it is important to limit radiation exposure
- For very small lesions (< 1cm) where MRI may be more accurate.
Like CEUS,
MRI is particularly useful in assessing septal thickening and enhancement in cystic renal lesions; this is of importance when there is uncertainty in differentiating Bosniak IIF from Bosniak III lesions on CT,
both of which would ensue different clinical management.
- Papillary RCC is often hypointense on T2-weighted sequences and demonstrates low-level homogenous contrast enhancement (figure 10).

Fig. 10: Type I papillary RCC demonstrating very mildly heterogenous enhancement (left). Bosniak IV cystic lesion within the contralateral kidney (right) also excised revealing papillary RCC.
- Clear cell RCC on the other hand appears hyperintense on T2-weighted sequences with intense heterogenous contrast enhancement and early wash-out.
- Oncocytoma demonstrates variable MRI signal characteristics and cannot reliably be differentiated from RCC.
- Fat suppressed images and signal loss on opposing phase sequences will identify fat within a classic AML.
Signal loss on opposing phase sequences however cannot reliably differentiate fat poor AML from RCC since clear cell RCC also contains small amounts of intracellular fat.
The presence of ‘India ink artefact’ at the interface between the lesion and renal cortex (appearing as a thick black line) can differentiate a fat poor AML from RCC.14 Both fat poor AML and papillary cell RCC demonstrate hypointense T2 signal on MRI,
however a differentiating feature but AML demonstrates rapid enhancement,
where papillary RCC shows more progressive enhancement reaching peak enhancement at a later point.15
Percutaneous Biopsy
Ultrasound or CT guided percutaneous biopsy is a safe and effective procedure with a negligible risk of tumour seeding.16 Biopsy is particularly helpful for assessing a renal lesion indeterminate after CT/MRI assessment,
particularly where clinical management would be strongly influenced.
With the emerging use of thermal ablative techniques (largely cryoablation and radiofrequency ablation),
it is essential to perform percutaneous biopsy prior to ablation in order to prevent the unnecessary treatment of histologically benign lesions.
Modern immunocytochemistry techniques are now able to reliably differentiate oncocytoma from RCC.
In view of the fact that up to 20% of small renal masses are benign and are mostly oncocytomas,
biopsy could also avoid unnecessary surgical intervention.






![Fig. 6: Enhancement characteristics of individual tumour subtypes. Papillary RCC enhances the least
[ref - 11. Zhang J, Lefkowitz RA et al. Solid renal cortical tumors: differentiation with CT. Radiology. 2007 Aug;244(2):494-504.]](https://epos.myesr.org/posterimage/esr/ecr2018/144151/media/754476?maxheight=150&maxwidth=150)