Acute pancreatitis is an acute inflammatory condition,
with a range of severity as well as various local and systemic complications.
Gallstones and alcohol are the first and second most common causes.
The revised Atlanta classification requires that two or more of the following criteria be met for the diagnosis of acute pancreatitis:
- abdominal pain suggestive of pancreatitis,
- serum amylase or lipase level greater than three times the upper normal value,
- characteristic imaging findings.
IMAGING TECHNIQUES
US: - It is indicated in an early episode of acute pancreatitis to evaluate the presence of colelithiasis/coledocolithiasis.
- The assessment of the pancreatic region is often limited by the presence of intestinal bloating;
CT: The CT should be made after 48-72 h of the onset of symptoms (the time needed to develop necrosis).
CT protocol is based on a pre-contrast scan,
followed by the injection of a bolus of contrast medium at high injection rate.
Post contrast study should include late arterial phase acquired about 40 sec from the injection start (better contrast between viable parenchyma and necrotic areas),
venous phase at 65-70 sec (for evaluating patency of the peripancreatic veins and peripancreatic regions).
According to CT findings we can classify the pancreatitis in mild,
moderate,
and severe using the CT severity index. According to this score,
the severity of pancreatitis is categorized as mild (0–2 points),
moderate (4–6 points) or severe (8–10 points).
In the 2012 revised Atlanta classification,
new definitions were included.
Acute pancreatis is divided into interstitial edematous pancreatitis (IEP) and necrotizing pancreatitis.
There are included four types of collections.
- Acute peripancreatic fluid collections (APFCs) and pseudocysts occur in IEP.
- Acute necrotic collections (ANCs) and walled-off necrosis (WON) occur in patients with necrotizing pancreatitis.
Phases of Acute Pancreatitis
Ø The early phase, occurs in the 1st week after onset,
with the disease manifesting as a systemic inflammatory response.
In the 1st week after the onset of pancreatitis,
imaging findings correlate poorly with clinical severity,
and imaging sensitivity for necrotizing pancreatitis is decreased in the first few days.
Any local complications detected in the 1st week generally do not necessitate intervention because treatment is based on supportive measures and management of organ failure.
Ø The late phase, generally starts in the 2nd week and can last for weeks to months,
and occurs only in patients with moderately severe or severe pancreatitis.
In the revised classification system acute pancreatitis is divided in IEP (intersticial edematous pancreatitis) and necrotizing pancreatitis.
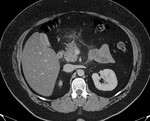
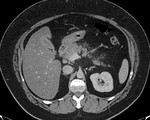
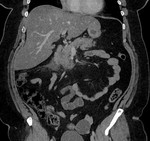
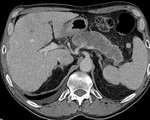
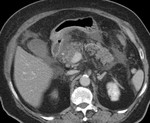
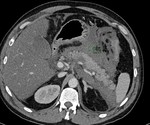
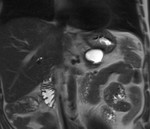
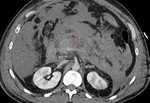
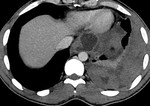
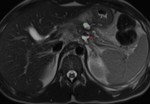
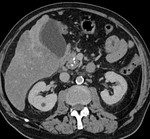
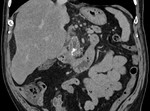
IEP: - represents nonnecrotizing inflammation of the pancreas.
- usually manifests with focal or diffuse pancreatic enlargement and is typically surrounded by wispy peripancreatic inflammation or a small amount of fluid.
- there may be surrounding fluid-containing collections.
(Fig.
1,2,3)
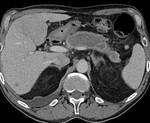
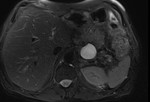
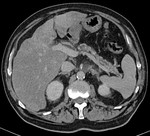
Necrotizing pancreatitis: - accounts for 5%–10% of cases of acute pancreatitis.
- necrosis may involve either the pancreatic parenchyma or the peripancreatic tissues.
- There are three subtypes of necrotizing pancreatitis based on the anatomic area of necrotic involvement:
(a) pancreatic only,
(b) peripancreatic only,
and
(c) combined pancreatic and peripancreatic. (Fig.
4,5)
PANCREATIC AND PERIPANCREATIC COLLECTIONS
The important distinctions for classifying collections correctly are the time course (< 4 weeks or >4 weeks from onset of pain) and the presence or absence of necrosis at imaging.
Acute peripancreatic fluid collections are defined as homogeneous fluid attenuation without capsulation in the first 4 weeks.
In case of heterogeneous,
variably loculated and non encapsulated fluid collections in pancreatic parenchyme or extrapancreatic area are defined as acute necrotic collection.
Once an enhancing capsule starts to develop,
persistent peripancreatic fluid collections are referred to as pseudocysts,
and acute necrotic collections are referred to as walled-off necrosis,
respectively.
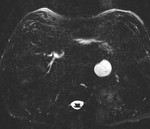
APFC (acute peripancreatic fluid collection) and Pseudocyst
APFCs
- occur during the first 4 weeks and are present only in patients with IEP.
- homogeneous fluid-attenuation collections that lack a wall and tend to conform to the retroperitoneal spaces.
- they are always peripancreatic in location.
- Most of them resolve spontaneously.
(Fig.6),
(Fig.7).
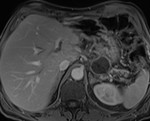
PSEUDOCYST - occur after the first 4 weeks.
- they are well-circumscribed,
homogeneous,
low-density fluid collections.
- have a capsule that manifests as an enhancing wall at CECT.
- MRI: uniformly hyperintense on T2-WI with no solid components or debris in the fluid and may have a connection to the pancreatic ductal system.
( Fig.
8,9,10,11)
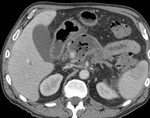
ANC (acute necrotic collection)
- collection is seen within the pancreatic parenchyma.
- occur within the first 4 weeks of symptoms onset
- poorly organized necrotic collections that occur only in necrotizing pancreatitis.
- they are often multiple,
with a loculated appearance,
and may extend inferiorly as far as the pelvic sidewalls.
- When intravenous contrast material is contraindicated,
the presence of fat attenuation within a collection at NECT is helpful for identifying necrosis and diagnosing ANCs.
WON (walled-off necrosis)
- occur after 4 weeks of symptoms onset
- a WON contains fluid and has a thick enhancing wall.
- They contain necrotic fat and/or pancreatic tissue,
which are well demonstrated at both CECT and MRI as nonliquefied debris within the fluid.
- WON may be confined to the pancreatic parenchyma but more commonly occurs in the peripancreatic space and can often occur in both locations,
with a coalescent collection extending from the lesser sac into a portion of parenchyma.
(Fig.
12).
COMPLICATIONS OF ACUTE PANCREATITIS
1. INFECTION
Any fluid collection described above can become infected,
although this complication is more common in ANC and WON.
On CECT,
there are no reliable signs of infection,
although the presence of gas within the collection should alert for this scenario.
In doubtful cases,
aspiration and culture of the fluid may be necessary.
2. HEMORRHAGIC PANCREATITIS
§ Rare and late sequelae of pancreatitis
§ Causes including erosion of pancreatic and peripancreatic vessels,
leakage or ruptured pseudoaneurysm,
diffuse bleeding in necrotic pancreas,
hemorrhagic pseudocysts
§ Need for embolization and/or surgery according to its severity (Fig.13)
3. CHRONIC PANCREATITIS
§ Irreversible inflammatory damage to morphology and function of pancreas
§ Approximately 90% of calcific pancreatitis caused by alcoholism
§ CT features : atrophy of body-tail of parenchyma,
varied enhancement due to degree of fibrosis,
dilatated main pancreatic duct with ductal calculi,
intra-and peripancreatic pseudocysts.
Splenic vein thrombosis,
thickening of peripancreatic fascia
§ MR : hypointense or signal void of calcification and intraductal calculi on fat-sat T2WI,
heterogeneous enhancement pattern of parenchyma on T1WI with enhancement.
§ US ; dilatated main pancreatic duct (MPD) with/without echogenic foci(calcifications) with posterior acoustic shadowing (Fig.16,17,18).
4. VASCULAR COMPLICATION OF ACUTE PANCREATITIS
§ Result of enzymatic digestion from pancreatic secretions or infection.
o Arterial complications encompass direct erosion into a local artery,
pseudoaneurysm formation as well as local and distant ischemic sequelae.
a) Peri-pancreatic haemorrhage
b) Pseudoaneurysm formation:
-3.5-10% of patients
- as a result of enzymatic digestion of the vessel well,
or,
when a visceral artery becomes incorporated into the wall of a pseudocyst
-Splenic artery : the most commonly affected artery (40%),
-Gastroduodenal artery : 2nd most commonly affected artery (30%)
o Vascular thrombosis
-Splenic vein thrombosis : 10-40% ( Fig.13)
-Hepatic infarction due to portal vein thrombosis
EXTRAABDOMINAL COMPLICATIONS
Fistula formation
a) Abnormal communication between the pancreas and other organs
b) Causes: leakage of pancreatic secretions from damaged pancreatic ducts or from pseudocysts
c) Most common: disruption of the pancreatic duct due to chronic pancreatitis
d) External pancreatic fistula: communication with the exterior of the body,
pancreaticocutaneous fistula
e) Internal pancreatic fistula: communication with other internal organs or space,
PPF
Extrapancreatic and extraabdominal pseudocyst
- Rarely occur in peripancreatic area extraabdominal area
- Intrasplenic pseudocyst : extrapancreatic fluid dissects along the vasculature towards the splenic hilum
- Mediastinal pseudocyst : rupture of pancreatic duct posteriorly into the retroperitoneal space
- CT : thin-walled low attenuation cystic mass extending from the body of pancreas into the mediastinum,
peripancreatic fluid collection located along the fistulous tract that dissected into the mediastinum through esophageal or aortic hiatus,
usually normal size of pancreas with features of chronic pancreatitis
- MRCP : method of choice for suspected PPF (sensitivity 80%),
identification of connection between the mediastinal and abdominal pseudocysts
- ERCP : 2nd effective modality of diagnosis for PPF,
invasive and limited when exists anatomical abnormality
- Diagnosis : elevated amylase level in the aspirated fluid (Fig.
14,15)