In 2011 the U.S.
Food and Drug Administration approved the first immune checkpoint inhibitor (ICI) for cancer care.
Subsequently there was a rapid expansion of ICIs in clinical use to such an extent that these agents are now standard therapies for several malignancies.
Since immunotherapy with ICIs has only recently been introduced in clinical practice,
radiologists are not familiar with the radiological signs of irAEs.
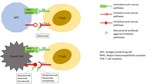
Mechanism of action
The tumor microenvironment can overexpress molecules capable of inhibiting the T-cell–mediated immune response.
Central to this process are three molecules:
- cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4)
- programmed death 1 (PD-1) and
- his ligand PD-L1.
The basic principle of ICIs is to block the CTLA-4 and the PD-1/PD-L1 pathways in order to enhance T-cell action against the cancer cells (Figure 1).
By unbalancing the immune system,
ICIs may cause dysimmune toxicities also called irAEs.
The latter can potentially affect any tissue,
but most frequently involve skin,
colon,
endocrine organs,
liver,
and lungs.
Many of these toxicities are associated with radiologic manifestations.
Current indications for ICI treatment
To date,
seven ICIs have been approved by the United States Food and Drug Administration (US FDA) for the treatment of various tumours at different stages: one anti–CTLA-4 antibody (Ipilimumab),
three anti–PD-1 antibodies (Pembrolizumab,
Nivolumab,
Cemiplimab),
and three anti–PD-L1 antibodies (Atezolizumab,
Avelumab,
and Durvalumab) (Figure 2).
Grading scale for irAEs
The grading scale for irAEs is based on the Common Terminology Criteria for Adverse Events (CTCAE).
IrAEs are very common for all ICIs and across different tumor types: events of any grade happen in about 90% of patients treated with anti–CTLA-4 ipilimumab and approximately 70% of those treated with any anti–PD-1 or anti–PD-L1 antibody.
For combined ICI treatment the rates of grades 3 and 4 irAEs are significantly higher.
Management of irAEs
Management of irAEs is based on clinical judgement and follows the CTCAE guidelines.
As a general strategy,
ICIs are interrupted in grade 2.
For grade 3 and 4,
high dose steroids are given.